US Pharm. 2016; 41(10):15-17.
Sinus bradycardia, also known as bradyarrhythmia, is medically defined as a sinus rhythm with a resting heart rate of <60 beats per minute (BPM) in adults.1 It is often asymptomatic and may be caused by sinus node dysfunction (SND)—most often seen in elderly patients with concomitant cardiovascular disease or diabetes—or by certain medications, including beta-adrenergic antagonists (i.e., beta-blockers).1,2 Though usually prescribed for cardiovascular conditions, beta-blockers found in topical ophthalmic formulations commonly used to treat glaucoma can induce bradycardia, or increase its risk in persons who have particular medical conditions or are taking certain other drugs. Pharmacists can identify those at risk and assist in the close monitoring of patients in order to minimize the potentially life-threatening adverse effects associated with these drugs.
Bradycardia: Causes, Symptoms, and Complications
Sinus bradycardia typically does not cause symptoms (TABLE 1), until the heart rate drops below 50 BPM; when symptomatic, it may cause dizziness, fatigue, weakness, and, at very low rates, syncope.1 Often an incidental finding in an otherwise healthy individual, the causes of sinus bradycardia include the following:

Sinus Node Dysfunction: Seen most commonly, SND involves a disturbance in the ability of the sinus node to generate or transmit an action potential to the atria. Conditions that occur frequently in the elderly may contribute to such a conduction disturbance (TABLE 2), including age-related myocardial tissue damage, hypertension, hypothyroidism, and certain medications.3 SND follows an unpredictable course and is associated with a number of conditions causing physiologically inappropriate atrial rates.2 Although some patients manifest SND as a result of electrical remodeling induced by periods of atrial fibrillation (AF), others develop progressive atrial structural remodeling that gives rise to both conditions together.4 In persons with AF, a pacemaker placement will regulate heart rhythm in patients who manifest non-drug-induced cerebral symptoms (e.g., dizziness, syncope) secondary to ventricular pauses; in AF patients with drug-induced symptomatic bradycardia, the causative drug is removed prior to pacemaker placement.5

Medications: Those most commonly responsible for bradycardia include therapeutic and supratherapeutic doses of digitalis glycosides, beta-blockers, and calcium channel-blockers; other cardiac drugs include class I antiarrhythmic agents and amiodarone. A broad variety of other drugs including lithium, paclitaxel, toluene, dimethyl sulfoxide (DMSO), topical ophthalmic acetylcholine, fentanyl, alfentanil, sufentanil, reserpine, and clonidine can also cause bradycardia.1
Other Causes: Conditions such as hypothermia, hypoglycemia, and sleep apnea, and, less commonly, diphtheria, rheumatic fever, and viral myocarditis can cause bradycardia.1
Complications of untreated bradycardia vary, and they depend upon the extent to which the heart rate has decreased, the location of the conduction problem, and the type of myocardial tissue damage.3 Potential complications of bradycardia include frequent syncope, heart failure, and sudden cardiac arrest or sudden death.3 It is recommended that older patients with heart disease and unexplained syncope undergo an electrophysiological study.5 Of note, theophylline and hydralazine are options that are used to increase the heart rate in healthy, younger patients who have bradycardia without syncope.2
Ophthalmic Beta-Blockers: Symptomatic Bradycardia
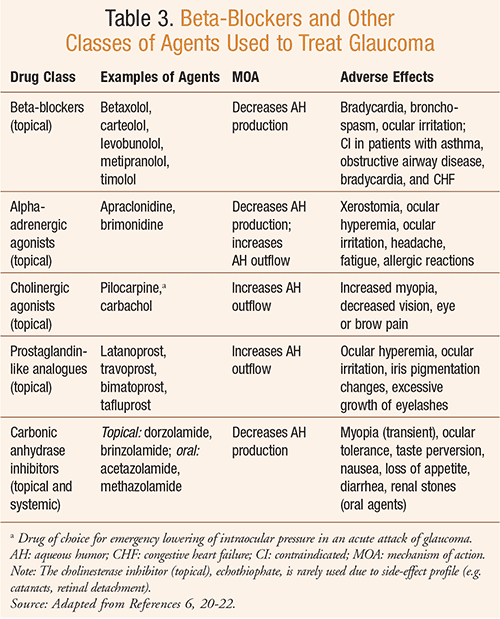
The majority of drugs used to treat glaucoma (TABLE 3; Patient Resources), generally considered a disease of the aging eye, are in topical ophthalmic form and may cause numerous untoward systemic effects in the elderly.6 While it is often believed that topical administration of ophthalmic preparations safeguards against harmful systemic effects, the reality is that approximately 80% of the drug in the ophthalmic products is systemically absorbed and first-pass metabolism is avoided.7 While systemic concentration after administration of topical beta-blockers is low in comparison to that achieved with oral beta-blockers, it is well known that topical therapy can induce cardiovascular, respiratory, central nervous system, and metabolic side effects.8,9
Topical application of ophthalmic timolol is widely prescribed in the treatment of glaucoma either alone or in combination ophthalmic formulations, many of which have been launched relatively recently.7 Ophthalmic timolol may cause serious adverse effects, such as symptomatic bradycardia, various conduction disorders in the heart, orthostatic hypotension, syncope and falls, all of which can be particularly troublesome in the frail elderly.7
