US Pharm. 2011;36(9):HS-13-HS-16.
Preterm birth, or the birth of an infant of less than 37 weeks’ gestation, is a leading cause of neonatal mortality in the United States.1-3 Nearly half of all preterm births are preceded by preterm labor, but preterm birth may also occur following membrane rupture or in the presence of other medical conditions.1-4 Preterm labor is regular contractions that occur prior to 37 weeks’ gestation and are associated with cervical changes.1,3 The causes of preterm labor are unclear, but the consequences are well defined. Preterm birth is responsible for 35% of health care spending for infants and 10% of spending for children in the U.S.1 Nearly 500,000 preterm births occur annually in the U.S., accounting for nearly 13% of live births.1,2,4
Significance of Preterm Labor and Birth
Preterm birth is responsible for three-quarters of cases of neonatal mortality and one-half of cases of neurologic impairment in children.1,2,5 The risk of long-term neurodevelopmental and medical disabilities is high among children born preterm.6 Gestational age is inversely proportional to the risk of neonatal morbidity and mortality.1 Infant mortality is significantly higher among neonates born before 32 weeks’ gestation than in those born later.6 Birth prior to 32 weeks’ gestation accounts for the most neonatal deaths and the most long-term morbidity.2 However, neonates born between 34 and 36 weeks’ gestation constitute the majority of neonatal ICU (NICU) admissions.6
Despite advances in obstetric and neonatal care, the incidence of preterm birth continues to increase.1 The rate of preterm birth has risen 20% in the last two decades, with most births occurring between 34 and 36 weeks’ gestation.6 This may be partly due to greater numbers of indicated preterm births attributed to maternal conditions and of multiple gestations following assisted conception.2
Risk Factors for Preterm Labor and Birth
The causes of preterm labor and birth are unclear but likely are influenced by a combination of genetic and environmental factors.3,6 Risk factors for preterm birth include nonwhite race, short interpregnancy interval, maternal physical or emotional stress, and maternal thinness. Previous preterm birth is a significant risk factor for a subsequent preterm birth.2,6 Other medical conditions, such as bacterial vaginosis, intrauterine infection, nongenital tract infection, maternal abdominal surgery, maternal endocrine disorders, multiple gestation, periodontal disease, cervical length shorter than 3 cm, tobacco use, uterine abnormalities, placenta previa, and placental abruption, contribute to the onset of preterm labor and preterm birth.3,6
Of preterm births, roughly half occur after spontaneous labor with intact membranes, one-quarter occur after preterm premature rupture of membranes (PPROM), and one-quarter are iatrogenic.2,6 PPROM may be caused by infection or inflammation, vascular disease, uterine overdistention, or immunologic disorders. Iatrogenic factors include elective preterm delivery for gestational hypertension, intrauterine growth restriction, placental abruption, and nonreassuring fetal status.2,6
Assessment of Preterm Labor
Identifying women at risk for preterm birth is difficult. Several decision tools and clinical nomograms for predicting the risks of preterm labor and preterm birth are available, but most lack accuracy and precision.1,7 The most useful tools for predicting risk are ultrasonography to determine cervical length and fetal fibronectin testing.1 A shortened cervix and increased concentrations of fibronectin—a protein found in cervical and vaginal secretions—are strong predictors of preterm labor and birth.2,3
The decision to initiate pharmacologic therapy for preterm labor involves several factors: the probability of progressive labor, gestational age, and the maternal and fetal or neonatal risks associated with treatment. In general, the decision to intervene with pharmacologic management is made when regular uterine contractions do not diminish with bed rest or hydration, when cervical changes occur, or in the case of cervical dilation upon presentation.1
