US Pharm. 2007;32(12):62-65.
Diabetes mellitus, the fifth
deadliest disease in the United States, is the result of
abnormalities in protein, fat, and carbohydrate metabolism that manifest as
hypergly cemia.1 Type I diabetes, which commonly occurs in
children and young adults, is characterized by beta-cell destruction resulting
in absolute insulin deficiency.2 Insulin resistance coupled with a
relative lack of insulin secretion characterizes Type II diabetes. In both
types, if the blood sugar is not adequately controlled, microvascular and
macrovascular complications can occur.2
Glycemic control has important
implications for clinical outcome in diabetes. The risk of diabetes-related
complications such as blindness, amputation, and organ failure is reduced by
35% for every 1-percentage-point decrease in hemoglobin A1c (HbA1c).3
Self-monitoring of blood glucose plays a vital role in assessment of glycemic
control by providing information about therapeutic efficacy and aiding in the
prevention of hypoglycemia.4 Current methods for evaluating blood
glucose levels include finger-stick measurements and HbA1c-detection tests.
The finger-stick measurement can be inconvenient and painful, and the reading
it supplies is accurate for the moment of testing only.5
Conversely, the HbA1c test provides an average glucose level over two to three
months, although results can be misleading: Hypoglycemic episodes can cause
the HbA1c to appear normal even though the patient's glucose control is
suboptimal. To overcome these limitations, continuous glucose monitoring (CGM)
devices have been developed that can track daily trends or fluctuations,
affording the opportunity for interventions. The information provided by CGM
systems can help patients with diabetes achieve glycemic control, thus
reducing the risk of associated complications.6,7
Continuous Glucose
Monitoring
Developed by
Medtronic, Inc., the Guardian REAL-Time CGM System was approved by the FDA in
July 2006 (Figure 1).6 This physician-prescribed device
allows patients to track fluctuations in their glucose levels, as well as the
rate at which these changes occur. Patients can see how medications, exercise,
diet, and lifestyle affect their glycemic control.5,7 Although
these observations can serve as a guide for interventions, adjustments to
therapy should not be made based solely on the information supplied by the
monitor. The levels provided by the monitor indicate when the patient may need
to do a confirmatory finger-stick test. Interventions should be implemented
only after confirmatory measurements are obtained with a standard glucose
meter.

SOF-Sensor: Easy
and virtually painless insertion of the sensor is achieved using the
autoinsertion device, called the Sen-serter, provided with each system; manual
insertion is optional, but not recommended.8-10 The patient inserts
the sensor in an area with adequate subcutaneous tissue and replaces it with a
new sensor every 72 hours. The new insertion site should be at least 2 inches
from the previous site.8,9 Potential risks associated with sensor
insertion include bleeding, swelling, irritation, and/or infection.8
The sensor measures glucose levels in the interstitial fluid through the use
of two biological reagents (less than 0.4 mcg of glucose oxidase and less than
0.7 mcg of bovine serum albumin). Due to the small amount of each reagent, the
risk of viral transmission and tissue reactions is minimal. After sensor
insertion, five minutes should elapse before the MiniLink REAL-Time
Transmitter is snapped into the sensor.
MiniLink Transmitter:
The transmitter powers the sensor and collects data. Almost a third the size
of earlier Medtronic transmitters, it can be easily worn under clothing.
7,8,11 A tester included with the system determines whether the
transmitter is working properly.9 The transmitter has a
rechargeable battery, so a MiniLink Charger is included. Recharging the
battery for 20 minutes provides three days of continued use, and a full
recharge provides 14 days; however, it is recommended that the battery be
recharged after each use.7,10,11
The MiniLink Transmitter is
designed to withstand common electromagnetic interference such as microwaves
and airport security systems. However, some electronic devices that transmit
in a similar frequency, such as 900-MHz cordless phones and most cellular
phones, may prevent the monitor from receiving glucose data.9,11
This will not harm the Guardian System, nor will incorrect data be sent to the
monitor. Interference problems can be resolved by ensuring that the
transmitter and the receiving device are less than 6 feet apart and that all
radiofrequency-transmitting devices are turned off.