Despite evidence linking the use of topical beta-blockers to bradycardia, one large study of elderly patients—a bradycardia hospitalization cohort—with a mean age of 82, found that codispensing of ophthalmic beta-blockers with medications that can cause or exacerbate bradycardia (e.g., oral beta-blockers, calcium channel blockers, digoxin, and antiarrhythmics) is common.10 The researchers noted that bradycardia is a potential adverse event following the initiation of timolol, and they recommend that healthcare providers carefully consider patient history before choosing a glaucoma regimen (TABLE 3). As a means of minimizing adverse effects and avoiding hospitalizations, close monitoring is required after treatment initiation with a topical nonselective beta-blocker ophthalmic preparation.10 The researchers had previously found that 36% of individuals with glaucoma who were dispensed the calcium channel blocker verapamil were codispensed ophthalmic timolol, a contraindication that can exacerbate bradycardia.11,12
Lewis found that healthy elderly volunteers treated with levobunolol showed significantly lower average heart rates during exercise and the postexercise recovery period, compared with people treated with betaxolol or placebo.13 Case reports also provide evidence of bradycardia caused by levobunolol.14 Further, Chun et al indicated that topical administration of levobunolol could cause syncope, bradycardia, and first-degree sinoatrial block.15 One of the most frequently prescribed first-line topical therapies for the reduction of intraocular pressure was found to be timolol, and case reports attribute bradycardia to topical timolol, reportedly owing to systemic absorption.16,17
Pratt et al indicate that interventions with prescribers are necessary to raise awareness of these potential adverse events.10 The researchers point out that the majority of glaucoma agents are prescribed by ophthalmologists, while adverse events may be managed by primary care practitioners; challenges are sure to be raised regarding how to address these adverse drug events across the continuum of care.10 In order to optimize patient care with improved communication among ophthalmologists, primary care physicians, pharmacists, and patients regarding the history of cardiac diseases and glaucoma treatment, the researchers emphasize the need for cross-specialty cooperation.10 They summarize that bradycardia is a potential adverse event following timolol initiation; practitioners should carefully review the patient history—with an awareness of comorbidities—before selecting a glaucoma regimen, and closely monitor patients after treatment initiation with topical nonselective beta-blocker ophthalmic drops to minimize adverse events and potentially avoid hospitalizaions.10,11
Beta-Blocker Toxicity
Beta-blockade is one of the toxic systemic (beta-blocking) effects of topical ophthalmic timolol found to be stronger and longer lasting in the elderly.10 Severe beta-blocker toxicity is defined as bradycardia with associated hypotension and shock (systolic blood pressure <80 mm Hg, heart rate <60 BPM). Other than the direct effects of the beta-adrenoreceptor blockade, toxicity may result from other mechanisms, including sodium and calcium channel blockade, centrally mediated cardiac depression, and alteration of cardiac myocyte energy metabolism.18
In beta-blocker toxicity, the goal of therapy is the restoration of perfusion to critical organ systems by increasing cardiac output; this may be accomplished by improving myocardial contractility, increasing heart rate, or both.19 Owing to the potential for rapid deterioration, only asymptomatic patients—observed for a period of 6 hours—should be considered stable for transfer; if intensive-care monitoring or therapy is not available, the unstable patient should be transferred to the closest medical center with the necessary capabilities for care, including a medical toxicologist.19 For a detailed discussion of the treatment for beta-blocker toxicity, refer to Reference 19.
Conclusion
Topical beta-blocker ophthalmic agents are effective treatments for glaucoma; however, they may often be an unrecognized cause of bradycardia in the frail elderly and individuals with cardiac disease.14 Pharmacists must engage patients and their caregivers in such a way as to elicit complete medication lists, including any and all topical ophthalmic preparations that might be overlooked, with the aim of minimizing adverse events and potentially avoiding hospitalizations.