Management of Preterm Labor
Preventing preterm birth is a major challenge for modern obstetric medicine.6 Spontaneous preterm labor often occurs without warning or previous indication, but the goal of preterm labor management is to prolong pregnancy as safely as possible to permit fetal development and maturation.3
Corticosteroids: One of the most useful tools in the obstetric armamentarium for reducing morbidity and mortality related to preterm birth, corticosteroids are the best-documented beneficial agents for use in preterm labor.1 The use of corticosteroids is indicated to prevent preterm birth in women between 26 and 34 weeks’ gestation who are at risk for preterm birth owing to preterm labor, PPROM, or severe preeclampsia or other medical conditions that necessitate preterm delivery.2
In combination with delivery in a facility with a level III NICU, corticosteroids consistently improve outcomes of preterm birth.6 Corticosteroids administered as late as 12 to 24 hours before preterm birth reduce the incidence of neonatal complications after preterm labor, including respiratory distress syndrome (RDS), intraventricular hemorrhage, and necrotizing enterocolitis.1-3,8 Administered prior to 35 weeks’ gestation, corticosteroids activate the hypothalamic-pituitary-adrenal axis to enhance functional maturity in the fetus, specifically lung maturity.9
The American College of Obstetricians and Gynecologists (ACOG) recommends either betamethasone or dexamethasone to promote fetal lung maturity.1,2 Intramuscular administration of betamethasone 12 mg every 24 hours for two doses or dexamethasone 6 mg every 12 hours for four doses is indicated for women at risk for preterm birth between 26 and 34 weeks’ gestation.1,3 Repeat doses of corticosteroids are not beneficial for improving outcomes following preterm birth.3
Antibiotics: Preterm labor is often associated with infections and inflammation, and subclinical infection is associated with PPROM.1,10 However, most studies have not found that prophylactic antibiotic treatment confers a substantial benefit in preventing preterm labor or birth. Additionally, antibiotics do not reduce the occurrence of neonatal RDS or sepsis.1
Antibiotics do not affect preterm labor in patients with intact membranes.6 Among women with PPROM, antibiotics may reduce the risk of preterm birth within 48 hours of administration, but little evidence of benefit exists for other outcomes. Specifically, antibiotic administration may delay delivery after PPROM, which provides time for corticosteroid administration.2,10 Amoxicillin-clavulanate should be avoided in women who are at risk for preterm birth because of the increased chance of neonatal necrotizing enterocolitis.10 The ACOG does not support the use of antibiotics for pregnancy prolongation in women with intact membranes.11
Bacterial vaginosis, an overgrowth of anaerobic bacteria, is associated with an increased risk of preterm birth; treatment of the vaginosis reduces the risk.2 Symptomatic bacterial vaginosis is often treated with oral clindamycin 300 mg twice daily for 7 days, metronidazole 500 mg twice daily for 7 days, or metronidazole 250 mg three times daily for 7 days.6
Antibiotics are appropriate during preterm labor as prophylaxis against group B streptococcus (GBS) sepsis.1 Current guidelines consider IV administration of one dose of ampicillin 2 g followed by ampicillin 1 g every 6 hours for 48 hours to be adequate GBS prophylaxis in women who have tested positive for GBS, had a previous infant with GBS disease, had GBS bacteriuria during the current pregnancy, or whose GBS status is unknown.12