Discreet and easy to wear, the
monitor can be concealed in a pocket or under clothing.12 A
watertight seal is formed when the transmitter and the sensor are connected,
allowing them to be worn while bathing, showering, and even swimming to a
depth of up to 8 feet for up to 30 minutes. The Guardian monitor, blue tester,
and MiniLink Charger are not water-resistant and should not be immersed in
water.9-11
Every five minutes, the
MiniLink Transmitter wirelessly sends glucose readings to the monitor,
producing 288 REAL-Time readings per day.7,9 It is recommended that
an Energizer alkaline AAA battery be used to power the monitor.
Calibration and
Capturing Events: Calibration of the system requires a meter blood
glucose measurement two hours after insertion of a new sensor; subsequent
calibrations on day 1 should occur within six and 12 hours, respectively,
after initial calibration. Calibrations must be performed at least every 12
hours on days 2 and 3, but more than four calibrations per day should be
avoided.9,13 The best times to calibrate are when blood glucose
levels are not changing rapidly: before meals, at least two hours after a
meal, and before bedtime. Daily events like noncalibration blood glucose
measurements, carbohydrate consumption, exercise, and insulin injections can
be entered into the Guardian REAL-Time System along with information regarding
when patients take medications and when they feel ill or stressed.
CareLink Personal
Therapy Management Software: The Guardian System features CareLink
Personal Therapy Management software, a secure Web-based tool capable of
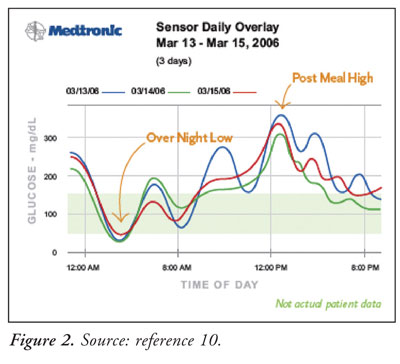
integrating logbook, meter, and CGM data. Charts, graphs, and tables are
generated that illustrate patterns and problems with glucose control (
Figure 2).7,10 The ComLink connector, which is installed on the
patient's computer, allows the patient to upload data to the CareLink system.
9 Detailed reports created by this software can be shared with
physicians, and this information can be incorporated into the patient's
electronic medical record. This feature is not available in all locations.
7,9
Features:
Features that are unique to the Guardian REAL-Time CGM System include
predictive and rate-of-change alerts and comprehensive, expanded trend graphs (
TABLE 1).7 The comprehensive trend graphs generated by the
Guardian reveal glucose patterns in 3-, 6-, 12-, and 24-hour increments,
allowing patients to ascertain how daily activities affect their glycemic
control. Predictive alerts forewarn the patient if his or her blood glucose is
approaching a critical level: Alarms can be set according to patient
preference at five-minute intervals, beginning at five minutes and ending at
30 minutes.9

Efficacy
To date, there have
been no clinical trials of the Guardian REAL-Time CGM System. However, there
have been studies involving the REAL-Time System's predecessor, the Guardian
RT System. Since the REAL-Time System uses the same algorithm as the RT System
to determine glucose levels, discussion of these studies is appropriate.9
Sensor Accuracy:
In a single-site, in-clinic study, 16 patients with Type 1 diabetes whose age
ranged from 18 to 65 years and who had a mean HbA1c of 8.2% were evaluated
while wearing two Guardian RT systems simultaneously. One system was
calibrated approximately 3.5 times per day; the other system was calibrated an
average of five times per day. Values from the Guardian RT System were
compared with plasma glucose values obtained from a reference method, the YSI
2300 STAT Plus glucose analyzer.9 Guardian RT readings were taken
every five minutes; YSI measurements were obtained every 30 minutes. Each YSI
value was paired with the Guardian RT value that was closest in time to it.
