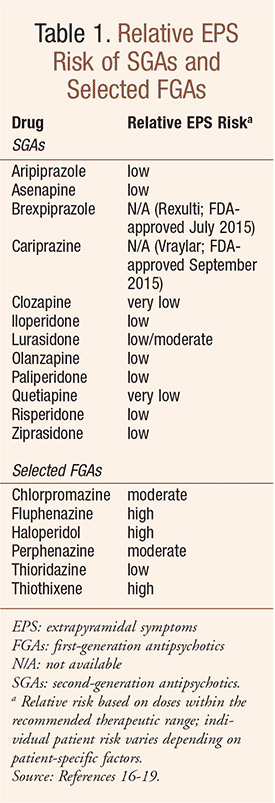
Second-generation antipsychotic agents (SGAs, TABLE 1), also referred to as atypical antipsychotics, are widely used in the United States in the treatment of schizophrenia, psychotic episodes, and other conditions. These agents, as compared with first-generation antipsychotics (FGAs), are associated with a dramatic decline in drug-induced movement disorders (DIMDs) such as akathisia (subjective motor restlessness), dystonia (dyskinetic muscle tonicity), parkinsonism, and tardive dyskinesia (TD; TABLE 2).1

However, the use of SGAs for populations that were antipsychotic agent-naïve, (i.e., elderly, adolescents, children) and an ever-widening adult population with conditions such as bipolar disorder and major depressive disorder, together with a lack of long-term clinical trials that would precisely indicate the rate of DIMD in these populations, portends an increase in the actual number of cases.2
In addition, the literature on identifiable risk factors associated with treatment-emergent movement disorders is highly variable.3 Recognition of DIMDs is essential in the treatment of these disorders.3 Pharmacists can play a role not only in identifying cases of DIMDs, but importantly, in guiding appropriate SGA dosing in light of their continued widening use in vulnerable populations such as the elderly, in the hope of reducing the incidence of, or preventing, these disorders.2
With the recent FDA approval of the newest SGA, cariprazine (Vraylar) capsules to treat schizophrenia and bipolar disorder in adults, reviewing the issues related to DIMDs is timely indeed. The development of a DIMD often reduces the quality of life in a geriatric patient.3 In particular, elderly patients with dementia are at greater risk than patients without dementia for persistent, drug-induced extrapyramidal symptoms (EPS). Sedation, orthostatic hypotension, anticholinergic effects, and akathisia may occur in up to 20% of elderly patients taking an antipsychotic agent, and drug-induced parkinsonism can persist for 6 to 9 months after the agent is discontinued.4 Further, tens of thousands of patients previously exposed to more potent antipsychotic agents have TD, which remains untreatable and irreversible.1 Of additional concern, the medications used in the management of DIMDs, including anticholinergic (antiparkinsonism) agents, benzodiazepines, and beta-blockers, require careful consideration in this vulnerable population.1,5
FGAs and SGAs: Focus on Mechanism of Action
In general, antipsychotic drugs block at dopaminergic and serotonergic receptors as well as at adrenergic, cholinergic, and histamine-binding neuroreceptors.6 FGAs are more likely associated with movement disorders, particularly for drugs that bind tightly to dopamine receptors (D2), such as haloperidol, and less likely for those agents that bind weakly, such as chlorpromazine.7 The SGAs block dopamine receptors more selectively than conventional FGAs.7
According to Caroff, the dominance in clinical practice of the newer SGAs has significantly reduced the frequency of EPS, whether by a reduced liability for EPS because of weaker dopamine receptor–binding affinity or a more complex “atypical” mechanism affecting multiple neurotransmitters.1 According to Schulz, although greater serotonergic binding was initially thought to contribute to the efficacy of SGAs, studies suggest this binding is unrelated to efficacy or adverse-effects profile; SGAs also do the following7:
• Have the tendency to alleviate positive symptoms (e.g., delusions, hallucinations)
• Potentially lessen negative symptoms to a greater extent than do FGAs (although clinicians may question this difference)
• Potentially cause less cognitive blunting
• Are less likely to have extrapyramidal adverse effects
• Are associated with a lower risk of TD
• Increase prolactin slightly or not at all (except risperidone)
Of note, while SGAs have fewer EPS than FGAs (TABLE 1), SGAs are associated with a higher risk of metabolic side effects, such as diabetes, hyperlipidemia, and weight gain.6,7
The Clinical Antipsychotic Trials of Intervention Effectiveness (CATIE) Schizophrenia Trial provided additional perspective on the relative risks and clinical value of the SGAs and FGAs; the study showed that perphenazine, a mid-potency FGA used at modest doses, was not significantly different from four SGAs in the risk of developing EPS.1 Caroff indicates that the dichotomy between FGAs and SGAs and the “atypical” concept based on liability of EPS may be oversimplified; instead, he suggests “antipsychotic drugs could be conceptualized as a single drug class with a spectrum of risk for EPS depending upon receptor-binding affinities, especially for dopamine and acetylcholine receptors, and individual patient susceptibility.”1
Schizophrenia and Antipsychotics in the Elderly
