US Pharm. 2016;41(4):30-33.
ABSTRACT: Genital herpes continues to be a relevant public-health concern because of its prevalence and its association with an increased risk of acquiring other sexually transmitted diseases. Genital herpes may present as vesicular lesions that can rupture and become painful; however, more than 80% of individuals are asymptomatic, which makes it difficult to contain the spread of disease. The treatment of genital herpes serves to shorten symptom duration and improve quality of life, but it does not cure the disease. Strategies to prevent transmission include chronic suppressive therapy in serodiscordant couples, safe-sex practices, and avoidance of sexual intercourse during outbreaks. Currently available therapies are well tolerated and highly effective, and some newer therapeutic options are being investigated.
Genital herpes is a major public-health concern because of its recurrent nature, its ability to be transmitted asymptomatically, and its potential for complications.1 This sexually transmitted disease (STD) is caused by herpes simplex virus types 1 and 2 (HSV-1 and HSV-2), in most cases by HSV-2.2 Genital herpes is the most common cause of genital ulceration in the United States; more than 50 million Americans are infected, and an estimated 776,000 new cases occur each year.1,3 Nationally, 15.5% of persons aged 14 to 49 years are infected with HSV-2, and the prevalence of genital herpes may be higher owing to increasing rates of HSV-1 causation.4 Genital herpes is also a significant public-health concern for the global community, with more than 500 million people infected worldwide.5 These estimates likely are low; many cases of genital herpes are unreported since people often are unaware that they have been infected.
Demographically, HSV-2 infection is more common in women than in men, and in the non-Hispanic black population, there is an increased prevalence in both sexes. Even among persons with similar numbers of lifetime sexual partners, this disparity between the ethnicities remains.3 Most people with symptomatic genital herpes will experience recurrent infections within the first year of the primary infection, and the median recurrence rate is four to six episodes per year.6
Signs and Symptoms
The vast majority of people infected with HSV-1 or HSV-2 virus are asymptomatic or have mild symptoms that go unnoticed or are mistaken for a different dermatologic condition, such as an ingrown hair or pimple.2,3 More than 80% of patients found to be seropositive have not been previously diagnosed with genital herpes.3,5 Symptomatic patients have one or more vesicular lesions around the genitals, rectum, or mouth. These lesions generally are painless but may rupture, becoming painful, shallow ulcers that take 2 to 4 weeks to heal. The occurrence of symptoms is commonly referred to as an outbreak.2,3,7
Before ulceration, up to 20% of patients experience prodromal symptoms, which may include flulike symptoms such as fever, swollen lymph nodes, headache, and body aches.2,3,7 The patient may also experience mild tingling up to 48 hours before lesion appearance or a shooting pain in the buttocks, legs, or hips up to 5 days beforehand.6 During these clinical episodes, genital lesions or ulcers develop after viral release from nerve endings into the epithelium.5 The average incubation period after exposure is 4 days (range, 2-12 days).3 Up to 60% of infected persons will asymptomatically shed the virus.7
Symptoms differ between the initial outbreak and subsequent outbreaks. Generally, the initial outbreak involves a longer duration of lesions, increased viral shedding, more severe localized symptoms, and systemic symptoms.3,5 The risk of acquiring HSV-2 is correlated with increased viral shedding. Recurrent outbreaks are typically shorter in duration and involve milder symptoms; however, they are more likely to occur during the first year of infection.3 Although the virus remains dormant indefinitely, the number of outbreaks typically decreases over time.3,7 Recurrences and asymptomatic viral shedding are less common in genital HSV-1 infection than in genital HSV-2 infection.3
Pathophysiology and Transmission
HSV-1 and HSV-2 infections are chronic and lifelong.4 When the infection develops, the person’s natural immunologic response is rarely sufficient to eradicate it, resulting in a latent viral infection.8 HSV infection has five key stages: primary mucocutaneous infection, infection of the ganglia, establishment of latency, reactivation, and recurrent infection. The virus replicates by spreading to adjoining cells and peripheral sensory nerves, and latency develops within sensory or autonomic nerve root ganglia. It has not been established which factors help maintain latency; however, immune-system responses and emotional and physical stressors appear to activate the virus.1
