US Pharm. 2010;35(2):HS28-HS32.
Heart disease is a broad term that describes a range of diseases that affect the cardiovascular system and result in approximately 630,000 deaths annually in the United States.1 Cardiovascular disease (CVD) is the leading cause of death of both men and women above the age of 35 among all racial and ethnic groups.2 While risk factors such as increased age, male gender, and family history are nonmodifiable, others, such as smoking, high cholesterol, hypertension, physical inactivity, obesity, diabetes mellitus, and stress are preventable.2 Another possible modifiable risk factor, vitamin D deficiency, has been identified and has caused debates in the literature as the protective role of vitamin D on the cardiovascular system continues to be investigated. This article will expand on this proposed phenomenon, providing deeper insight into recently published literature, as well as discuss the pharmacist’s role in preventing vitamin D deficiency.
Vitamin D: The Basics
Vitamin D refers to two biologically inactive precursors: vitamin D3 (cholecalciferol), produced mainly in the skin post exposure to ultraviolet (UV) radiation, and vitamin D2 (ergocalciferol), produced exogenously and entering the circulation solely after gastrointestinal absorption.3 Both vitamin D2 and D3 require hydroxylation reactions in the liver and kidney, closely regulated by the parathyroid hormone (PTH), to form the biologically active metabolite of vitamin D, 1,25(OH)2D (calcitriol).4 Vitamin D status is best measured by 25(OH)D concentrations or levels and not calcitriol levels for several reasons, including longer half-life (~3 weeks compared to ~8 hours for calcitriol), higher circulating serum concentrations, and tight regulation of calcitriol by PTH, resulting in falsely elevated calcitriol levels despite vitamin D deficiency.5 Currently, there is no universally accepted “normal” measure of 25(OH)D levels; however, it has been suggested that concentrations above 30 ng/mL are associated with decreased fracture rates and maximal parathyroid suppression.6
Although the classic function of vitamin D has been to increase the intestinal absorption of calcium for proper bone health, its role in health maintenance is beginning to expand with the finding of vitamin D receptors (VDRs) in many cells throughout the body, including cardiomyocytes, vascular smooth muscle, and endothelium.5,7 Additionally, recent studies have found that individuals with vitamin D deficiency have increased incidence of CVD.7
Prevalence
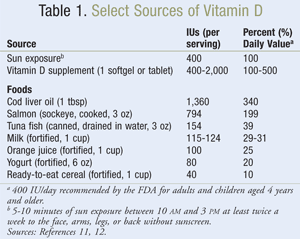
There is a large body of evidence indicating that vitamin D deficiency is more prevalent than previously anticipated. Data from the Third National Health and Nutrition Examination Survey (NHANES III) revealed that in the U.S., over 40% of men and over 50% of women are vitamin D deficient.8 Furthermore, this survey revealed that vitamin D deficiency (<30 ng/mL) was more common in women, elderly persons, and racial or ethnic minorities, and in patients with hypertension, diabetes mellitus, hypertriglyceridemia, and hypercholesterolemia. Other studies have identified that vitamin D deficiency is also present in up to 50% of young adults and healthy children.9 Unfortunately, there are limited foods that naturally contain vitamin D (e.g., salmon, mackerel, sardines, cod liver oil), and only a few foods are fortified with vitamin D (e.g., milk, orange juice, some breads and cereals), resulting in increased prevalence of deficiency in patients of all age groups in the U.S. and Europe.10 It should be noted that the risk of vitamin D deficiency is also dependent on geography, seasonality, latitude, and altitude, linking increased prevalence to reduced exposure to sunlight.4,5,10 Interestingly, CVD risk is also noted to be higher in areas of increased geographic latitude and during the winter months, which further supports the need for investigation of the relationship between vitamin D and CVD risk.9 Please refer to TABLE 1 for a summary of vitamin D sources.11,12
Hyperparathyroidism and Increased CVD Risk
There is strong evidence that in humans, secondary hyperparathyroidism (SHPT) in end-stage renal disease (ESRD) originates from vitamin D deficiency, resulting in increased risk for CVD.4,7,9 The concentrations of 25(OH)D are inversely related to PTH levels, with the threshold for PTH elevation being the 25(OH)D concentration of <30 ng/mL.9 As 25(OH)D concentrations decrease below 30 ng/mL, the decrease in calcium levels triggers PTH release, resulting in transport of calcium from bone and an increase in renal tubular calcium reabsorption in an attempt to elevate serum calcium levels, all of which inevitably leads to elevating renal production of 1,25(OH)D.7,9 Dialysis patients (and those with stage 4 or 5 chronic kidney disease [CKD]) with an estimated glomerular filtration rate of <30 mL/min/1.73 m2 often cannot produce sufficient amounts of 1,25(OH)D, resulting in SHPT. In patients with ESRD, SHPT is considered an important risk factor for the development of CVD.4 It has been observed that adjusted cardiovascular mortality is 10 to 20 times more likely to occur in patients receiving hemodialysis or peritoneal dialysis then in the general population.4