Tocolytics: Tocolytic drugs inhibit uterine contractions.1,2 Four main classes of tocolytics, with varying degrees of safety and effectiveness, are used: beta-adrenergic agonists, magnesium sulfate, calcium channel blockers, and nonsteroidal anti-inflammatory drugs (NSAIDs).1,3 General contraindications to tocolysis include severe preeclampsia, maternal instability, placental abruption, intrauterine infection, lethal congenital or chromosomal abnormalities, advanced cervical dilation, fetal compromise or distress, and placental insufficiency.1,6 The choice of tocolytic should be based on maternal condition, potential adverse effects, gestational age, and cost.1,13 Once treatment is initiated, the patient’s response to tocolysis, including adverse effects, should be continuously monitored.13 Prolonged use of tocolytic agents is not recommended.1
Despite widespread use, tocolysis lacks robust evidence for the prevention of preterm birth. Most tocolytic drugs prolong gestation for 2 to 7 days.1,2,6,14 This delay in preterm birth allows sufficient time for the administration of corticosteroids and maternal transfer to a hospital with an appropriate NICU, if such interventions are warranted.1,4,6,13 Tocolytic agents potentiate the effects of corticosteroids, and the concomitant use of tocolytics and corticosteroids is widespread.6,9 Tocolytic therapy offers no benefit in neonatal RDS or mortality.14 TABLE 1 provides a summary of common tocolytics and their doses, administration, and adverse effects.1,2,6,15
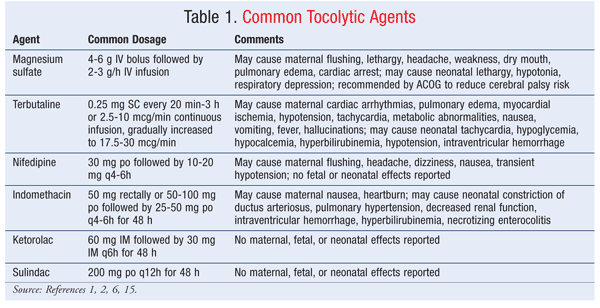
Magnesium sulfate, one of the most common obstetric drugs in the U.S., is used primarily for seizure prophylaxis in preeclampsia.16 Despite its lack of proven efficacy, magnesium sulfate is also the most commonly used tocolytic agent in the U.S.4,13 Magnesium sulfate can cause maternal lethargy, drowsiness, double vision, nausea, and vomiting.1 More serious maternal adverse effects include pulmonary edema, hypotension, muscle paralysis, tetany, cardiac arrest, and respiratory depression.3 Magnesium sulfate can cause fetal toxicity at high doses.3,16
While most tocolytics do not show a benefit in neonatal outcomes, magnesium sulfate has gained attention for its use in preventing cerebral palsy. Several studies have shown that, among women at risk for preterm birth, low-dose administration of magnesium sulfate reduced the risk of cerebral palsy among surviving neonates.16-18 The neuroprotective effect of magnesium sulfate results from a reduction in vascular instability and prevention of hypoxic and amino acid damage.17 The ACOG supports the administration of magnesium sulfate prior to anticipated preterm birth to reduce the risk of cerebral palsy.15 The recommended dose of magnesium sulfate is an IV bolus of 4 g to 6 g followed by 2 g to 3 g per hour.1
Terbutaline, a beta-adrenergic agonist, is a potent cardiovascular (CV) stimulant that is associated with an increased risk of pulmonary edema and maternal and fetal CV abnormalities. A bronchodilator, it is approved to prevent and treat bronchospasms associated with asthma, bronchitis, and emphysema.19 The ACOG does not support the use of terbutaline to prevent preterm labor but states that, when it is used as a tocolytic, terbutaline should be administered as a subcutaneous 0.25-mg dose every 20 minutes to 3 hours.1 As a continuous infusion for tocolysis, terbutaline is initiated at a rate of 2.5 mcg/min to 10 mcg/min, and the rate can be increased gradually every 10 to 20 minutes to a maximum of 17.5 mcg/min to 30 mcg/min.20
Terbutaline is not FDA approved as a tocolytic agent. In February 2011, the FDA released a safety announcement advising that terbutaline be used for tocolysis no longer than 48 to 72 hours owing to the risk of serious maternal CV problems that could lead to death. The FDA required that a boxed warning and contraindication be added to the labeling of both injectable and oral terbutaline. The FDA acknowledged that clinicians may continue to use terbutaline on an off-label basis to prevent preterm birth in urgent situations, but warned that the drug should never be used in the outpatient setting. The FDA’s statement was based on postmarketing surveillance data that included 16 reports of maternal death and 12 cases of maternal CV events following terbutaline administration since the drug’s approval in 1976.19
