US Pharm. 2016;41(12):HS9-HS14.
ABSTRACT: Pancreatic cancer is the fourth most common cause of cancer-related death in both men and women. Although long-term survival is possible with early diagnosis, most patients present with more advanced disease. Pancreatic cancer continues to have low response rates to chemotherapy agents. Fortunately, some progress has been made in advancing treatment options. Patients continue to have complications of pain, pancreatic insufficiency, blood clots, and other issues that pharmacists can work with other healthcare professionals to mitigate.
Pancreatic cancer has been a historically difficult cancer to manage, with overall poor response to traditional therapies. Over the last 10 years, advances in chemotherapy have increased tumor response rates from 5% to 10% with single-agent chemotherapy to over 25% with newer multiagent regimens.1 Although there have been some improvements in response rates and 1-year survival rates, overall outcomes for patients with advanced pancreatic cancer remain poor.
Treatment of pancreatic cancer varies according to the stage, type, and associated complications. Management of pancreatic cancer often requires a multimodal approach ranging from surgery, chemotherapy, and radiation therapy to symptom relief and prophylaxis. This article will review the principles of management for pancreatic cancer, with a focus on advanced-stage disease.1
Epidemiology
In 2016, it is estimated that about 305,000 Americans will be diagnosed with a malignancy of the digestive tract.2 Colon cancer is the most prevalent digestive tract malignancy with 95,270 estimated cases, followed by pancreatic cancer with 53,070. In addition, pancreatic cancer is estimated to cause 41,780 of the anticipated 153,030 deaths from digestive tract tumors in 2016.2
Men and women are equally affected by pancreatic cancer.2 It is the ninth most common cancer in women, but is not in the top 10 for men. However, pancreatic cancer is the fourth most common cause of cancer-related death in both men and women, accounting for 7% of cancer deaths. This highlights the high relative death rate of pancreatic cancer in patients. Pancreatic cancer rates increased 1.2% per year between 2000 and 2012, but the etiology of the rise is unclear. It is a malignancy afflicting an older population, with a median age at diagnosis of >60 years.2
There are several factors that may contribute to a patient’s risk of developing pancreatic cancer. Smokers have double the risk of nonsmokers, and smokeless tobacco may also increase this risk.2 Family history of pancreatic cancer can increase the risk in some patients. Medical history of chronic pancreatitis, obesity, and diabetes increases the risk as well. Dietary factors such as high consumption of red meat and diary products have been associated with increased pancreatic cancer risk. Some additional genetic risk factors include Lynch syndrome (hereditary nonpolyposis colorectal cancer) and germline mutations in breast cancer genes 1 and 2 (BRCA1 and BRCA2).1,2
Presentation
Early stages of pancreatic cancer are often undetected and asymptomatic or are associated with nonspecific symptoms such as vague abdominal pain, nausea, and vomiting. Pancreatic cancer often presents at more advanced stages with symptoms such as jaundice (with or without symptoms of cholangitis), fatigue, weight loss, abdominal pain, back pain, nausea, and vomiting. Uncommonly, pancreatic cancer may affect glucose metabolism, causing patients to present with hyperglycemia.1,2
The workup for suspected pancreatic cancer involves a multitude of testing, including endoscopic ultrasound and/or abdominal CT scan (to identify any masses, blockage, and extent of disease), chest x-ray, liver function tests, cancer antigen (CA) 19-9, basic metabolic panel, and CBC. If patients have jaundice at presentation or as part of progressive disease, either an MRI/MRCP (magnetic resonance cholangiopancreatography) or endoscopic retrograde cholangiopancreatography (ERCP) is warranted. These procedures can provide diagnostic information as well as resolve the jaundice by stenting any blockages in the bile duct that may be present.1,2
Diagnosis will be made from tissue obtained during a procedure such as the ERCP, laparoscopic biopsy, or surgery. Pancreatic cancer most commonly originates in the head of the pancreas versus the body or tail of the organ. Additionally, pathology reveals most pancreatic cancers are adenocarcinomas arising from the ductal epithelium of the pancreas (>95%), with a small portion arising from the exocrine glands of the pancreas exhibiting neuroendocrine features (<5%). Molecular analysis reveals several common mutations in pancreatic cancer cells, including K-Ras (>90%), p53, cyclin-dependent kinase inhibitor 2A (CDKN2A), BRCA2, and SMAD4.3 Mutations that lead to uncontrolled growth include K-Ras, whereas mutations in tumor suppressor genes such as p53, CDKN2A, and SMAD4 lead to loss of activity.4