Although the link between ESRD and CVD is likely multifactorial, there is a large body of evidence that explains that the increase in CVD is related to calcification of the heart valves, mitral annulus, and myocardium.9,13-15 The effect of SHPT on CVD outcomes was demonstrated in a study that revealed that the incidence of CVD was two times higher in patients with a PTH level >250 pg/mL than in patients with PTH levels of <250 pg/mL.16 Another observational study in elderly individuals showed that CVD-related mortality rates doubled in those patients with elevated PTH, as compared to patients with normal PTH levels.17 Furthermore, studies that evaluated the repletion of vitamin D levels in patients with CKD have demonstrated improved outcomes. One such study found that in patients with ESRD who were undergoing hemodialysis, supplementation with 1alpha-hydroxyvitamin D3 at 0.25 to 1.5 mcg significantly lowered the risk of death from CVD (P = .003).18 Another recent study in patients with CKD and SHPT showed that supplementation with oral 1,25(OH)2D3 (calcitriol) also significantly improved overall survival.19
Atherosclerosis and Inflammation
Although the association of ESRD and increased CVD risk has been well documented, the effect of elevated PTH levels induced by vitamin D deficiency on CVD risk in the absence of ESRD continues to be under investigation. A recent cross-sectional study evaluated 654 adults between the ages of 55 and 96 years without a history of coronary heart disease, revascularization, or stroke. Results revealed that those with higher concentrations of 25(OH)D had a significantly dose-dependent decrease of the intima-media wall thickness of the carotid artery (P = .036), linking vitamin D deficiency to development of subclinical atherosclerosis.20 This finding is supported by research stating that vitamin D deficiency increases systemic inflammation, as confirmed by elevated levels of C-reactive protein and interleukin-10.4,6,9 In addition, the administration of vitamin D analogues has been shown to downregulate the inflammatory markers and reduce plaque production and instability.8,9
Hypertension
Vitamin D deficiency and insufficiency have been observed to upregulate the renin-angiotensin-aldosterone system (RAAS), resulting in hypertension, a well-known risk factor for CVD.21 In an animal study emulating vitamin D deficiency, vitamin D receptor knockout mouse models showed increased blood pressure, increased serum angiotensin-converting enzyme levels, and tissue renin content.8,22 Human studies have shown that 1,25(OH)2D has an inhibitory effect on renin synthesis, which decreases blood pressure.23 It has also been demonstrated that exposure to UVB, but not UVA, radiation on a regular basis both elevated serum 25(OH)D levels above 100 nmol/L and decreased blood pressure by 6 mmHg in hypertensive patients.24 (In this study, patients were exposed to UVB radiation in a tanning bed, 3 times per week, for a total of 3 months.) Another study in elderly women demonstrated that supplementation with vitamin D and calcium, compared to calcium alone, resulted in a significant increase in 25(OH)D concentrations (P <.01), a decrease in PTH levels (P <.05), a decrease in blood pressure by 9.3% (P <.025), and a decrease in heart rate by 5.4% (P <.025).25 Furthermore, the NHANES III study found that those individuals with higher serum 25(OH)D concentrations had a self-reported mean systolic blood pressure approximately 3 mmHg lower compared to patients with lower concentrations.8
In contrast, several smaller studies noted no such benefits of vitamin D supplementation on blood pressure decline.5 The Women’s Health Initiative study, recently conducted in the U.S., revealed that no blood pressure changes were noted in women randomized to receive vitamin D (400 IU) and calcium at the end of a 7-year follow-up period.26 It should be noted, however, that the vitamin D dose administered to study subjects was lower than recommended in clinical practice (for adults >70 years, supplementation is recommended to be at least 800 IU daily).27 In addition, the authors also reported that study subjects had low adherence to the study drug. Nevertheless, these results should not be ignored; further studies are needed to clarify the clinical importance of vitamin D supplementation as an antihypertensive agent.
