US Pharm. 2014;39(4):HS2-HS7.
ABSTRACT: The management of hydrocephalus, the
accumulation of cerebrospinal fluid (CSF) in the ventricles of the
brain, often includes shunt implantation to prevent long-term effects.
Shunts are drainage devices designed to sustain intracranial pressure
and prevent further CSF accumulation, but they can introduce pathogens
into the central nervous system. This can lead to infections and
sequelae, such as seizures, shunt failure, meningitis, sepsis, and
encephalitis. Infections may be prevented by using aseptic techniques,
chemoprophylaxis, and/or antibiotic-eluting shunts. Treatment of a shunt
infection may include removal of the infected hardware, placement of a
drainage device, and use of IV or intraventricular antibiotics.
Hydrocephalus is a condition characterized by accumulation of cerebrospinal fluid (CSF) in the ventricles or subarachnoid space in the brain, leading to increased intraventricular pressure and ventricular dilation. A common etiology of hydrocephalus is impairment of CSF drainage.1,2 The occurrence of hydrocephalus in the United States and Europe has been estimated at 0.5 to 0.8 per 1,000 births and is often secondary to spina bifida, intracranial hemorrhage, or congenital etiology.1,3 Other potential causes of hydrocephalus include myelodysplasia, tumors, meningitis, posterior fossa cyst, Dandy-Walker malformation, or aqueductal stenosis.4
Mechanical shunting is the primary treatment for hydrocephalus. Hydrocephalus-related shunt placements are the most common neurosurgical procedure performed in children, but they also have the highest failure rate.5 Over 40,000 shunt placements are performed annually in the U.S., with an estimated inpatient mortality rate of 2.7%.5,6 Introduction of common skin flora and other pathogens, both perioperatively and postoperatively, can lead to shunt infections and result in shunt failure.
The rate of shunt infections is estimated at 1.6% to 16.7% in adult patients but up to 20% in pediatric patients.2,7 This increased occurrence in the pediatric population is thought to be due to lack of immunity (particularly <1 year of age), prematurity (<35 weeks’ gestational age), exposure to potential pathogens, concomitant morbidities, and other physiological factors.3,4 Pediatric patients tend to develop shunt infections sooner than adults, with most infections occurring within the first 30 to 60 days post placement.1
Patients who are <5 years of age and adults aged 50 to 59 years appear to have the greatest incidence of shunt infections.1 Risk factors for shunt infection may include younger age, postoperative CSF leak, intraventricular hemorrhage, multiple shunts, shunt revision surgery in the past 90 days, shunts placed immediately post meningitis, prolonged hospital stay (>1 month), and previous shunt infection.2,4,7,8 Complications related to shunt infections include shunt malfunctions, short- or long-term cognitive impairment, sepsis, meningitis, and lengthened hospital stay.1,4,8
Shunt Types
The most common types of shunts are summarized in TABLE 1. This article will focus on the two most utilized shunts: ventriculoperitoneal (VP) and ventriculoatrial (VA) shunts. VP shunts, the more common of the two, work by diverting CSF to the peritoneum, whereas VA shunts shift the CSF to the atrium of the heart.2,9,10

Pathogenesis of Shunt Infections
Shunt contamination is often caused when the proximal end of the shunt comes in contact with normal skin flora. This can occur postoperatively due to a wound infection or perioperatively during surgical implantation.11 Gram-positive bacteria, such as coagulase-negative Staphylococcus epidermidis and Staphylococcus aureus, are responsible for 45% to 60%1-3,12 and 20% to 33%2,3 (with one report up to 80%4) of shunt infections, respectively. These pathogens are closely associated with early shunt infections (occurring within 4 weeks of shunt surgery).2
Late shunt infections can occur several months after shunt surgery and account for approximately 15% of shunt-related infections.3 In many cases, these late infections can be caused by either gram-positive bacteria, like Streptococcus or Enterococcus species, or gram-negative bacteria, most commonly Pseudomonas aeruginosa, if the patient has experienced a bowel perforation or peritonitis (primarily seen in VP shunts).2 The most common pathogens associated with shunt infections are summarized in TABLE 2.1-4,11 Infections increase the likelihood of shunt-related complications and failures.

