US Pharm. 2009;34(11):HS10-HS19.
Traumatic brain injury (TBI), defined as a strong impact to the head or a penetrating head injury that alters the function of the brain, affects the lives of approximately 1.4 million people in the United States each year.1 TBI currently accounts for an estimated 1.1 million emergency department admissions and 50,000 deaths annually. Individuals at highest risk for TBI are young children 6 months to 4 years old, males between the ages of 15 and 30 years, elderly individuals (aged 75 years or older), and certain military personnel. According to the CDC, the leading causes of TBI include falls (28%), motor vehicle crashes (20%), events in which a person is struck by or against an object (19%), and assaults (11%).1
Types of Injuries
To appropriately treat and provide care to patients with TBI, it is important to understand the mechanism and pathophysiology behind the injury. There are three key mechanisms of injury—blunt, penetrating, and blast, which are further divided into primary or secondary injuries.2 Blunt injuries are the most common cause of TBI and are frequently the result of motor vehicle (i.e., automobile, motorcycle, pedestrian) crashes, falls, sports-related injuries, and assaults. Penetrating injuries result when any sharp or blunt object penetrates the scalp or skull (e.g., gunshot wounds). Lastly, blast wounds are the result of a combination of blunt and penetrating injuries.
In addition to identifying the mechanism of injury, TBI is also separated into either primary or secondary injury.2 A primary injury occurs at the time of injury and directly damages neuronal tissue. Resultant lesions from a primary injury are either focal, where the injury is in a localized area of the brain, or diffuse, in which the injury is large and widespread. The secondary injury that occurs from TBI is not initially seen; instead it is the result of a normal physiological response to the primary injury, such as tissue hypoxia, that develops over a period of time from hours to months following the primary injury.2
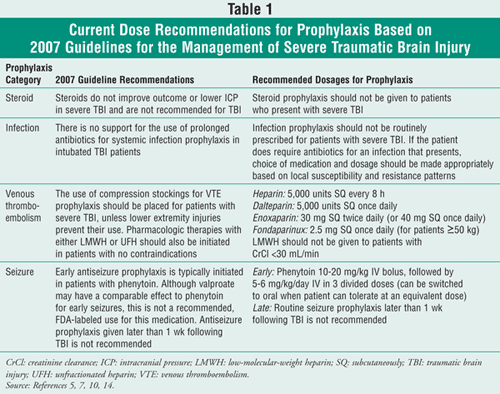
For decades, the concentration of TBI management was on maintaining the control of intracranial pressure (ICP) and cerebral perfusion pressure. As will be discussed, the focus of TBI management no longer resides in one central area; instead, managing all aspects of TBI are now important areas of therapeutic decision making. Building upon the 2007 Guidelines for the Management of Severe Traumatic Brain Injury, this article will focus on prophylactic therapies currently being used and recommended in patients with TBI (TABLE 1).
Prophylactic Steroids
Beneficial effects of corticosteroids in TBI were originally discovered in the 1960s. At that time, corticosteroids were thought to be favorable through a significant reduction in brain edema, a decrease of cerebrospinal fluid production, and the lessening of free radical production. Since then, there have been numerous clinical studies examining the role that corticosteroids play in neurological procedures, as well as in TBI. In 1976, two trials were conducted comparing low- and high-dose glucocorticoids in patients with severe TBI.3,4 Results of these trials demonstrated favorable response to high-dose glucocorticoid groups and favorable dose-related effects on mortality. Successively, in the 1980s and 1990s, there were further trials that evaluated clinical outcomes in patients with TBI, ICP, or both. None of the studies demonstrated a sizeable benefit in using glucocorticoids in those patients.5
More recently, in 2004, the CRASH (Corticosteroid Randomization After Significant Head Injury) trial authors described the results of an international randomized, controlled trial in patients with TBI using high-dose methylprednisolone (2-g IV methylprednisolone followed by 0.4 mg/h for 48 h) or placebo.6 The study was concluded early due to an interim analysis that demonstrated a harmful effect of methylprednisolone, particularly with regard to mortality. The authors thus concluded that although the cause of increase in mortality was unclear, the detrimental effects were not different among groups of patients further classified by injury severity.