Nifedipine, a calcium channel blocker, reduces the risk of preterm delivery within 7 days of treatment prior to 34 weeks’ gestation. Compared with beta-agonists, nifedipine also lowers the risk of RDS, necrotizing enterocolitis, intraventricular hemorrhage, neonatal jaundice, and NICU admission.21 Nifedipine poses few maternal or fetal risks, but its use with magnesium sulfate has led to CV collapse in some pregnant women. For tocolysis, the ACOG recommends a loading dose of nifedipine 30 mg administered orally followed by 10 mg to 20 mg every 4 to 6 hours.1
NSAIDs act as tocolytic agents by blocking the inflammatory process that triggers labor.2 NSAIDs have few maternal adverse effects, but they are associated with oligohydramnios and premature closure of the ductus arteriosis in the fetus.1,2 Also, neonates of mothers treated with NSAIDs may experience intraventricular hemorrhage, necrotizing enterocolitis, and bronchopulmonary dysplasia.2 These adverse effects are more common when NSAIDs are used after 32 weeks’ gestation, and NSAIDs are generally reserved for tocolysis earlier in pregnancy.2,14
The most commonly used NSAID for tocolysis is indomethacin. According to practice guidelines, a loading dose of 50 mg rectally or 50 mg to 100 mg orally followed by 25 to 50 mg orally every 4 to 6 hours for 48 hours is used for tocolysis.1,6 Ketorolac, also used as a tocolytic, is administered intramuscularly as a 60-mg loading dose followed by 30 mg every 6 hours for 48 hours. As a tocolytic, sulindac is administered at a dose of 200 mg orally every 12 hours for 48 hours.1
Progesterone: Maternal progesterone declines before labor, and low maternal progesterone levels are associated with miscarriage and preterm labor.2,3 Exogenous progesterone supplementation is indicated for women at risk for preterm labor and birth.2 Specifically, progesterone lowers the risk of preterm labor and birth by maintaining uterine inactivity.6 Administered as weekly intramuscular injections of 250 mg of 17-alpha-hydroxyprogesterone caproate (17-OHPC), progesterone reduces the risk of recurrent preterm birth by up to 50% when begun between 16 and 20 weeks’ gestation.2,6,22,23 Women with multiple gestations do not benefit from 17-OHPC treatment, and 17-OHPC does not prolong pregnancy in women with PPROM.6,24
Natural progesterone vaginal suppositories reduce the risk of preterm birth by up to 45% and decrease the incidence of respiratory distress and neonatal morbidity and mortality in pregnant women with a shortened cervix.6,23 In this situation, progesterone is commonly delivered at a dose of 100 mg daily, beginning between 16 and 24 weeks’ gestation and continuing through 34 to 36 weeks’ gestation.6,20,23 The ACOG supports the use of progesterone for prolonging pregnancy only in women with intact membranes and a history of spontaneous birth prior to 37 weeks’ gestation.6,24
Although several interventions for managing preterm labor and preventing preterm birth are available, most are not universally effective, offering benefits in only a specific group of women or presenting safety concerns for the mother or fetus.2 Large-scale research is needed to conclusively define the best choices for managing preterm labor and preventing preterm birth.3
The Role of the Pharmacist
Providing pharmacologic care to women during pregnancy can be rewarding, but it is often challenging. The benefits of drug therapy for managing preterm labor and preventing preterm birth must be weighed against the risks associated with such treatment. The health of the pregnant woman, as well as the health of the fetus, must be taken into account when drug therapy is being considered. Pharmacists must remain up-to-date on current clinical guidelines and the use of drugs during pregnancy in order to ensure healthy outcomes for mother and child. A collaborative approach between the obstetrician, neonatologist, pharmacist, and other health care providers is essential to reduce the burdens of preterm labor and birth.3
REFERENCES
1. ACOG Committee on Practice Bulletins. ACOG Practice Bulletin. Clinical management guidelines for obstetrician-gynecologist. Number 43, May 2003. Management of preterm labor. Obstet Gynecol. 2003;101:1039-1047.