The average age at onset of schizophrenia is early to mid-20s in women and somewhat earlier in men; about 40% of males have their first episode before age 20.7 With regard to the elderly, late-life onset schizophrenia, sometimes referred to as paraphrenia, may occur.7 Most geriatric patients treated for schizophrenia, however, have been living with the symptoms for decades, including psychosis (loss of contact with reality), hallucinations (false perceptions), delusions (false beliefs), disorganized speech and behavior, flattened affect (restricted range of emotions), cognitive deficits (impaired reasoning and problem solving), and occupational and social dysfunction.7
Furthermore, large numbers of geriatric patients previously treated with haloperidol and other FGAs have TD; these agents are still used in psychiatry; in medical settings for delirium and agitation; and in developing nations.1
While all antipsychotic agents can reduce paranoia, they have the potential to worsen confusion; elderly patients, especially women, are at increased risk of TD, which is often irreversible.4 Since antipsychotics are considered a drug category of concern in the elderly, according to Ruscin and Linnebur, antipsychotics should be used only for psychosis; in nonpsychotic, agitated patients, they indicate that antipsychotics control symptoms only marginally better than placebo and can have severe adverse effects.4
In people with dementia, studies have shown antipsychotics increased mortality and risk of stroke, moving the FDA to issue a black box warning on their use in this population; in general, dementia-related behavior problems, such as wandering, yelling, and uncooperativeness, do not respond to antipsychotics.4
Regarding further caution in the elderly, antipsychotics may exacerbate or cause syndrome of inappropriate antidiuretic hormone secretion or hyponatremia; it is recommended that sodium levels be closely monitored when starting or changing dosages in older adults.8 When an antipsychotic agent is used with two or more CNS-active drugs (antipsychotics; benzodiazepines; nonbenzodiazepines, benzodiazepine receptor agonist hypnotics; tricyclic antidepressants, selective serotonin reuptake inhibitors, and opioids), there is an increased risk of falls; avoid a total of three or more CNS-active drugs and minimize the number of CNS-active drugs.8
SGA Dosing in the Geriatric Patient
When initiating an antipsychotic agent in an elderly person, the initial dose should be about one- quarter of the usual starting adult dose; it should then be increased gradually with frequent monitoring for response and adverse effects.4 Upon clinical response by the patient, the dose should be decreased, if possible, to the lowest effective dose; it should be discontinued if it is ineffective. Clinical trial data are limited with regard to dosing, efficacy, and safety of these drugs in the elderly.4 As previously mentioned, elderly patients with dementia are at greater risk than patients without dementia for persistent drug-induced EPS.1
Drug-Induced Movement Disorders: EPS
Management requires cooperation from the patient, family, and other caregivers that can be fostered early in the patient’s care through appropriate informed consent. According to Caligiuri, the risks and benefits of antipsychotic treatment in the elderly patient need to be communicated to the patient and the family.3 Once TD develops, there is at present no consistently effective treatment; therefore, attention should focus on its prevention and close monitoring. The pharmacological management of DIMDs requires careful consideration of the precautions and warnings associated with interventions such as anticholinergics, beta-blockers, and benzodiazepines. Refer to the 2015 American Geriatrics Society Updated Beers Criteria for Potentially Inappropriate Medication Use in Older Adults for guidance.8 DIMDs include:
Akathisia (i.e., motor restlessness), the most common extrapyramidal symptom, is often mistaken for anxiety or exacerbation of psychosis, and remains a problem even with newer SGA drugs.1,5 The desire to be in motion and the subsequent inability to sit or stand still, resulting in pacing, is so distressing and frightening to patients that it may induce suicidality.5 As indicated in a representative sample of trials, akathisia developed at an incidence rate of about two to seven times higher with haloperidol (15% to 40%) when compared with SGAs (0% to 12%).1 Of note, cigarette smoking in women has been associated with increased incidence.5
While risk factors for akathisia have not been well established, the following may entail greater risk: increasing age, female gender, negative symptoms, cognitive dysfunction, iron deficiency, history of akathisia, concomitant parkinsonism, and mood disorders.9,10 In most clinical trials, SGAs have been shown to have a significantly lower incidence of akathisia compared with FGAs.11
While this EPS usually occurs early in the course of antipsychotic treatment, it may persist even after the offending agents are discontinued. While anticholinergic agents (e.g., trihexyphenidyl 2-5 mg orally three time daily) are used to treat akathisia, a benzodiazepine (e.g., clonazepam 0.5-1 mg orally three times daily) is often considered first-line treatment; propranolol 30-80 mg orally/d, diazepam 5 mg orally three times daily, or amantadine 100 mg orally three times daily may be required in resistant cases.5,7 Refer to Reference 19 for geriatric dosing considerations.