Prevention of Shunt Infections
Methods employed to prevent a shunt infection include using sterile technique, systemic antibiotics, and antibiotic-impregnated catheters. Aseptic surgical technique has been shown to help reduce the risk of shunt infections. A standardized surgical protocol, including preoperative washing of the patient’s hair with chlorhexidine shampoo, double gloving by all members of the surgical team, and the use of antibiotic-impregnated shunts (AIS), that was implemented by four children’s hospitals demonstrated an infection reduction rate from 8.8% to 5.7%.13
Systemic prophylactic antibiotics may be utilized during the procedure and in the initial 24 hours after shunt placement. According to a meta-analysis, the use of systemic, prophylactic antibiotics and antibiotic-impregnated catheters reduced the rate of shunt infections.9 This same study compared use of systemic, prophylactic antibiotics versus placebo for the reduction of shunt infection and found that the antibiotics decreased the rates of shunt infections (odds ratio [OR], 0.52).9 While the most appropriate prophylactic antibiotic for shunt placement was not well defined in the past, most clinicians use IV cefazolin or vancomycin for surgical prophylaxis.2,14 Recent guidelines recommend cefazolin for neurosurgical fluid-shunting procedures, with clindamycin and vancomycin as alternatives for allergic patients.15
AIS catheters have commonly been used to prevent shunt infections since their introduction in 2002.16 Most devices are impregnated with a combination of rifampicin and clindamycin.4,16,17 They are designed to target the colonization of gram-positive bacteria that produce a glycoprotein biofilm that adheres to the catheter surface.16 One meta-analysis looked at the use of antibiotic-impregnated catheters versus standard catheters and found that the use of antibiotic-impregnated catheters was associated with decreased rates of shunt infection (OR, 0.21). It was concluded in this meta-analysis that the use of both systemic antibiotic prophylaxis and antibiotic-impregnated catheters could be beneficial in the reduction of shunt infections.9
A retrospective trial was also conducted in pediatric patients who had undergone shunt insertion during a 3-year time frame. For the first 18 months, patients received nonantibiotic-impregnated catheters; for the next 18 months, any new patient requiring shunt placement received antibiotic-impregnated catheters. All children were followed for 6 months after shunt placement. The children with nonantibiotic-impregnated catheters had an infection rate of 12%, whereas infection was seen in only 1.4% of the children with antibiotic-impregnated catheters.18 A more recent review of 15 trials also concluded that antibiotic-coated catheters reduce the clinical infection rate, particularly in patients who have long-term catheters, which was different from some previous analyses.19
Preventing shunt infections through prophylaxis and the use of AIS catheters has been shown to reduce cost of patient care.20 It is estimated that the cost to treat a shunt infection can range from $17,300 to $48,454 per patient, leading to an estimated annual hospital cost in the U.S. of $2 billion.16,17 Although each AIS catheter costs an average of $400, the amount of money saved by preventing infection and/or reinfection can provide significant long-term savings.16 These savings are due to a 2.4-fold decreased likelihood of infection and decreased time in the hospital as a result of infection. It has been reported that these savings would amount to over $440,000 per 100 patients shunted.17
Diagnosing Shunt Infections
General shunt infection symptoms include headache, nausea, vomiting, inflammation of the surgical site, neurologic abnormality, fever, and seizure.2,3 Patient-specific clinical manifestations of shunt infections depend on the type of shunt implanted. VP shunts can be associated with peritonitis, anorexia, irritability, and abdominal pain,8 while VA shunts can be associated with sepsis and endocarditis.2,3 If clinical manifestations such as altered mental status, erythema surrounding the shunt site, fluid along the shunt track, purulent discharge, papilledema, bulging fontanel, nuchal rigidity, or obvious CSF leakage are seen, the patient should be evaluated with CSF analysis, blood cultures, and cerebral imaging.8
CSF analysis predictive for infection includes increased white blood cell count, increased protein concentrations, and decreased glucose levels; the CSF should also be cultured and gram-stained. Blood cultures are more useful indicators of VA shunt infections as opposed to VP shunt infections.11 Cerebral imaging can be used to identify ventriculitis or evidence of CSF obstruction. Abdominal imaging may also be used to identify VP shunt malfunction.