In general, the use of prophylactic corticosteroids is not recommended at this time for improving patient outcomes or reducing ICP/edema in patients with TBI.5 Furthermore, it is concluded that high-dose methylprednisolone may be correlated with an increase in mortality. Due to the results of recent trials, there is little interest in pursuing further research in this area at this time.
Infection Prophylaxis
With an increased incidence of mechanical ventilation and other invasive aspects of patient monitoring and treatment, patients with TBI are at a significantly increased likelihood of developing infections. The issue of infection prophylaxis has been evaluated by numerous sources and has revolved around different possibilities of infection development.
One possible infection source is through the insertion of ICP devices. The incidence of ICP device infection can range from less than 1% to 27%.7 Even with these striking infection rates, there are conflicting data concerning what the appropriate prophylactic measures should contain for patients with TBI. Most studies cited by the 2007 guidelines evaluating prophylactic antibiotic usage in patients with external ventricular drainage have shown no difference in infection rates. Additionally, one study showed that patients receiving bacitracin flushes experienced a significantly higher infection rate than those without prophylactic measures.7
Prophylactic antibiotic use in patients with TBI have shown no meaningful reduction in nosocomial infections.7 In addition, an increase in serious gram-negative infections was noted in this population. The guidelines also cite data showing an increase in resistant or gram-negative nosocomial pneumonias. This was seen when prophylactic antibiotics were given for longer than 48 hours in general trauma patients. In contrast, one study showed a decrease in pneumonias when prophylactic antibiotics were given to patients with TBI. No difference in mortality was noted, so it is difficult to assess the usefulness of this therapeutic avenue.7
Since publication of the 2007 guidelines, one further study in patients with TBI has been completed.8 This study retrospectively evaluated the use of antibiotic prophylaxis in patients with ICP monitor implantation. Of the 155 patients included in the analysis, only two developed CNS infections, and these were both in the group that received prophylactic antibiotics. Additionally, both complications from infections and multidrug resistant infections were significantly increased in the group that received antibiotics.
In summary, there are currently no convincing data to support infection prophylaxis in patients with TBI, especially in light of data suggesting that prophylaxis might predispose patients to more severe infections when infections would arise.7,8 To be fair, there is a relative lack of data to give any definitive statement on the use of prophylactic antibiotics, but at this time this practice cannot be supported.
Venous Thromboembolism Prophylaxis
Venous thromboembolism (VTE) is a topic that is very pertinent to patients with TBI, as they are at an increased risk of developing blood clots due to their typically persistent immobile state. The development of deep vein thrombosis (DVT) in patients with TBI who do not receive prophylaxis is as high as 25%.9 However, there remains some uncertainty about the appropriateness of VTE prophylaxis due to the questionable safety of anticoagulation in patients who have TBI. Mechanical means of VTE prophylaxis such as sequential compression devices seem to carry less risk in patients with TBI, but concomitant lower limb injuries can prevent their use in some patients. Per the 2007 guidelines, mechanical VTE prophylaxis is recommended in all severe patients with TBI who have no contraindications to usage, such as an intracranial bleed.10
Concerning pharmacologic options, the guidelines suggest that low-molecular-weight heparin (LMWH) or low-dose unfractionated heparin (UFH) should be used in addition to mechanical means of VTE prophylaxis when no contraindications to LMWH or UFH exist. It is important to note, however, that the guidelines do not elicit a preferred agent, dosage, or timing of pharmacologic prophylaxis and also warn of the increased risk of intracranial hemorrhage expansion.10
Three studies have been published since the guidelines on pharmacologic VTE prophylaxis in patients with TBI.11-13 A 2007 prospective study evaluated the use of dalteparin in trauma patients in which 23% were patients with TBI.11 These patients received prophylactic dalteparin (5,000 units SQ once daily) if there was no active or progressing bleed on CT scan. No patients developed or had increased intracranial bleeding after initiation of dalteparin, while only 3.9% of patients had evidence of DVT and 0.8% had evidence of pulmonary embolism. A second prospective study published in 2008 evaluated the use of enoxaparin (30 mg SQ twice daily) in patients with TBI.12 Progressive hemorrhagic injury was seen in 3.4% of the TBI population, with 67% of those patients having clinically insignificant hemorrhagic changes. In addition, one patient died from a potential side effect from enoxaparin usage. The authors of these two studies concluded that enoxaparin and dalteparin are safe options in patients with TBI and that both agents have a relatively low risk of significant bleeding complications.11,12
A retrospective study published in 2009 looked at a comparison of the risk of DVT in patients with and without TBI utilizing either LMWH or UFH (doses not specified).13 This study showed a three- to fourfold increase in DVTs in patients with TBI using relative risks with 95% confidence intervals. The highest rate was noted when prophylaxis was started greater than 48 hours after insult.