Acute dystonias, sustained contractions of muscles leading to twisting, distorted postures, usually occur early in the course of antipsychotic therapy, with younger patients bearing the highest risk.5,6 Late occurrence is seen in individuals who had previously suffered early severe dystonic reactions and an affective disorder, which underscores the importance of thorough medical and medication history-taking in the development of pharmacotherapy care plans. Bizarre muscle spasms of the head, neck, and tongue are most common: torticollis, oculogyric crisis, difficulties in chewing and swallowing, and spasms of the masseter muscle. Particularly dangerous is spasmodic closure of the larynx. While dystonia is less frequent than in years past, it may be encountered with high-potency antipsychotic agents (e.g., haloperidol).1 For acute episodes, diphenhydramine 50 mg is administered intramuscularly; this is followed by the mildly sedating benztropine mesylate, 2 mg orally twice daily, for several weeks, followed by gradual tapering.5
Parkinsonism secondary to drug therapy is reversible, although not distinguishable from idiopathic parkinsonism. Parkinsonism is quite debilitating and more likely to be seen when antipsychotics are used in elderly patients.1 Characteristically, patients present with apathy, akinesia of the face and arms (i.e., appearing as depression), rigidity, salivation, postural abnormalities, shuffling gait, and pill-rolling tremor of the hands; catatonia may also present.5 Symptoms frequently can be eliminated with a reduction in dose or change to a lower-potency antipsychotic; an anticholinergic, while helpful for drooling, can cause confusion and delirium; however, one may be added if necessary.12 While maximum therapeutic response is usually seen in 3 to 10 days, more severe symptoms may take longer (e.g., 4-6 weeks) to respond. In the elderly, anticholinergics are not for use as prophylaxis against EPS. Amantadine use requires dosing based on renal function. Quetiapine may be a useful SGA alternative for this population.12
Tardive dyskinesia (TABLE 2), is more common in female patients, and age is the most consistent risk factor.6,7 Other risk factors, including previous brain injury and alcoholism, are outlined in TABLE 3. TD may be seen in up to 30% of patients receiving long-term FGAs.13-15

While TD is most often mild, it is irreversible, with limited options for treatment.1 Due to this risk of TD, patients receiving long-term maintenance therapy should be evaluated at least every 6 months; rating instruments may be used.7
While beyond the scope of this topic, a proposed treatment algorithm for TD may be found in Reference 1.
Neuroleptic malignant syndrome (NMS) is an abrupt, life-threatening, idiosyncratic patient response to a therapeutic dose of an antipsychotic agent; intramuscular administration is a risk factor.6 NMS represents an extremely rare form of EPS combining features of advanced parkinsonism and catatonia; symptoms include exhaustion, agitation, hyperthermia (>38ºC), mental status change, muscle rigidity and other movement disorders, autonomic dysregulation, and elevated creatine kinase (CK).1,7 Lithium, in combination with an antipsychotic, may increase vulnerability.5 NMS requires control of fever and fluid support. The following have been used as treatment, although controversy still exists regarding them: bromocriptine, amantadine, dantrolene, calcium channel blockers, and benzodiazepines; ECT is used in resistant cases, while clozapine is used as an SGA for patients with a history of NMS.5 Patients and caregivers should be counseled about this adverse reaction and made aware that it requires emergency medical attention.