Diabetes and Metabolic Syndrome
Revealed in many animal and human cross-sectional studies, another proposed mechanism for increased CVD risk is the association between vitamin D deficiency and obesity, glucose intolerance, and metabolic syndrome.28,29 It has been known for several years that a relationship exists between normal insulin secretion from the pancreatic beta-cells and vitamin D.4 It is proposed that the pancreas possesses the VDR gene and therefore has the ability to convert circulating 25(OH)D to 1,25(OH) 2D, resulting in increased insulin production.5,30 Studies have shown that allelic variation in the VDR gene increases the risk of developing both obesity and diabetes mellitus.31,32 Others have proposed that increased body fat results in sequestration of vitamin D in adipose tissue, lowering serum vitamin D concentrations and ultimately leading to insulin resistance and metabolic syndrome.33
One study observed that there was a 60% increase in insulin sensitivity in individuals with serum concentrations of 25(OH)D of 30 ng/mL compared to 10 ng/mL; in addition, concentrations <20 ng/mL were associated with decreased beta-cell function.34 The NHANES III study found that individuals with a 25(OH)D concentration of <21 ng/mL doubled their odds ratio for diabetes, compared to those with concentrations above 37 ng/mL.8 Furthermore, a study comparing daily intake of 800 IU of vitamin D versus <400 IU of vitamin D showed that those receiving higher doses of vitamin D daily reduced their risk of type 2 diabetes by one-third.35 Although the evidence is intriguing, it is limited, and future clinical trials are needed to determine the role of vitamin D in the prevention or treatment of diabetes and metabolic syndrome.
Correlation Between Vitamin D and Cardiovascular Events
Overall, the literature points to the observation that a low 25(OH)D concentration is closely associated with CVD. As previously discussed, several mechanisms explain the protective effects of vitamin D on the cardiovascular system: involvement of the vitamin D-PTH axis, the regulation of inflammation with its link to atherosclerosis, the regulation of the RAAS system, and the effect on insulin secretion, insulin sensitivity, and metabolic syndrome. Understanding these mechanisms assists pharmacists and other health care providers in interpreting recent studies that evaluate vitamin D deficiency and adverse cardiovascular events.
A recent investigation of the general U.S. population found that 25(OH)D deficiency (lowest quartile, <17.8 ng/mL) increased the risk of all-cause mortality by 26%, and although not statistically significant, increased the risk of CVD mortality in this group as well.28 Data from NHANES (2001-2004) revealed that adults with lower 25(OH)D concentrations were more likely to have CVDs such as coronary heart disease, heart failure, stroke, and peripheral arterial disease.36 It was discussed, however, that lower vitamin D levels might have been observed in patients with heart failure and heart disease due to limited physical activity, leading to decreased sunlight exposure, suggesting that lower vitamin D was a result of CVD, and not vice versa. However, another prospective observational study, with a mean follow-up period of 5.4 years, found that subjects with low vitamin D levels without CVD at baseline had a 53% to 80% higher rate of a CVD end point (fatal or nonfatal myocardial infarction [MI], ischemia, stroke, and heart failure).30 Additionally, in the Health Professionals Follow-up Study, it was found that men without CVD at baseline but with vitamin D deficiency, defined as 25(OH)D <15 ng/mL, experienced a two-fold increased rate of MI during a 10-year follow-up period.37
Although these trials are important stepping-stones to our insight into this widely debated topic in the literature, future research is necessary to reveal the relationship between vitamin D and cardiovascular health.