HSV is a double-stranded DNA virus. HSV-1 and HSV-2 are distinct serotypes of HSV that are antigenically related.1 HSV-1 is commonly acquired during childhood, with an estimated seroprevalence of 90%.8 HSV-1 is most commonly associated with oropharyngeal disease, whereas HSV-2 is usually associated with genital disease; however, each virus is capable of causing infections in both anatomical areas.1,3 As previously mentioned, HSV-1–related genital herpes is increasing in prevalence, especially in young women and in men who have sex with men.4 Humans are the only known reservoir for HSV, which is spread through contact with lesions, mucosal surfaces, oral secretions, or genital secretions. Genital herpes can be spread though vaginal, anal, or oral sex with an infected person.2,3
Transmission frequently occurs when an infected person without visible sores has sexual contact with a seronegative partner. In patients with asymptomatic HSV-2 infection, genital HSV shedding occurs on 10% of days, frequently without associated symptoms.3 It was previously thought that HSV-2 virus is inactive when a patient is asymptomatic; however, HSV-2 may be constantly replicating in a small number of neurons, which may result in a nearly continuous release of the virus into genital epithelia, rendering infection possible.8 HSV-2 infection is more easily transmitted from men to women than from women to men.3
HSV-2 is a strong risk factor for acquisition of HIV-1 infection.3,8,9 HSV-2 promotes HIV replication in genital epithelial tissue and, through genital ulcers, increases the risk of transmission fivefold.8 Ulcers resulting from a herpes infection reduce the protection that these tissues typically provide, and genital ulcers can easily bleed.3 HSV-2 can also accelerate the progression of HIV-1, and the high burden of HSV-2 infection worldwide is believed to have contributed to the prevalence of HIV-1 infection. In turn, HIV-1 can increase the shedding frequency and quantity of HSV-2 viral particles in a coinfected person.9
Screening for HSV Infection
The CDC currently does not recommend routine screening for HSV-2 in the general population. This is due to several factors, such as the expense of testing, the risk of false-positives, and a lack of evidence of reduced spread of disease when asymptomatic patients are tested. Patients who should be screened for HSV-2 include those whose sexual partner has a known history of genital herpes and those who have multiple sexual partners.10 Tissue culture is the most specific and sensitive method for diagnosing the initial episode of genital herpes; however, it is relatively insensitive when used during later stages of genital-ulcer healing or during recurrent infections. In those situations, viral culture is preferred. Although diagnosis can be confirmed only by laboratory testing, less stringent diagnostic criteria (e.g., vesicular or ulcerative genital lesions and clinical history) are frequently used in practice.1
Treatment
The acyclic guanosine analogues, including acyclovir, valacyclovir, and famciclovir, are the most commonly used therapeutic options for HSV-1 and HSV-2. These drugs are competitive inhibitors of viral DNA polymerase that result in termination of replication. The recommended dosing strategies from the CDC’s STD guidelines are outlined in TABLES 1 to 3.11 Therapy should be administered promptly, ideally within 24 to 72 hours of symptoms. The goal of therapy is not eradication, but rather to shorten the duration of aforementioned symptoms and improve quality of life.
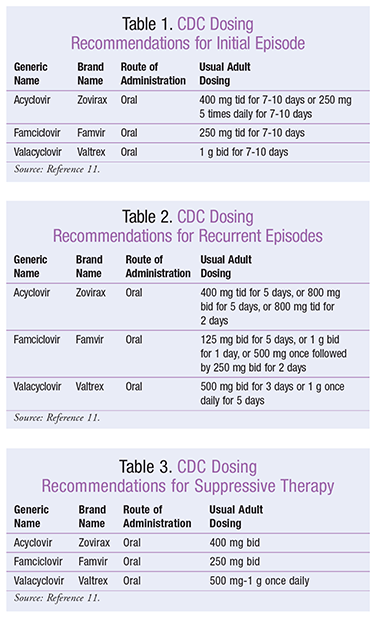
Although the drugs in this class are considered therapeutically equivalent, there are pharmacokinetic differences that could lead to the selection of one agent over another.11 The oral bioavailability of acyclovir is approximately 5% to 15% compared with valacyclovir, which has a three- to fivefold improved bioavailability. Famciclovir, a prodrug of penciclovir, has the highest oral bioavailability (approximately 77%). These differences result in the need for comparatively less frequent dosing. As a class, these agents are well tolerated, and the most common adverse effects are nausea, diarrhea, rash, and headache. Finally, of the agents described below, only acyclovir is available in an IV formulation, which should be reserved for serious infections such as central nervous system disease or disseminated disease. Although acyclovir is also available in topical formulations, topical administration is not associated with clinical benefit and is not recommended by current guidelines.