Conclusion
The development of a DIMD often reduces the quality of life in a geriatric patient. Pharmacists have an opportunity to implement recommendations to appropriately individualize selection and dosing of an antipsychotic agent, monitor and reassess benefit and risk periodically, and recommend deprescribing where appropriate. Those who are antipsychotic-naïve and especially those who are vulnerable with regard to risk, such as the elderly, often require particular attention.
REFERENCES
1. Caroff SN, Hurford I, Lybrand J, et al. Movement disorders induced by antipsychotic drugs: implications of the CATIE schizophrenia trial. Neurol Clin. 2011; 29(1):127-viii. www.ncbi.nlm.nih.gov/pmc/articles/PMC3018852.
2. Factor SA, Leffler JB, Murray CF. Drug-induced movement disorders: a clinical review. Medscape, 2009. www.medscape.org/viewarticle/586881.Accessed September 22, 2015.
3. Caligiuri MR, Jeste DV, Lacro JP. Antipsychotic-induced movement disorders in the elderly: epidemiology and treatment recommendations. Drugs Aging. 2000;17(5):363-384.
4. Ruscin M, Linnebur SA. Drug categories of concern in the elderly. Merckmanuals.com. Last full review/revision June 2014. www.merckmanuals.com/professional/geriatrics/drug-therapy-in-the-elderly/drug-categories-of-concern-in-the-elderly. Accessed October 27, 2015.
5. Williams N, DeBattista C. Psychiatric disorders. In: Papadakis MA, McPhee SJ, Rabow MW, eds. 2016 Current Medical Diagnosis & Treatment. 55th ed. New York, NY: McGraw Hill Medical; 2015:1050-1056.
6. Harvey RA. Pharmacology. 5th ed. Lippincott Williams & Wilkins: Philadelphia, PA. 2012:161-168.
7. Schulz, C. Schizophrenia. Merckmanuals.com. Last full review/revision July 2013. www.merckmanuals.com/professional/psychiatric-disorders/schizophrenia-and-related-disorders/schizophrenia. Accessed October 27, 2015.
8. American Geriatrics Society 2015 Updated Beers Criteria for potentially inappropriate medication use in older adults. American Geriatrics Society 2015 Beers Criteria Update Expert Panel. Published Oct 8, 2015. http://onlinelibrary.wiley.com/doi/10.1111/jgs.13702/full. Accessed October 27, 2015.
9. Bratti IM, Kane JM, Marder SR. Chronic restlessness with antipsychotics. Am J Psychiatry. 2007;164(11):1648-1654.
10. Poyurovsky M. Acute antipsychotic-induced akathisia revisited. Br J Psychiatry. 2010;196(2):89-91.
11. Kane JM, Fleischhacker WW, Hansen L, et al. Akathisia: an updated review focusing on second-generation antipsychotics. J Clin Psychiatry. 2009;70(5):627-643.
12. Mathews M, Gratz S, George V, et al. Antipsychotic-induced movement disorders: evaluation and treatment. Psychiatry (Edgmont). 2005;2(3):36-41.
13. Jeste DV, Caliguiri MP. Tardive dyskinesia. Schizophrenia Bull. 1993;19:303-315.
14. Wojcik JD, Gelenberg A, LaBrie RA, et al. Prevalence of TD in an out-patient population. Compr Psychiatry. 1980;21:370-380.
15. Barnes TRE. Movement disorders associated with antipsychotic drugs: the tardive syndromes. Int Rev Psychiatry. 1990;2:355-366.
16. Woods SW. Chlorpromazine equivalent doses for the newer atypical antipsychotics. J Clin Psychiatry. 2003;64(6):663-667.
17. Vraylar Full Prescribing Information. Actavis Pharma, Inc. Parsippany, NJ. 2015. http://pi.actavis.com/data_stream.asp?product_group=2028&p=pi&language=E. Accessed October 19, 2015.
18. Rexulti Full Prescribing Information. Otsuka Pharmaceutical Co., Ltd., Tokyo, 101-8535 Japan. July 2015. www.rexulti.com. Accessed September 18, 2015.
19. Semla TP, Beizer JL, Higbee MD. Geriatric Dosage Handbook. 19th ed. Hudson, OH: Lexicomp; 2014.
To comment on this article, contact rdavidson@uspharmacist.com.