Staging and Treatment
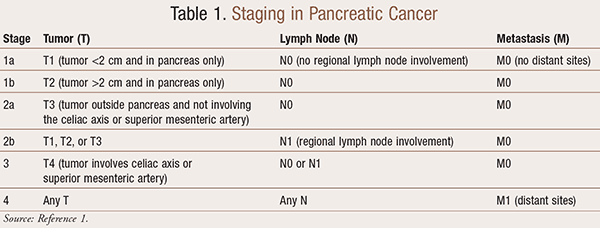
Staging is based on TNM classification and takes into consideration the tumor size (T), lymph node involvement (N), and any metastatic spread (M) of disease (TABLE 1).1 Considering treatment options overall, surgery is potentially curable for early-stage disease. Patients with more advanced stages who cannot have surgical intervention are not curable. Chemotherapy and radiation are used after surgery to reduce recurrences, and chemotherapy is the primary method to treat advanced stages of the disease.1
The National Comprehensive Cancer Network (NCCN) uses a system of guidelines to categorize the level of scientific evidence and consensus for treatment recommendations.1 They include: Category 1 (high level of evidence with uniform consensus); Category 2A (lower level of evidence with uniform consensus); Category 2B (lower level of evidence without a uniform consensus but with no major disagreement); and Category 3 (any level of evidence but with major disagreement).1
Stages 1-2: During the evaluation process, patients will be identified as surgical candidates with resectable, borderline resectable, or unresectable tumors. The surgery for patients with resectable tumors involves an extensive surgery called the Whipple procedure. During this procedure, a portion of the pancreas is removed and several anatomical changes are necessary to reroute gastrointestinal (GI) tract functions. Successful surgical resection is essential for a patient to have a chance of cure. Unfortunately, only a small proportion of patients present with stage 1 or 2.1
Patients with early-stage disease who undergo surgery are candidates for additional therapy after surgical healing and recovery. Therapy with adjunctive chemotherapy and/or radiation is an option to delay recurrence and extend survival. Chemotherapy with gemcitabine, followed by radiation therapy, then additional gemcitabine have shown to extend overall survival as compared to chemo-therapy alone to 20.4 months versus 16.6 months (hazard ratio [HR] 2.42, CI 1.17-5.01).4 An additional phase II study did not identify a survival difference (i.e., 24 months in both groups); however, it did find combined chemo-therapy and radiation therapy following surgery reduced local recurrences from 24% to 11%.5 Adjuvant chemotherapy options in this setting include gemcitabine (Category 1), fluorouracil (5-FU)/leucovorin (Category 1), continuous infusion 5-FU (Category 2B), or capecitabine (Category 2B).1 Following therapy, patients are seen every 3 to 6 months for 2 years, then every 6 to 12 months ongoing to monitor for recurrence.1
Stage 3: Patients with locally advanced pancreatic cancer that is not resectable are often candidates for chemotherapy and/or radiation therapy. If a patient is in relatively good health, neoadjuvant chemotherapy with gemcitabine is indicated to shrink the tumor to such size that surgical excision may be successful.1 If this outcome is achieved, patients may receive more chemotherapy and/or radiation following surgery as above. If a patient is not felt to be able to tolerate surgery, chemotherapy and radiation combined may have some benefit. In a study of 74 patients with locally advanced unresectable pancreatic cancer, patients were randomized to gemcitabine alone or gemcitabine with radiation therapy. Quality of life and toxicities were similar; however, median survival was 9.2 months with gemcitabine versus 11.1 months with gemcitabine and radiation (P = .017).6
Stage 4: There are numerous chemotherapy options for patients with advanced-stage disease (TABLE 2).1 The NCCN guidelines have several preferred (Category 1) regimens, including FOLFIRINOX (5-FU, leucovorin, irinotecan, oxaliplatin) or gemcitabine plus albumin-bound paclitaxel in patients with good performance status.1

In studies with FOLFIRINOX, a median overall survival of 10.2 months was associated with an overall response rate of 26% to 31%, and a median response duration of 9.3 months has been noted.7 FOLFIRINOX and gemcitabine have been shown to increase emotional functioning of patients and decrease pain, insomnia, and constipation during therapy. Additionally, FOLFIRINOX has been shown to increase global health status (P < .001). FOLFIRINOX therapy was associated with a more prolonged response time until a decrease in quality-of-life measures as compared with gemcitabine.8
When weighing the option of FOLFIRINOX versus gemcitabine in combination with albumin-bound paclitaxel, a review of response rates, survival rates, and toxicities can be helpful. Overall response rates are 31.6% for FOLFIRINOX and 23% for gemcitabine plus albumin-bound paclitaxel.9 In clinical trials, median survival was 11.1 versus 8.5 months, and 1-year survival was 48% versus 35%, respectively, for FOLFIRINOX versus gemcitabine plus albumin-bound paclitaxel. Toxicities such as fatigue and neutropenia are higher with FOLFIRINOX.9