Following treatment for the initial episode, the patient may opt for chronic suppressive therapy versus an episodic approach. Chronic suppressive therapy may be most appropriate for patients who experience frequent recurrences (>6 per year), but it may also improve quality of life in patients with fewer recurrences.12,13 This strategy decreases the frequency of recurrences by up to 80%.14-17 Another benefit of chronic suppressive therapy is reduced transmission in serodiscordant couples.18 In contrast, episodic therapy is less costly and might be more suitable for patients with adherence issues.19 As with treatment of the initial episode, all of the agents are considered interchangeable; however, some data suggest that famciclovir may be inferior with regard to reducing viral shedding.20
Despite the frequency of use of these agents in clinical practice, resistance is an infrequent problem.21,22 Resistance can develop through several mechanisms, one of which is viral thymidine kinase deficiency (partial or complete). Some viral strains may have DNA polymerase mutations, although this is much less common. Resistance to one of these agents typically confers resistance to others in the class and necessitates the use of alternative drugs, such as foscarnet.
Future Therapeutic Direction
Although the therapeutic approach has changed little in the past 15 years, several recent studies have suggested that new therapeutic and preventive options may be on the horizon. Pritelivir, a HSV-2 helicase inhibitor, has shown promising results with regard to reducing transmission; however, the development of this drug has been suspended owing to dermatologic and hematologic toxicities in animals.23 A gel formulation of tenofovir, another nucleotide inhibitor, has demonstrated significant protection against acquisition of HSV-2 infection.24 Finally, vaccine research is ongoing; however, to date, most vaccines have failed to impart consistent efficacy.25
Counseling for Prevention
The effectiveness of condoms in preventing HSV-2 transmission is less established than their effectiveness for HIV prevention; however, correct and consistent use of latex condoms is moderately effective in preventing HSV-2 transmission.3,9 The effectiveness of condoms in prevention is limited, as outbreaks can occur in areas not covered by the condom. The most effective way to prevent genital herpes transmission is to abstain from sexual contact with partners when genital sores or prodromal symptoms are present. Counseling is critical to ensure that the patient knows that even if he or she has no symptoms, a sexual partner can still be infected. Partners of infected persons should be made aware that although condom use reduces the risk, they still may become infected when condoms are used.3,4 Counseling of both infected persons and their sexual partners is essential in managing genital herpes.4 Patients should be strongly encouraged to inform sexual partners of their status and the risk involved.
Counseling should focus not only on the prevention of disease spread, but also on the psychological effects associated with genital herpes.4,6 Recurrence of genital herpes may be associated with psychological factors that can increase viral shedding owing to their suppression of the immune system. Patients who have labile moods, experience high levels of emotional distress, and are less able to regulate emotional states are more likely to experience recurrence.6 Genital herpes is associated with a tremendous stigma, resulting in anxiety, depression, guilt, shame, and difficulty developing and managing relationships in persons infected.6 These psychological symptoms should be monitored by practitioners, and interventions should be conducted when such symptoms are identified.
Conclusion
Owing to a high rate of asymptomatic infections, the number of new cases of genital herpes is likely to continue to increase in prevalence; however, the disease remains treatable. Identifying patients with genital herpes and treating them with chronic suppression therapy or episodic therapy is imperative, as treatment may prevent future infection with other STDs. Strategies to prevent disease transmission include chronic suppressive therapy in discordant couples, safe-sex practices, and avoidance of sexual intercourse during outbreaks.
REFERENCES
1. Knodel LC. Sexually transmitted diseases. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 9th ed. New York, NY: McGraw-Hill; 2014.
2. CDC. Genital herpes—CDC fact sheet. www.cdc.gov/std/herpes/stdfact-herpes.htm. Accessed October 28, 2015.
3. CDC. Genital herpes—CDC fact sheet (detailed). www.cdc.gov/std/herpes/stdfact-herpes-detailed.htm. Accessed October 28, 2015.