Treatment Options
Currently, there are no practice guidelines for the treatment of shunt infections; however, the removal of the infected hardware, placement of an external ventricular drain, cultures, and treatment with IV or intraventricular anti-biotics are all shown to be part of an effective management process.2
Current recommendations for the empirical treatment of central nervous system (CNS) infection in the presence of a shunt recommend using IV vancomycin in combination with an agent that has adequate gram-negative coverage, such as cefepime, ceftazidime, cefotaxime, or meropenem. The ability of a medication to penetrate the CSF as well as the activity of the antibiotic against the bacterial biofilm are also important to consider for antibiotic choice.21 Such agents should be administered until the pathogen is identified and definitive treatment determined.22,23 In a VP shunt, the distal tip of the shunt lies within the peritoneal cavity and is more susceptible to gram-negative bacteria; gentamicin may be added for synergy.2 Empirical antimicrobial therapy and dosing recommendations for shunt infections are summarized in TABLE 3.2,18,22,23
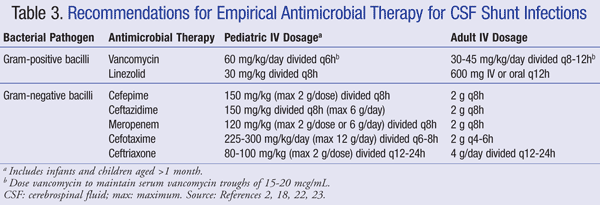
For patients refractory to vancomycin therapy, linezolid 10 mg/kg every 8 hours has been shown to be effective as monotherapy in pediatric patients.21 The addition of rifampin as adjunctive therapy may also be considered due to its penetration into the CNS.24 The length of antibiotic therapy depends largely on the surgical approach used, the type of shunt, and the pathogen involved, with one study reporting a duration of therapy range of 4 to 47 days.25,26 More specifically, 7 to 10 days has been suggested for treatment duration,23 with a longer course (10-14 days) recommended for gram-negative infections.2
In the case of complicated or treatment-resistant shunt infections, clinicians should consider intrathecal or intraventricular administration of antibiotics for increased efficacy due to their ability to achieve higher bactericidal concentrations within the CNS.23 Vancomycin and gentamicin are commonly used in this situation; however, there are no definitive recommendations on their use.22,23
Fungal Shunt Infections
A fungal shunt infection is considered to be rare, with one study reporting an incidence rate of 0.01%.27 The most common pathogens responsible for a fungal shunt infection are Candida species (including C albicans, C parapsilosis, and C tropicalis), which cause 75% of fungal cases. Other organisms include Histoplasmosis species, Cryptococcus neoformans, and Aspergillus species. The primary treatment for fungal shunt infections is liposomal amphotericin B 3 to 5 mg/kg/day for at least 12 weeks.27 For patients intolerant of amphotericin B, 5-flucytosine may be an alternative.28 This is often followed by treatment with fluconazole 600 to 800 mg/day or itraconazole 200 mg two to three times daily for up to 1 year in immunocompetent patients and potentially life-long therapy in patients who are immunocompromised.27
Conclusion
Shunt infections are a risk factor associated with hydrocephalus treatment. Infection prevention has been demonstrated with empirical antibiotics and the use of AIS catheters, combined with proper aseptic technique, to significantly decrease the rate of infection and reduce the risk of comorbid sequelae. There are currently no guidelines for the treatment of shunt infections; however, IV empirical therapy involving both gram-positive and gram-negative coverage, such as vancomycin and a third-generation cephalosporin, would be recommended. Intraventricular antibiotics may also be needed for maximal bactericidal CNS concentration.
REFERENCES
1. Langley JM, Gravel D, Moore D, et al. Study of
cerebrospinal fluid shunt-associated infections in the first year
following placement, by the Canadian Nosocomial Infection Surveillance. Infect Control Hospital Epidemiol. 2009;30:285-288.
2. Wells DL, Allen JM. Ventriculoperitoneal shunt infections in adult patients. AACN Adv Crit Care. 2013;24:6-12.
3. Lee JK, Seok JY, Lee JH, et al. Incidence and risk
factors of ventriculoperitoneal shunt infections in children: a study of
333 consecutive shunts in 6 years. J Korean Med Sci. 2012;27:1563-1568.
4. Parker SL, Attenello FJ, Sciubba DM, et al. Comparison
of shunt infection incidence in high-risk subgroups receiving
antibiotic-impregnated versus standard shunts. Childs Nerv Syst. 2009;25:77-83.
5. Treatment of hydrocephalus. Hydrocephalus Association.
www.hydroassoc.org/hydrocephalus-education-and-support/learning-about-hydrocephalus/treatment-of-hydrocephalus/.
Accessed February 11, 2014.
6. Patwardhan RV, Nanda A. Implanted ventricular shunts in
the United States: the billion-dollar-a-year cost of hydrocephalus
treatment. Neurosurgery. 2005;56:139-144.