Additional options include gemcitabine and erlotinib, gemcitabine and capecitabine, gemcitabine alone, or 5-FU monotherapy as well as several others. NCCN guidelines currently recommend gemcitabine alone or in combination with erlotinib if the FOLFIRINOX or gemcitabine plus albumin-bound paclitaxel is not used (TABLE 3).1

Bevacizumab has been studied in combination with gemcitabine therapy for advanced pancreatic cancer. Outcomes of therapy showed overall response rates of 21% and median survival of 8.8 months; however, grade 3 and 4 hypertension (19%), thrombosis (13%), GI perforation (8%), and bleeding (2%) limit the ability to tolerate the regimen.10 A large phase III CALGB (Cancer and Leukemia Group B) study in 602 patients confirmed a lack of improvement in overall survival with bevacizumab and gemcitabine combination therapy. Overall response rates were 13% with the combination and 10% with monotherapy in this confirmation study.11
In a phase III study of gemcitabine versus gemcitabine and capecitabine, overall survival was 7.2 versus 8.4 months (P = 0.234), respectively. In patients with very good performance status (>90% on the Karnofsky Performance Scale), the combination was favored over monotherapy in a subset analysis. Overall response rates were 7.8% with monotherapy versus 10.0% with combination therapy.12 In a larger phase III study, objective response rates were 12.4% versus 19.4% (P = 0.034), favoring the combination. Progression-free survival was significantly improved in the combination group (HR 0.78, CI 0.68-0.93); however, overall survival was not improved.13
Gemcitabine monotherapy is a Category 1 recommendation for both good and poor prognosis patients as compared to most gemcitabine combinations (TABLE 2).1
A meta-analysis of chemotherapy for locally advanced and metastatic pancreatic cancer included >9,000 patients from 51 clinical trials.14 This study found that chemotherapy improved survival as compared to best supportive care (HR 0.64, CI 0.42-0.98). Gemcitabine-based therapy improved survival (HR 0.91, CI 0.85-0.97), whereas 5-FU–based therapy did not. This analysis was done before FOLFIRINOX was investigated.14
In patients with poor performance status, gemcitabine monotherapy is preferred, with best supportive care as an alternative (TABLE 2).1 A randomized, phase III, controlled study reported 55 patients who chose gemcitabine-based therapy versus pancreatic enzyme replacement therapy (PERT).15 Median survival was 14.3 months with gemcitabine-based therapy as compared to 4.3 months with PERT alone. One-year survival rates were 56% with chemotherapy versus 16% with PERT alone. Finally, quality-of-life ratings favored the chemotherapy group as well (P < .01).15
Refractory Disease: For patients who relapse following gemcitabine-based therapy, oxaliplatin, leucovorin, and 5-FU have been evaluated in the CONKO-003 trial.16 In this study, 168 patients were randomized to leucovorin and 5-FU alone or in combination with oxaliplatin. Median overall survival was 3.3 months versus 5.9 months with the oxaliplatin combination (HR 0.66, CI 0.48-0.91). Toxicity was similar, except for increased rates of neurotoxicity with oxaliplatin, 38.2% versus 7.1%, respectively.16
Capecitabine in combination with erlotinib 150 mg daily has been evaluated in 30 patients with refractory pancreatic cancer.17 The overall response rate was 10.0%, with a median survival duration of 6.5 months. Common toxicities of therapy included diarrhea, skin rash, fatigue, and hand-foot syndrome.17
More recently, ruxolitinib (a JAK1/JAK2 inhibitor) has been investigated in combination with capecitabine in gemcitabine-refractory patients, with some positive results.18 An early trial with vismodegib (a Hedgehog pathway inhibitor) failed to produce positive effects with gemcitabine.19
Biomarkers as predictors of response are being evaluated as well. The biomarker hENT1 (human equilibrative nucleoside transporter 1) may predict responses to gemcitabine, and the SMAD4 gene may predict responses to radiation in locally advanced disease. The SPARC gene may predict benefit for the combination of gemcitabine and albumin-bound paclitaxel in advanced disease. C-reactive protein elevations may predict response to ruxolitinib in refractory patients.1,9
Overall, a number of agents and regimens are available for metastatic pancreatic cancer. Patients will often start with FOLFIRINOX or gemcitabine plus albumin-bound paclitaxel and note prolonged response and increased survival benefits. Second-line therapy is often gemcitabine-based if patients have not received it previously. Unfortunately, response rates and duration of response in relapse are shorter. Consideration for a clinical trial in this setting is appropriate.