9 A total of 3,941 paired glucose measurements were obtained. Guardian
RT readings, were found, on average, to be 15 mg/dL lower than YSI readings.
To evaluate the clinical implications of the difference between the YSI values
and Guardian RT values, the Clarke Error Grid, which consists of a correlation
plot divided into five zones, was used. Clinically acceptable values fall
within Zones A and B of the Error Grid, while readings that are in Zones C, D,
and E represent clinically significant errors. Of the 3,941 paired
measurements, 96% of the values fell within Zone A (61.7%) and Zone B (34.4%).
Agreement between YSI and Guardian RT values tended to be closer when glucose
values were at mid-range, versus high or low glucose levels.
The precision of the Guardian
RT values was estimated using 11,475 paired readings from the two Guardian RT
systems. On average, there was a 17.2% difference between the readings
generated by the two systems.
The effects of calibration
frequency were evaluated by looking at the specificity and sensitivity of
alarm performance. This analysis revealed that, when units were calibrated
less often, specificity in the hyperglycemic range decreased from 0% to 2% and
increased 2% to 4% in the hypogly cemic range. Less- frequent calibrations
caused sensitivity to increase by 5% to 9% in the hypergly cemic range,
decrease by 3% to 7% when alarms were set to between 85-100 mg/dL, and
decrease by 7% to 16% when the alarms were set to 80 mg/dL or lower.
GuardControl Study:
This randomized, controlled trial involved 81 pediatric patients and 81 adult
patients with stable Type 1 diabetes who had an A1c level equal to or greater
than 8.1% despite intensive insulin therapy with multiple daily insulin
injections or continuous subcutaneous insulin infusions.14 Patients
were randomized into one of three groups for three months: arm 1, which used
the Guardian RT continuously; arm 2, which used the Guardian RT for three days
every two weeks; and placebo, which used conventional self-monitoring of blood
glucose (SMBG). Any treatment adjustments made by physicians or by patients in
the placebo group were based on SMBG values; treatment adjustments made in
arms 1 and 2 were based on real-time glucose profiles. Prior to taking
corrective action if hypo- or hyperglycemic alarms or symptoms occurred,
patients were instructed to perform confirmatory SMBG measurements. After one
and three months, A1c was centrally measured. Calibration of the device was
achieved by using SMBG reference values. The high glucose alert level was set
at 170-250 mg/dL; after the first 10 days, the upper alarm was lowered to 200
mg/dL. The low glucose alert level was set at 50-80 mg/dL.
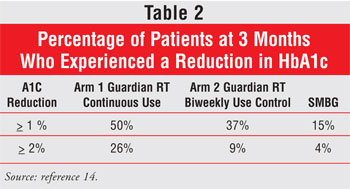
Significant reductions in A1c
were observed with the use of the Guardian RT System (TABLE 2). At one
and three months, a mean reduction in SMBG per day was seen in arm 1, but this
was not statistically significant compared with arm 2 and placebo. The average
daily insulin dose at 3 months was not significantly different from baseline
in the three groups. Severe hypoglycemia occurred once in arm 1 and once in
arm 2. The patient in arm 1 experienced hypoglycemia despite a confirmatory
low SBMG and corrective carbohydrate consumption; the other patient was not
wearing the device when the episode occurred. In arms 1 and 2, 82% of patients
at one month and 95% at three months reported using real-time CGM information
to make dietary, lifestyle, or insulin adjustments. Exact changes to patients'
diabetes management were not recorded on a daily basis, so the link between
improvement of glycemic control and the use of real-time CGM cannot be
described in detail.
Pediatric Guardian
Real-Time CGM System
Medtronic has
specifically targeted the pediatric diabetic population with its recently
FDA-approved pediatric model of the Guardian REAL-Time System.3,15
The pediatric model is indicated for patients 7 to 17 years of age and has a
special feature: a low alert threshold that cannot be set below 90 mg/dL,
allowing additional time for patients to react to hypoglycemic episodes.