4. CDC. Genital HSV infections. www.cdc.gov/std/tg2015/herpes.htm. Accessed October 28, 2015.
5. Shin H, Iwasaki A. Generating protective immunity against genital herpes. Trends Immunol. 2013;34:487-494.
6. Horn EE, Turkheimer E, Strachan E. Psychological distress, emotional stability, and emotion regulation moderate dynamics of herpes simplex virus type 2 recurrence. Ann Behav Med. 2015;49:187-198.
7. Roett MA, Mayor MT, Uduhiri KA. Diagnosis and management of genital ulcers. Am Fam Physician. 2012;85:254-262.
8. Bernstein DI, Bellamy AR, Hook EW III, et al. Epidemiology, clinical presentation, and antibody response to primary infection with herpes simplex virus type 1 and type 2 in young women. Clin Infect Dis. 2013;56:344-351.
9. Looker KJ, Magaret AS, Turner KM, et al. Global estimates of prevalent and incident herpes simplex virus type 2 infections in 2012. PLoS ONE. 2015;10:e114989.
10. CDC. Genital herpes screening FAQ. www.cdc.gov/std/herpes/screening.htm. Accessed October 28, 2015.
11. Workowski KA, Bolan GA; CDC. Sexually transmitted diseases treatment guidelines, 2015. MMWR Recomm Rep. 2015;64:1-137.
12. Fife KH, Almekinder J, Ofner S. A comparison of one year of episodic or suppressive treatment of recurrent genital herpes with valacyclovir. Sex Transm Dis. 2007;34:297-301.
13. Mills J, Mindel A. Genital herpes simplex infections: some therapeutic dilemmas. Sex Transm Dis. 2003;30:232-233.
14. Diaz-Mitoma F, Sibbald RG, Shafran SD, et al; Collaborative Famciclovir Genital Herpes Research Group. Oral famciclovir for the suppression of recurrent genital herpes: a randomized controlled trial. JAMA. 1998;280:887-892.
15. Mertz GJ, Loveless MO, Levin MJ, et al; Collaborative Famciclovir Genital Herpes Research Group. Oral famciclovir for suppression of recurrent genital herpes simplex virus infection in women. A multicenter, double-blind, placebo-controlled trial. Arch Intern Med. 1997;157:343-349.
16. Reitano M, Tyring S, Lang W, et al; International Valaciclovir HSV Study Group. Valaciclovir for the suppression of recurrent genital herpes simplex virus infection: a large-scale dose range-finding study. J Infect Dis. 1998;178:603-610.
17. Romanowski B, Marina RB, Roberts JN; Valtrex HS230017 Study Group. Patients’ preference of valacyclovir once-daily suppressive therapy versus twice-daily episodic therapy for recurrent genital herpes: a randomized study. Sex Transm Dis. 2003;30:226-231.
18. Corey L, Wald A, Patel R, et al. Once-daily valacyclovir to reduce the risk of transmission of genital herpes. N Engl J Med. 2004;350:11-20.
19. Corey L, Bodsworth N, Mindel A, et al. An update on short course episodic and prevention therapies for herpes genitalis. Herpes. 2007;14(suppl 1):5A-11A.
20. Wald A, Selke S, Warren T, et al. Comparative efficacy of famciclovir and valacyclovir for suppression of recurrent genital herpes and viral shedding. Sex Transm Dis. 2006;33:529-533.
21. Reyes M, Shaik NS, Graber JM, et al. Acyclovir-resistant genital herpes among persons attending sexually transmitted disease and human immunodeficiency virus clinics. Arch Intern Med. 2003;163:76-80.
22. Danve-Szatanek C, Aymard M, Thouvenot D, et al. Surveillance network for herpes simplex virus resistance to antiviral drugs: 3-year follow-up. J Clin Microbiol. 2004;42:242-249.
23. Wald A, Corey L, Timmler B, et al. Helicase-primase inhibitor pritelivir for HSV-2 Infection. N Engl J Med. 2014;370:201-210.
24. Abdool Karim SS, Abdool Karim Q, Kharsany AB, et al. Tenofovir gel for the prevention of herpes simplex virus type 2 infection. N Engl J Med. 2015;373:530-539.
25. Johnston C, Koelle DM, Wald A. HSV-2: in a pursuit of a vaccine. J Clin Invest. 2011;121:4600-4609.
To comment on this article, contact rdavidson@uspharmacist.com.