Complications of Therapy
Pancreatic cancer patients often experience a number of disease-related complications, either at presentation or as the cancer progresses. Bile duct obstruction can occur as the disease progresses and presents with jaundice, nausea, and vomiting with or without fever. An ERCP or MRCP is used to stent the bile duct and allow for bile drainage and resolution of symptoms. If internal stenting in not successful, percutaneous stents may be placed to resolve bile duct obstruction as well. If patients also present with fever, antibiotic management for cholangitis is indicated with broad-spectrum coverage of gram-negatives, coliforms, and anaerobes.1
GI obstruction can occur as well, presenting with severe nausea, vomiting, and abdominal pain. It is often managed with bowel rest and decompression. If obstruction continues, a gastrostomy or gastrojejunostomy tube can be placed for decompression. Pancreatic function may also be compromised either by the cancer itself or as a complication of surgical and other therapies. PERT may be needed in some patients who are experiencing difficulty digesting food, particularly fats in the diet.2
Depression, pain, and malnutrition are common symptoms of advancing pancreatic cancer. Pancreatic cancer often spreads to the liver and local areas, sometimes affecting the celiac plexus, leading to severe pain that is resistant to opioid therapy. Traditional pain management with opioids is often required, but may not fully control the pain in a given patient. For patients who fail to respond to opioids, interventional pain services such as celiac nerve blocks can relieve pain in the more severe cases. Supportive therapy with oral nutritional supplements is appropriate for patients, but IV nutritional therapy is not commonly used. Antidepressant therapy can be indicated in patients found to have depression; when choosing agents to manage depression, choices that also treat neuropathic pain can benefit both issues for a patient. Typical antidepressants used are selective serotonin reuptake inhibitors (SSRIs).1
Venous thromboembolism (VTE) is a common complication of advanced pancreatic cancer. As a standard of practice, prophylaxis in the ambulatory population is not indicated. Deep venous thrombosis (DVT) prophylaxis is warranted when a patient is admitted to the hospital, as long as he or she does not have contra-indications to anticoagulation. When a patient does develop a blood clot, therapy options include heparin and low-molecular-weight heparin (LMWH). For long-term anticoagulation, LMWH has been shown as superior to warfarin in preventing clot recurrences. Newer oral agents such as direct thrombin inhibitors and anti-factor Xa agents have limited data in the cancer population.1
It has been documented that in cancer patients under-going chemotherapy, the incidence of VTE is increased. A recent study of 312 patients used prophylactic enoxaparin with chemotherapy versus chemotherapy alone for advanced pancreatic cancer.20 In the study, the rate of venous blood clots was 6.4% in the enoxaparin group versus 15.1% in the chemotherapy alone group (HR 0.40, CI 0.19-0.83; P = .01). Enoxaparin dosing was therapeutic for 3 months, and then reduced to 40 mg SC daily until disease progression. Treatment outcomes were not affected in the study, and the number of major bleeding events was equivalent.20 These data may lead to changes in VTE prophylaxis recommendations in pancreatic cancer patients undergoing chemotherapy.