2. Denney JM, Culhane JF, Goldenberg RL. Prevention of preterm birth. Womens Health (Lond Engl). 2008;4:625-638.
3. DiPiro JP, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 7th ed. New York, NY: McGraw-Hill Medical; 2008.
4. Mercer BM, Merlino AA. Magnesium sulfate for preterm labor and preterm birth. Obstet Gynecol. 2009;114:650-668.
5. Sanu O, Lamont RF. Periodontal disease and bacterial vaginosis as genetic and environmental markers for the risk of spontaneous preterm labor and preterm birth. J Matern Fetal Neonatal Med. Epub Jan 24, 2011.
6. Sayres WG Jr. Preterm labor. Am Fam Physician. 2010;81:477-484.
7. Allouche M, Huissoud C, Guyard-Boileau B, et al. Development and validation of nomograms for predicting preterm delivery. Am J Obstet Gynecol. 2011;204:242.e1-8.
8. Onland W, de Laat MW, Mol BW, Offringa M. Effects of antenatal corticosteroids given prior to 26 weeks’ gestation: a systematic review of randomized controlled trials. Am J Perinatol. 2011;28:33-44.
9. Hallman M, Peltoniemi O, Kari MA. Enhancing functional maturity before preterm birth. Neonatology. 2010;97:373-378.
10. Kenyon S, Boulvain M, Neilson JP. Antibiotics for preterm rupture of membranes. Cochrane Database Syst Rev. 2010;(8):CD001058.
11. ACOG Committee opinion no. 445: antibiotics for preterm labor. Obstet Gynecol. 2009;114:1159-1160.
12. Verani JR, McGee L, Schrag SJ. Prevention of perinatal group B streptococcal disease—revised guidelines from CDC, 2010. MMWR Recomm Rep. 2010;59:1-36.
13. Blumenfeld YJ, Lyell DJ. Prematurity prevention: the role of acute tocolysis. Curr Opin Obstet Gynecol. 2009;21:136-141.
14. Haas DM, Imperiale TF, Kirkpatrick PR, et al. Tocolytic therapy: a meta-analysis and decision analysis. Obstet Gynecol. 2009;113:585-594.
15. ACOG Committee opinion no. 455: magnesium sulfate before anticipated preterm birth for neuroprotection. Obstet Gynecol. 2010;115:669-671.
16. Pryde PG, Mittendorf R. Contemporary usage of obstetric magnesium sulfate: indication, contraindication, and relevance of dose. Obstet Gynecol. 2009;114:669-673.
17. Rouse DJ, Hirtz DG, Thom E, et al. A randomized, controlled trial of magnesium sulfate for the prevention of cerebral palsy. N Engl J Med. 2008;359:895-905.
18. Crowther CA, Hiller JE, Doyle LW. Magnesium sulphate for preventing preterm birth in threatened preterm labour. Cochrane Database Syst Rev. 2002;(4):CD001060.
19. FDA drug safety communication: new warnings against the use of terbutaline to treat preterm labor. www.fda.gov/Drugs/DrugSafety/
20. Lacy CF, Armstrong LL, Goldman MP, Lance LL. Drug Information Handbook. 18th ed. Hudson, OH: Lexi-Comp, Inc; 2009.
21. Conde-Agudelo A, Romero R, Kusanovic JP. Nifedipine in the management of preterm labor: a systematic review and metaanalysis. Am J Obstet Gynecol. 2011;204:134.e1-20.
22. Mason MV, Poole-Yaeger A, Krueger CR, et al. Impact of 17P usage on NICU admissions in a managed Medicaid population—a five-year review. Manag Care. 2010;19:46-52.
23. Hassan SS, Romero R, Vidyadhari D, et al. Vaginal progesterone reduces the rate of preterm birth in women with a sonographic short cervix: a multicenter, randomized, double-blind, placebo-controlled trial. Ultrasound Obstet Gynecol. 2011;38:18-31.
24. ACOG Committee opinion no. 419 October 2008 (replaces no. 291, November 2003): upse of progesterone to reduce preterm birth. Obstet Gynecol. 2008;112:963-965.
To comment on this article, contact rdavidson@uspharmacist.com.