In summary, it is currently acceptable to recommend mechanical and pharmacologic VTE prophylaxis in patients with TBI that have no contraindications to usage.10-13 The evidence is shifting in favor of quick initiation of prophylactic agents regardless of product selection. However, the breakpoint for deciding between mechanical and pharmacologic choices or agents is still unknown and should be a course of future study.
Seizure Prophylaxis
Posttraumatic seizure (PTS) is a common occurrence in patients with TBI. These seizures are broken into two groups: early (within 7 days of injury) and late (after 7 days). Certain risk factors have been shown to place patients with TBI at increased risk for PTS. These risk factors include: Glasgow Coma Score (a neurological scale used to assess level of consciousness) less than 10; cortical contusion; depressed skull fracture; subdural, intracerebral, and epidural hematoma; penetrating head wound; or seizure within 24 hours after injury.14
Phenytoin and valproate sodium have been studied in the prevention of early and late PTS. One study cited by the 2007 guidelines showed a significant reduction in early PTS without showing any significant effect on late PTS or survival with the use of phenytoin.14 In contrast, a randomized, double-blind study showed no early or late PTS benefit by using phenytoin. Valproate sodium has shown a similar rate of early PTS reduction when compared to phenytoin. However, a trend toward higher mortality in the valproate sodium group was noted and could be cause for concern.14
Since the publication of the guidelines, there have been two pertinent studies completed regarding seizure prophylaxis.15-16 The first, a study from 2008, compared the incidence of seizures in patients with TBI when randomized to either phenytoin or levetiracetam.15 Results indicated that there was no significant difference in seizure incidence. However, patients receiving levetiracetam showed increased incidence of electroencephalogram (EEG) abnormalities. An EEG was indicated if patients displayed persistent coma, decreased mental status, or clinical signs of seizures.
The second study evaluated the incidence of late PTS in patients with TBI who did or did not receive antiseizure prophylaxis upon initial presentation.16 This study was carried out in Italy, with phenobarbital being one of the main agents used in antiseizure prophylaxis. Interestingly, in the retrospective portion of the study, 29% of patients who received antiseizure prophylaxis developed late PTS, while only 13% of patients who did not receive prophylaxis developed late PTS. While the retrospective data were not statistically significant, the prospective data were even more striking. A significant difference was noted in the prospective group, where 39% of patients treated with antiseizure prophylaxis developed late PTS, while none of the patients who were not treated with antiseizure prophylaxis developed late PTS. However, phenobarbital is not commonly used in the U.S. in this regard because of its adverse-effect profile and multiple drug interactions, and because more appropriate antiepileptic selections are available; therefore, results must not be generalized too drastically.
In summary, current literature including the 2007 guidelines indicates that the incidence of early PTS appears to be reduced with the addition of prophylactic antiseizure medications.14 However, there is currently no evidence to indicate that prophylactic antiseizure medications alter mortality or incidence of late PTS, and it is still unknown whether or not this course of therapeutic prophylaxis is currently benefitting patients.