Adverse Events: Adverse events from chemotherapy are another area where pharmacists need to be aware of the toxicities that patients can experience and to offer assistance with management strategies. Some common agents that have been described already include gemcitabine, capecitabine, 5-FU, irinotecan, oxaliplatin, erlotinib, and albumin-bound paclitaxel. Capecitibine and 5-FU have similar adverse-event profiles, including mucositis, rash, fever, hand-foot syndrome, anorexia, myelosuppression, and nausea and vomiting.7,8,12,13,21 Gemitabine is typically associated with fever with infusion, myelosuppression, mild nausea and vomiting, and anorexia.4-6,8,10-13,18,21 Irinotecan is associated with flushing and sweating during the infusion, acute and delayed diarrhea, and myelo-suppresion.7,8,21
In addition, oxaliplatin therapy is associated with mild infusion reactions, peripheral neuropathy, mild nausea and vomiting, cold intolerance, and myelo-suppresion.7,8,21 Albumin-bound paclitaxel is associated with reduced incidence of infusion reactions as compared to standard paclitaxel, as well as with myelosuppresion, peripheral neuropathy, hair loss, mild nausea and vomiting, and anorexia.21
It is important for pharmacists to be prepared to offer interventions to assist patients with a variety of adverse events. Acetaminophen can reduce the fever associated with gemcitabine infusion.22 Mouth care regimens for mucositis include oral rinses with normal saline or sodium bicarbonate solutions, antifungal topical therapy, and pain management.22 Delayed diarrhea associated with irinotecan can be managed with OTC loperamide and fluid support, but if severe, referral to acute medical care is warranted.21,22 If patients develop pain, redness, or swelling in their hands and feet with capecitabine or 5-FU, referral to their primary provider is needed.22 Nausea and vomiting can be managed with prescription antiemetics such as ondasentron, prochlorperazine, metoclopramide, or lorazepam.23 Because of the risks of myelosuppression, if patients develop a fever while on therapy, immediate referral to their oncology provider is warranted to assess the risk for infection and possible hospitalization.24
A management plan for the pancreatic cancer patient needs to include an awareness of multiple possible complications, some of which require hospitalization and multidisciplinary collaboration.1 Pharmacists can help to manage these complications and enhance the supportive care of patients.
Outcomes
Historically, survival in pancreatic cancer is limited; for all stages, 1-year survival is 29%, with a 5-year survival rate of 7%.1,2 Five-year survival can be up to 27% for patients with early-stage disease (9%) who also undergo extensive surgical resection. When looking at patients with late-stage pancreatic cancer, 1-year survival is 15% and 5-year survival is 2%. The survival data are better for the small percentage of patients with neuroendocrine-type pancreatic cancer as compared to the majority of patients with adenocarcinomas (exocrine type).1,2
In advanced pancreatic cancer treated with combination chemotherapy, 1-year survival rates in studies range from 25% to 40%.1,2 This marks a significant improvement over historical data. Monotherapy with gemcitabine has been shown to enhance 1-year survival in patients with poorer functional status as well.1,2 With continued research, these benchmarks may change further.
Conclusion
Pancreatic cancer is a complex disease with a multitude of complications that may occur in a population of patients who are typically elderly. The foundation of treating early-stage disease is surgery followed by adjuvant therapy to enhance survival. Successful surgical excision can give patients a chance for cure in the early stage. Unfortunately, most patients present with late-stage disease and are not surgical candidates. Chemotherapy can extend survival and benefit quality of life, but there is a long way to go in improving outcomes in this disease. Enrollment in clinical trials is needed to further identify better therapy for patients in the future. Pharmacists can offer recommendations to manage the complications of pancreatic cancer, in particular pain management, depression treatment, nutritional supplementation, and thromboembolism management.
REFERENCES
1. National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology. Pancreatic adenocarcinoma, version 1.2016. www.nccn.org/professionals/physician_gls/f_guidelines.asp. Accessed August 1, 2016.
2. American Cancer Society. Cancer Facts & Figures 2016. Atlanta, GA: American Cancer Society; 2016. www.cancer.org/acs/groups/content/@research/documents/document/acspc-047079.pdf. Accessed August 1, 2016.
3. Khawaja MR, Kleyman S, Yu Z, et.al. Adjuvant gemcitabine and gemcitabine-based chemoradio-therapy versus gemcitabine alone after pancreatic cancer resection: the Indiana University experience. Am J Clin Oncol. 2014 Aug 13 [Epub ahead of print].
4. Polireddy K, Chen Q. Cancer of the pancreas: molecular pathways and current advancement in treatment. J Cancer. 2016;7(11):1497-1514.
5. Van Laethem JL, Hammel P, Mornex F, et al. Adjuvant gemcitabine alone versus gemcitabine-based chemoradiotherapy after curative resection for pancreatic cancer: a randomized EORTC-40013-22012/FFCD-9203/GERCOR phase II study. J Clin Oncol. 2010;28(29):4450-4456.
6. Loehrer PJ, Feng Y, Cardenes H, et al. Gemcitabine alone versus gemcitabine plus radiotherapy in patients with locally advanced pancreatic cancer: an Eastern Cooperative Oncology Group trial. J Clin Oncol. 2011;29(31):4105-4112.