3,10 The high alert threshold is set at a default of 280 mg/dL in this
model.
Conclusion
The Guardian
REAL-Time CGM System may benefit patients with Type 1, Type 2, or gestational
diabetes; the pediatric model may help children and teenagers identify and
reduce the duration of hypoglycemic episodes.3,12 Even though
finger-sticks are still necessary, the Guardian REAL-Time System provides
significantly more information than finger-stick measurements alone.12
Special features allow patients to track daily trends that impact glycemic
control; this information can be utilized by both patients and physicians to
improve treatment strategies.7 The GuardControl Study suggested
that the Guardian system can help reduce HbA1c, thereby producing a
significant impact on overall glucose control and diabetes-related
complications.
For more information,
visit Medtronic's Web site at www.minimed.com or call 1-800-MINIMED.
References
1. American Diabetes Association. The dangerous toll of diabetes. Available at: www.diabetes.org/diabetes-statistics/dangerous-toll.jsp. Accessed June 13, 2007.
2. Triplitt C, Reasner C, Isley W. Diabetes mellitus. In: DiPiro J, Talbert R, Yee G, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 6th ed. New York: McGraw-Hill; 2005:1333-1367.
3. Medtronic Inc. FDA approves new Medtronic continuous glucose monitoring devices for children and teenagers. News release. March 12, 2007. Available at: wwwp.medtronic.com/Newsroom/NewsRe leaseDetails.do?itemId=1173705787522&lang=en_US. Accessed June 17, 2007.
4. American Diabetes Association. Standards of medical care for patients with diabetes mellitus. Diabetes Care. 2002;25:213-229.
5. Medtronic Inc. The value of real-time continuous glucose readings. Available at: wwwp.medtronic.com/Newsroom/LinkedItemDetails.do?itemId=1168599428346&itemType=backgrounder&lang=en_IN. Accessed June 17, 2007.
6. Medtronic Inc. Medtronic receives FDA approval for Guardian® Real-Time Continuous Glucose Monitoring System. News release. June 17, 2006. Available at: wwwp.medtronic.com/Newsroom/NewsRe leaseDetails.do?itemId=1152884580852&lang=en_US. Accessed June 17, 2007.
7. Medtronic launches new Guardian® Real-Time System with miniature Transmitter and CareLinkTM Personal Software. News release. March 19, 2007. Available at: wwwp.medtronic.com/Newsroom/NewsReleaseDetails.do?itemId=1174306815850&lang=en_US. Accessed June 17, 2007.
8. Glucose Sensor User Guide. Available at: www.minimed.com/pdf/glucose_sensor_user_guide.pdf. Accessed June 17, 2007.
9. Guardian REAL-Time System User Guide. Available at: www.minimed.com/pdf/guardian_real_time_user_guide.pdf. Accessed June 17, 2007.
10. Medtronic Inc. Introducing theGuardian REAL-Time Continuous Glucose Monitoring System. Available at: www.minimed.com/products/guardian/. Accessed June 13, 2007.
11. MiniLink REAL-Time Transmitter User Guide. Available at: www.minimed.com/pdf/minilink_user_guide.pdf. Accessed June 17, 2007.
12. Medtronic Inc. Guardian REAL-Time Continuous Glucose Monitoring System. Patient brochure.
13. Guardian REAL-Time Getting Started Guide. Available at: www.minimed.com/ pdf/guardian_real_time_getting_started.pdf. Accessed June 17, 2007.
14. Deiss D, Bolinder J, Riveline JP, et al. Improved glycemic control in poorly controlled patients with Type 1 diabetes using Real-Time Continuous Glucose Monitoring. Diabetes Care. 2004;29:2730-2732.
15. American Diabetes Association.
Total prevalence of diabetes &prediabetes. Available at:
www.diabetes.org/diabetes-statistics/prevalance.jsp. Accessed June 13, 2007.
To comment on this article, contact
editor@uspharmacist.com.