Conclusion
Traumatic brain injury can be overwhelming and distressing to both patients and their family members. In addition to the emotional and social impacts that accompany TBI, it is important to identify and deliver prompt attention to the physical needs of the patient. Prophylactic medications play an important role in patients with TBI, yet as of now many categories lack definitive data to direct appropriate therapeutic choices. Future studies are needed to clarify this important issue in the management of patients with this condition.
REFERENCES
1. Langlois JA, Rutland-Brown W, Thomas KE. Traumatic Brain Injury in the United States: Emergency Department Visits, Hospitalizations, and Deaths. Atlanta, GA: Centers for Disease Control and Prevention, National Center for Injury Prevention and Control; 2006.
2. Nolan S. Traumatic brain injury: a review. Crit Care Nurs Q. 2005;28:188-194.
3. Faupel G, Reulen HJ, Muller D, et al. Double-blind study on the effects of steroids on severe closed head injury. In: Pappius HM, Feindel W, eds. Dynamics of Brain Edema. New York, NY: Springer-Verlag; 1976:337-343.
4. Gobiet W, Bock WJ, Liesgang J, et al. Treatment of acute cerebral edema with high dose of dexamethasone. In: Beks JW, Bosch DA, Brock M, eds. Intracranial Pressure III. New York, NY: Springer-Verlag; 1976:231-235.
5. Brain Trauma Foundation, American Association of Neurological Surgeons, Congress of Neurological Surgeons, et al. Guidelines for the management of severe traumatic brain injury. XV. Steroids. J Neurotrauma. 2007;24(suppl 1):S91-S95.
6. Roberts I, Yates D, Sandercock P, et al. Effect of intravenous cortico steroids on death within 14 days in 10,008 adults with clinically significant head injury (MRC CRASH trial): randomised placebo-controlled trial. Lancet. 2004;364:1321-1328.
7. Brain Trauma Foundation, American Association of Neurological Surgeons, Congress of Neurological Surgeons, et al. Guidelines for the management of severe traumatic brain injury. IV. Infection prophylaxis. J Neurotrauma. 2007;24(suppl 1):S26-S31.
8. Stoikes NF, Magnotti LJ, Hodges TM, et al. Impact of intracranial pressure monitor prophylaxis on central nervous system infections and bacterial multi-drug resistance. Surg Infect (Larchmt). 2008;9:503-508.
9. Denson K, Morgan D, Cunningham R, et al. Incidence of venous thromboembolism in patients with traumatic brain injury. Am J Surg. 2007;193:380-384.
10. Brain Trauma Foundation, American Association of Neurological Surgeons, Congress of Neurological Surgeons, et al. Guidelines for the management of severe traumatic brain injury. V. Deep vein thrombosis prophylaxis. J Neurotrauma. 2007;24(suppl 1):S32-S36.
11. Cothren CC, Smith WR, Moore EE, Morgan SJ. Utility of once-daily dose of low-molecular-weight heparin to prevent venous thromboembolism in multisystem trauma patients. World J Surg. 2007;31:98-104.
12. Norwood SH, Berne JD, Rowe SA, et al. Early venous thromboembolism prophylaxis with enoxaparin in patients with blunt traumatic brain injury. J Trauma. 2008;65:1021-1027.
13. Reiff DA, Haricharan RN, Bullington NM, et al. Traumatic brain injury is associated with the development of deep vein thrombosis independent of pharmacological prophylaxis. J Trauma. 2009;66:1436-1440.
14. Brain Trauma Foundation, American Association of Neurological Surgeons, Congress of Neurological Surgeons, et al. Guidelines for the management of severe traumatic brain injury. XIII. Antiseizure prophylaxis. J Neurotrauma. 2007;24(suppl 1):S83-S86.
15. Jones KE, Puccio AM, Harshman KJ, et al. Levetiracetam versus phenytoin for seizure prophylaxis in severe traumatic brain injury. Neurosurg Focus. 2008;25:E3.
16. Formisano R, Barba C, Buzzi MG, et al. The impact of prophylactic treatment on post-traumatic epilepsy after severe traumatic brain injury. Brain Injury. 2007;21:499-504.
To comment on this article, contact rdavidson@jobson.com.