REFERENCES
1. Livingston MW. Sinus bradycardia. Medscape.com. Updated December 18, 2015. http://emedicine.medscape.com/article/760220-overview?src=refgatesrc1#showall. Accessed September 14, 2016.
2. Mitchell LB. Sinus node dysfunction (sick sinus syndrome). Merckmanual.com. Last full review/revision July 2015. www.merckmanuals.com/professional/cardiovascular-disorders/arrhythmias-and-conduction-disorders/sinus-node-dysfunction. Accessed September 13, 2016.
3. Bradycardia. Mayoclinic.org. May 7, 2014. www.mayoclinic.org/diseases-conditions/bradycardia/basics/causes/con-20028373. Accessed September 12, 2016.
4. John RM, Kumar S. Sinus node and atrial arrhythmias. Circulation. 2016;133(19):1892-1900.
5. Aronow WS. Cardiac arrhythmias. In: Fillit HM, Rockwood K, Woodhouse K, eds. Brocklehurst’s Textbook of Geriatric Medicine and Gerontology. 7th ed. Philadelphia, PA: Saunders Elsevier; 2010:327-337.
6. Novack GD, O’Donnell MJ, Molloy DW. New glaucoma medications in the geriatric population: efficacy and safety. J Am Geriatr Soc. 2002;50(5):956-962.
7. Mäenpää J, Pelkonen O. Cardiac safety of ophthalmic timolol. Expert Opin Drug Saf. 2016;31:1-13.
8. Walia HS, Walia SS, Emanuel ME. Sick sinus syndrome associated with topical timolol maleate instillation. J Pharmacol Pharmacother. 2011;2(4):300-302.
9. Gerber SL, Cantor LB, Brater DC. Systemic drug interactions with topical glaucoma medications. Serv Ophthalmol. 1990;35:205-218.
10. Pratt NL, Ramsay EN, Kalisch Ellett LM, et al. Association between ophthalmic timolol and hospitalisation for bradycardia. J Ophthalmol. 2015;2015:567387. Epub March 22, 2015.
11. Roughead EE, Kalisch LM, Pratt NL, et al. Managing glaucoma in those with co-morbidity: not as easy as it seems. Ophthalmic Epidemiol. 2012;19(2):74-82.
12. Pringle SD, MacEwen CJ. Severe bradycardia due to interaction of timolol eye drops and verapamil. BMJ. 1987;294(6565):155-156.
13. Lewis SE. Controlled comparison of the cardiovascular effects of levobunolol 0.25% ophthalmic solution and betaxolol 0.25% ophthalmic suspension. J Glaucoma. 1994;3(4):308-314.
14. Lin L, Wang Y, Chen Y, Liu M. Bradyarrhythmias secondary to topical levobunolol hydrochloride solution. Clin Interv Aging. 2014;9:1741-1745.
15. Chun JG, Brodsky MA, Allen BJ. Syncope, bradycardia, and atrioventricular block associated with topical ophthalmic levobunolol. Am Heart J. 1994;127(3):689-690.
16. Anguita M, Torres F, Giménez D, et al. [Bradyarrhythmias secondary to the use of ophthalmic timolol. A report of 3 cases]. Rev Esp Cardiol. 1992;45(1):71-73.
17. Walia HS, Walia SS, Emanuel ME. Sick sinus syndrome associated with topical timolol maleate instillation. J Pharmacol Pharmacother. 2011;2(4):300-302.
18. Brubacher JR. Beta-adrenergic antagonists. In: Hoffman RS, Howland MA, Lewin NA, et al, eds. Goldfrank’s Toxicologic Emergencies. 10th ed. New York, NY: McGraw-Hill Education; 2015.
19. Sharma A. Beta-blocker toxicity. Medscape.com. Updated: May 2, 2016.
http://emedicine.medscape.com/article/813342-overview#showall. Accessed September 12, 2016.
20. Radhakrishnan R. Adrenergic antagonists. In: Whalen K, ed. Pharmacology. 6th ed. Philadelphia, PA: Wolters Kluwer; 2015:95-106.
21. Epocrates.com. Epocrates Plus Version 15.12.1. Updated June 23, 2016. Accessed September 20, 2016.
22. Semla TP, Beizer JL, Higbee MD. Geriatric Dosage Handbook. 20th ed. Hudson, OH: Lexicomp; 2015.
To comment on this article, contact rdavidson@uspharmacist.com.