The Pharmacist’s Role
There are currently no universal guidelines that standardize screening, prevention, or treatment of vitamin D deficiency for patients at risk for CVD. There are, however, recommendations for serum 25(OH)D concentrations that have been found to maintain optimal bone health and prevent rickets.8 Based on the current literature, pharmacists can assist in identifying patients at highest risk for vitamin D deficiency and provide recommendations for appropriate screening and treatment. As mentioned earlier, few foods naturally contain vitamin D, although some are fortified; thus, dietary consumption of vitamin D is not the optimal choice to increase vitamin D levels (Table 1).4,10 UVB exposure, on the other hand, is the best natural source of vitamin D.4 It is documented that “sensible” sun exposure (defined as 5-10 minutes of exposure of arms and legs, or hands, arms, and face, 2 to 3 times per week) is recommend daily to maintain adequate vitamin D levels.4 However, since there is a growing concern about any exposure to sunlight causing skin damage, including skin cancer and wrinkling, this recommendation is extremely controversial.10
It has been found that patients who do not receive adequate exposure to sunlight, as well those patients who are obese, use sunscreen, cover all of the skin with clothing, have skin pigmentation, are elderly, use certain medications (e.g., anticonvulsants, glucocorticoids, drugs that activate the steroid and xenobiotic receptor), or have hepatic failure and intestinal-fat malabsorption syndrome, all have an increased risk for vitamin D deficiency.7,38 For these patients it is recommended that 25(OH)D concentrations be checked yearly, preferably at the end of the fall season, to help reveal vitamin D deficiency.10
The FDA currently recommends 400 IU daily for children and adults of all ages.39 However, it has been found that this supplementation is inadequate and that most people, especially elderly adults and those living in higher altitudes and in extreme winter climates, need at least 800 to 2,000 IU of vitamin D daily to achieve optimal supplementation.9 It would be beneficial if future trials used adequate dosages (at least 800 IU) to determine the effect of vitamin D supplementation on potential benefits in CVD prevention. Prospective studies are warranted to assess the effect of vitamin D on the mortality risk associated with CVD. Until then, pharmacists will need to continue educating health care professionals and patients on the available data regarding this potential association.
REFERENCES
1. Heart disease. Centers for Disease Control and Prevention. www.cdc.gov/heartdisease/. Accessed December 1, 2009.
2. Risk factors and coronary heart disease. American Heart Association. www.americanheart.org/print_presenter.jhtml?identifier=4726. Accessed December 1, 2009.
3. Wolpowitz D, Gilchrest BA. The vitamin D questions: how much do you need and how should you get it? J Am Acad Dermatol. 2006;54:301-317.
4. Zittermann A. Vitamin D and disease prevention with special reference to cardiovascular disease. Prog Biophys Mol Biol. 2006;92:39-48.
5. Judd SE, Tangpricha V. Vitamin D deficiency and risk for cardiovascular disease. Am J Med Sci. 2009;338:40-44.
6. Michos ED, Melamed ML. Vitamin D and cardiovascular disease risk. Curr Opin Clin Nutr Metab Care. 2008;11:7-12.
7. Holick MF. Vitamin D deficiency. N Engl J Med. 2007;357:266-281.
8. Martins D, Wolf M, Pan D, et al. Prevalence of cardiovascular risk factors and the serum levels of 25-hydroxyvitamin D in the United States: data from the Third National Health and Nutrition Examination Survey. Arch Intern Med. 2007;167:1159-1165.
9. Lee JH, O’Keefe JH, Bell D, et al. Vitamin D deficiency: an important, common, and easily treatable cardiovascular risk factor? J Am Coll Cardiol. 2008;52:1949-1952.
10. Holick MF. Sunlight and vitamin D for bone health and prevention of autoimmune disease, cancers, and cardiovascular disease. Am J Clin Nutr. 2004;80(suppl 6):1678S-1688S.
11. Vitamin D. www.drugstore.com. Accessed January 8, 2010.
12. Dietary supplement fact sheet: vitamin D. Office of Dietary Supplements. National Institutes of Health. http://dietary-supplements.info.nih.gov/factsheets/vitamind.asp#h3. Accessed January 8, 2010.
13. Raggi P, Kleerekoper M. Contribution of bone and mineral abnormalities to cardiovascular disease in patients with chronic kidney disease. Clin J Am Soc Nephrol. 2008;3:836-843.
14. Andersson P, Rydberg E, Willenheimer R. Primary hyperparathyroidism and heart disease—a review. Eur Heart J. 2004;25:1776-1787.
15. Zittermann A, Schleithoff SS, Koerfer R. Vitamin D and vascular calcification. Curr Opin Lipidol. 2007;18:41-46.
16. Soubassi LP, Chiras TC, Papadakis ED, et al. Incidence and risk factors of coronary heart disease in elderly patients on chronic haemodialysis. Int Urol Nephrol. 2006;38:795-800.