7. Conroy T, Paillot B, Francois E, et al. Irinotecan plus oxaliplatin and leucovorin-modulated fluorouracil in advanced pancreatic cancer—a Groupe Tumeurs Digestives of the Federation Nationale des Centres de Lute Contre le Cancer study. J Clin Oncol. 2005;23(6):1228-1236.
8. Gourgou-Bourgade S, Bascoul-Mollevi C, Desseigne F, et al. Impact of FOLFIRINOX compared with gemcitabine on quality of life in patients with metastatic pancreatic cancer: results from the PRODIGE 4/ACCORD 11 randomized trial. J Clin Oncol. 2012;31(1):23-29.
9. Ko AH. Progress in the treatment of metastatic pancreatic cancer and the search for next oppor-tunities. J Clin Oncol. 2015;33(16):1779-1786.
10. Kindler HL, Friberg G, Singh DA, et al. Phase II trial of bevacizumab plus gemcitabine in patients with advanced pancreatic cancer. J Clin Oncol. 2005;23(31):8033-8040.
11. Kindler HL, Niedzwiecki D, Hollis D, et al. Gemcitabine plus bevacizumab compared with gemcitabine plus placebo in patients with advanced pancreatic cancer: phase III trial of the Cancer and Leukemia Group B (CALGB 80303). J Clin Oncol. 2010;28(22):3617-3622.
12. Herrmann R, Bodoky G, Ruhstaller T, et al. Gemcitabine plus capecitabine compared with gemcitabine alone in advanced pancreatic cancer: a randomized, multicenter, phase III trial of the Swiss Group for Clinical Cancer Research and the Central European Cooperative Oncology Group. J Clin Oncol. 2007;25(16):2212-2217.
13. Cunningham D, Chau I, Stocken DD, et al. Phase III randomized comparison of gemcitabine versus gemcitabine plus capecitabine in patients with advanced pancreatic cancer. J Clin Oncol. 2009;27(33):5513-5518.
14. Sultana A, Smith CT, Cunningham D, et al. Meta-analyses of chemotherapy for locally advanced and metastatic pancreatic cancer. J Clin Oncol. 2007;25(18):2607-2615.
15. Chabot JA, Tsai WY, Fine RL, et al. Pancreatic proteolytic enzyme therapy compared with gemcitabine-based chemotherapy for the treatment of pancreatic cancer. J Clin Oncol. 2009;28(12):2058-2063.
16. Oettle H, Riess H, Stieler JM, et al. Second-line oxaliplatin, folinic acid, and fluorouracil versus folinic acid and fluorouracil alone for gemcitabine refractory pancreatic cancer: outcomes from the CONKO-003 trial. J Clin Oncol. 2014;32(23):2423-2429.
17. Kulke MH, Blaszkowsky LW, Ryan DP, et al. Capecitabine plus erlotinib in gemcitabine refractory advanced pancreatic cancer. J Clin Oncol. 2007;25(30):4787-4792.
18. Hurwitz HI, Uppal N, Wagner SA, et al. Randomized, double-blind, phase II study of ruxolitinib or placebo in combination with capecitabine in patients with metastatic pancreatic cancer for whom therapy with gemcitabine has failed. J Clin Oncol. 2015;33(34):4039-4047.
19. Catenacci DV, Junttila MR, Karrison T, et al. Randomized phase1b/II study of gemcitabine plus placebo or vismodegib, a hedgehog pathway inhibitor, in patients with metastatic pancreatic cancer. J Clin Oncol. 2015;33(36):4284-4292.
20. Pelzer U, Opitz B, Deutschinoff G, et al. Efficacy of prophylactic low-molecular-weight heparin for ambulatory patients with advanced pancreatic cancer: outcomes from the CONKO-004 trial. J Clin Oncol. 2015;33(18):2028-2034.
21. AHFS Drug Information 2016. Bethesda, MD: American Society of Health-System Pharmacists; 2016.
22. National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology. Palliative care, version 1.2016. www.nccn.org/professionals/physician_gls/f_guidelines.asp. Accessed November 17, 2016.
23. National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology. Antiemesis guidelines, version 2.2016. www.nccn.org/professionals/physician_gls/f_guidelines.asp. Accessed November 17, 2016.
24. National Comprehensive Cancer Network. NCCN clinical practice guidelines in oncology. Prevention and treatment of cancer related infections, version 2.2016. www.nccn.org/professionals/physician_gls/f_guidelines.asp. Accessed November 17, 2016.
To comment on this article, contact rdavidson@uspharmacist.com.