7. Dallacasa P, Dappozzo A, Galassi E, et al. Cerebrospinal fluid shunt infections in infants. Childs Nerv Syst. 1995;11:643-648.
8. Rogers EA, Kimia A, Madsen JR, et al. Predictors of
ventricular shunt infection among children presenting to a pediatric
emergency department. Pediatr Emerg Care. 2012;28:405-409.
9. Ratilal BO, Costa J, Sampaio C. Antibiotic prophylaxis for surgical introduction of intracranial ventricular shunts. Cochrane Database Syst Rev. 2006;(3):CD005365.
10. Shunt systems. Hydrocephalus Association.
www.hydroassoc.org/hydrocephalus-education-and-support/learning-about-hydrocephalus/shunts.
Accessed February 12, 2014.
11. Forward KR, Fewer HD, Stiver HG. Cerebrospinal fluid shunt infection. A review of 35 infections in 32 patients. J Neurosurg. 1983;59:389-394.
12. Lan CC, Wong TT, Chen SJ, et al. Early diagnosis of
ventriculoperitoneal shunt infections and malfunctions in children with
hydrocephalus. J Microbiol Immunol Infect. 2003;36:47-50.
13. Kestle JR, Riva-Cambrin J, Wellons JC, et al. A
standardized protocol to reduce cerebrospinal fluid shunt infection: the
Hydrocephalus Clinical Research Network Quality Improvement Initiative.
J Neurosurg Pediatr. 2011;8:22-29.
14. Shah SS, Sinkowitz-Cochran RL, Keyserling HL, et al. Vancomycin use in pediatric neurosurgery patients. Am J Infect Control. 1999;27:482-487.
15. Bratzler DW, Dellinger EP, Olsen KM, et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Am J Health-Syst Pharm. 2013;70:195-283.
16. Parker SL, Anderson WN, Lilienfeld S, et al.
Cerebrospinal shunt infection in patients receiving
antibiotic-impregnated versus standard shunts. J Neurosurg Pediatr. 2011;8:259-265.
17. Attenello F, Garces-Ambrossi G, Zaidi H, et al.
Hospital costs associated with shunt infections in patients receiving
antibiotic-impregnated shunt catheters versus standard shunt catheters. Neurosurgery. 2010;66:284-289.
18. Sciubba DM, Stuart RM, McGirt MJ, et al. Effect of
antibiotic-impregnated shunt catheters in decreasing the incidence of
shunt infection in the treatment of hydrocephalus. J Neurosurg. 2005;103(suppl):131-136.
19. Babu MA, Patel R, Marsh WR, et al. Strategies to
decrease the risk of ventricular catheter infections: a review of the
evidence. Neurocrit Care. 2012;16:194-202.
20. Bratzler DW, Dellinger EP, Olsen KM, et al. Clinical practice guidelines for antimicrobial prophylaxis in surgery. Surg Infect (Larchmt). 2013;14:73-156.
21. Yilmaz A, Dalgic N, Musluman M, et al. Linezolid treatment of shunt-related cerebrospinal fluid infections in children. J Neurosurg Pediatr. 2010;5:443-448.
22. Tunkel AR, Hartman BJ, Kaplan SL, et al. Practice guidelines for the management of bacterial meningitis. Clin Infect Dis. 2004;39:1267-1284.
23. Van de Beek D, Drake JM, Tunkel AR. Nosocomial bacterial meningitis. N Engl J Med. 2010;362:146-154.
24. Hedberg A, Hardemark HG, Olsson-Liljequist B, et al.
Penetration of fusidic acid and rifampicin into cerebrospinal fluid in
low grade inflammatory meningitis caused by Staphylococcus epidermidis. Clin Microbiol Infect. 2004;10:765-768.
25. Antimicrobial prophylaxis for surgery. Treat Guidel Med Lett. 2004;2:27-32.
26. Simon TD, Hall M, Dean JM, et al. Reinfection following initial cerebrospinal fluid shunt infection. J Neurosurg Pediatr. 2010;6:277-285.
27. Veeravagu A, Ludwig C, Camara-Quintana JQ, et al.
Fungal infection of a ventriculoperitoneal shunt: histoplasmosis
diagnosis and treatment. World Neurosurg. 2013;80:222.e5-e13.
28. Chiou CC, Wong TT, Lin HH, et al. Fungal infection of ventriculoperitoneal shunts in children. Clin Infect Dis. 1994;19:1049-1053.
To comment on this article, contact rdavidson@uspharmacist.com.