17. Bjorkman MP, Sorva AJ, Tilvis RS. Elevated serum parathyroid hormone predicts impaired survival prognosis in a general aged population. Eur J Endocrinol. 2008;158:749-753.
18. Shoji T, Shinohara K, Kimoto E, et al. Lower risk for cardiovascular mortality in oral 1alpha-hydroxy vitamin D3 users in a haemodialysis population. Nephrol Dial Transplant. 2004;19:179-184.
19. Kovesdy CP, Ahmadzadeh S, Anderson JE, Kalantar-Zadeh K. Association of activated vitamin D treatment and mortality in chronic kidney disease. Arch Intern Med. 2008;168:397-403.
20. Reis JP, Muhlen D, Michos ED, et al. Serum vitamin D, parathyroid hormone levels, and carotid atherosclerosis. Atherosclerosis. 2009;207;858-590.
21. Pilz S, Tomaschitz A, Ritz E, et al. Vitamin D status and arterial hypertension: a systemic review. Cardiol. 2009;6:621-630.
22. Simpson RU, Hershey SH, Nibbelink KA. Characterization of heart size and blood pressure in the vitamin D receptor knockout mouse. J Steroid Biochem Mol Biol. 2007;103:521-524.
23. Li YC. Vitamin D regulation of the renin-angiotensin system. J Cell Biochem. 2003;88:327-331.
24. Krause R, Buhring M, Hopfenmuller W, et al. Ultraviolet B and blood pressure. Lancet. 1998;352:709-710.
25. Pfeifer M, Begerow B, Minne HW, et al. Effects of a short-term vitamin D3 and calcium supplementation on blood pressure and parathyroid hormone levels in elderly women. J Clin Endocrinol Metab. 2001;86:1633-1637.
26. Lacroix AZ, Kotchen J, Anderson G, et al. Calcium plus vitamin D supplementation and mortality in postmenopausal women: the Women’s Health Initiative calcium-vitamin D randomized controlled trial. J Gerontol A Biol Sci Med Sci. 2009;64:559-567.
27. Michos ED, Blumenthal RS. Vitamin D supplementation and cardiovascular disease risk. Circulation. 2007;115:827-828.
28. Melamed ML, Michos ED, Post W, Astor B. 25-hydroxyvitamin D levels and the risk of mortality in the general population. Arch Intern Med. 2008;18:1629-1637.
29. Tai K, Need AG, Horowitz M, et al. Vitamin D, glucose, insulin, and insulin sensitivity. Nutrition. 2008;24:279-285.
30. Wang TJ, Pencina MJ, Booth SL, et al. Vitamin D deficiency and risk of cardiovascular disease. Circulation. 2008;117:503-511.
31. Ford ES, Ajani UA, McGuire LC, Liu S. Concentration of serum vitamin D and the metabolic syndrome among U.S. adults. Diabetes Care. 2005;5:1228-1230.
32. Ortlepp JR, Lauscher J, Hoffmann R, et al. The vitamin D receptor gene variant is associated with the prevalence of type 2 diabetes mellitus and coronary artery disease. Diabet Med. 2001;18:842-845.
33. Martini LA, Wood RJ. Vitamin D status and the metabolic syndrome. Nutr Rev. 2006;64:479-486.
34. Chiu KC, Chu A, Go VL, Saad MF. Hypovitaminosis D is associated with insulin resistance and beta cell dysfunction. Am J Clin Nutr. 2004;79:820-825.
35. Pittas AG, Dawson-Hughes B, Li T, et al. Vitamin D and calcium intake in relation to type 2 diabetes in women. Diabetes Care. 2006;29:650-656.
36. Kim DH, Sabour S, Saga UN, et al. Prevalence of hypovitaminosis D in cardiovascular diseases (from the National Health and Nutrition Examination Survey 2001 to 2004). Am J Cardiol. 2008;102:1540-1544.
37. Giovannucci E, Liu U, Hollis BW, et al. 25-hydroxyvitamin D and risk of myocardial infarction in men: a prospective study. Arch Intern Med. 2008;168:1174-1180.
38. Holick MF. High prevalence of vitamin D inadequacy and implication for health. Mayo Clin Proc. 2006;81:353-373.
39. Huh SY, Gordon CM. Vitamin D deficiency in children and adolescents: epidemiology, impact and treatment. Rev Endocr Metab Disord. 2008;9:161-170.
To comment on this article, contact rdavidson@uspharmacist.com.