US Pharm. 2013;38(9):23-26.
ABSTRACT: Primary dysmenorrhea is the most common menstrual complaint, and it is frequently self-treated by patients of all ages. Teenagers are especially prone to experiencing dysmenorrhea symptoms and selecting OTC therapies without adult supervision. Nonsteroidal anti-inflammatory drugs and combined oral contraceptives represent first-line therapy for dysmenorrhea pain, but several dietary and herbal supplements also show promise. When possible, pharmacists should educate patients regarding proper drug selection and dosing in order to optimize patient outcomes.
Dysmenorrhea is the most common menstrual complaint in the United States, impacting more than 50% of women over the course of their lifetime.1 Primary dysmenorrhea is specifically defined as recurrent, crampy abdominal pain that occurs in the absence of abnormal pelvic pathology.2,3 Incidence of primary dysmenorrhea is highest during the teenage years, with 60% to 90% of this population reporting symptoms.4-6 It is estimated that 14% of teenagers with symptoms miss school because of pain, and the percentage increases to 50% if pain is severe.7 Other risk factors for primary dysmenorrhea include heavy or prolonged menstrual flow, family history of dysmenorrhea, smoking, nulliparity, and lower socioeconomic status.2,3
Despite reports that upward of 90% of teenagers self-treat primary dysmenorrhea, studies of United States and Canadian adolescents revealed that only 14% to 16% sought advice from a physician prior to self-treatment.5,7-9 In fact, 78% reported no adult supervision in choosing medication, instead relying on what was conveniently available in the medicine cabinet at home or on advice from friends.9 Furthermore, 53% were unsure of correct dosing for the OTC medications used to treat primary dysmenorrhea.6,8 At least 70% took their medications incorrectly with regard to dosage and/or dosing frequency, with the most frequent errors being ingestion of single doses that were larger than recommended or dosing intervals that were less frequent than recommended.6,8 Polypharmacy was also common, with 31% reporting use of two concurrent OTC medications and 15% reporting use of three or more medications.8
Because of the high incidence of primary dysmenorrhea in the adolescent population and the tendency of teenagers to self-treat the condition without adult supervision, pharmacists are uniquely positioned to offer care and advice to these patients at the time OTC medications are purchased.
DIAGNOSIS AND PATHOPHYSIOLOGY
Dysmenorrhea typically presents as crampy pain along the midline of the lower abdomen.3,4 Pain may be accompanied by complaints of diarrhea, nausea, vomiting, fatigue, dizziness, headache, and backache.3,4 Primary dysmenorrhea symptoms first present just after a stable menstrual cycle has been established; accordingly, any patient experiencing pain prior to or within the first 6 months of menarche should be referred for further evaluation. The same is true for patients presenting with first-time pain after several years of asymptomatic cycles.3
The patient with primary dysmenorrhea generally experiences reproducible symptoms from cycle to cycle.3 Symptoms typically start several hours before the menstrual flow begins.3,4 Pain severity peaks at the time of maximal menstrual flow (usually within first 2 days of menses) and quickly dissipates thereafter.3 Therefore, total pain duration usually ranges from 12 to 72 hours.3,4 Assuming that the patient presents with this typical pattern of symptoms, practice guidelines indicate that formal medical evaluation and pelvic examination are not needed for diagnosis.3 However, patients should be referred for assessment should symptoms occur outside this normal pattern or when symptoms do not respond to first-line OTC therapies.3 In such cases, the physician should conduct a history and physical examination in order to determine potential secondary causes of dysmenorrhea, the most common of which is endometriosis.3
The pain of primary dysmenorrhea is caused by excessive prostaglandin (PG) production within secretory endometrial cells.2,4,10 This PG release, in turn, causes uterine contractions, uterine muscle ischemia, and increased peripheral nerve sensitivity.2,4,10
TREATMENT
The therapeutic objective for treating primary dysmenorrhea is reduction of pain so that the patient can participate in usual activities of daily living without limitation. Common nonpharmacologic strategies include hot baths, heating pads, and exercise.8 Unfortunately, data supporting these methods are lacking, and drug therapy is often necessary.11-13 Drug treatments generally target inhibition of PG synthesis or reduction in the number of endometrial cells present at the time of menstruation.
First-Line Therapies
First-line therapies for primary dysmenorrhea generally involve nonsteroidal anti-inflammatory drugs (NSAIDs) or hormonal contraception. No studies have directly compared the two methods; thus, no conclusions may be reached regarding preferential selection between these two options based on efficacy alone.
NSAIDs: Simple analgesics (specifically NSAIDs) reduce primary dysmenorrhea pain via inhibition of PG synthesis and have been the mainstay of therapy for decades. Simple analgesics are most commonly used as self-treatment by adolescents, with acetaminophen, aspirin, ibuprofen, and naproxen being reported most often.6,8,9 These drugs may be coadministered with antihistamines or diuretics (e.g., pyrilamine or pamabrom) because of coformulation within many available OTC products; however, there are no studies demonstrating that these added ingredients improve patient response. Unfortunately, some of these medications are not effective for dysmenorrhea (see discussion below) or are contraindicated for use in children and adolescents (aspirin).
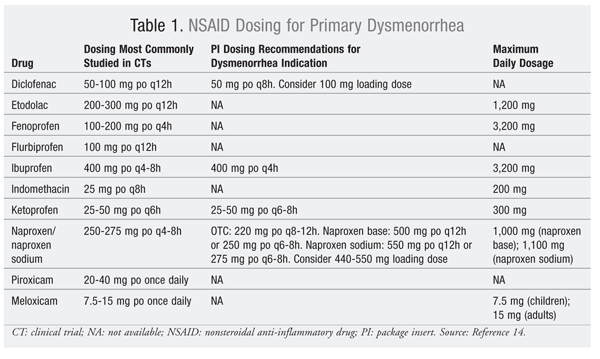
A systematic review of 73 randomized, controlled trials concluded that NSAIDs were superior to placebo for treatment of primary dysmenorrhea pain and reduced the number of days absent from school and work.14 It should be noted that these effectiveness data did not apply to aspirin or acetaminophen.14 Numerous NSAIDs have been studied for this indication, but no literature exists to indicate the effectiveness of any one agent over another.14 Consequently, NSAID choice is best guided by cost and availability. Most patients will be candidates for NSAID therapy, unless use is precluded by allergy or pre-existing gastrointestinal (GI) or renal issues.
Patients should generally be advised to begin NSAID therapy just prior to the start of menses and to continue dosing on a scheduled basis for 2 to 3 days following the onset of each cycle.2 Despite these recommendations, the average time to first dose is 30 minutes after the onset of pain for most teenagers, with only 16% taking the medications prophylactically.6 Therefore, education regarding proper dosing may help improve patient outcomes. Average study dosages for drugs available in the United States appear in TABLE 1. Although typical NSAID side effects such as GI upset and drowsiness were reported more often than placebo in controlled trials, incidence did not reach statistical significance, which was likely due in part to the short-term duration of therapy for the primary dysmenorrhea indication.14
Hormonal Contraception: Although NSAIDs provide pain relief for a large percentage of patients with primary dysmenorrhea, 20% to 25% will fail this therapy.14 When NSAIDs fail or are contraindicated, or when contraception is desired, hormonal contraception is a viable and effective method for treating primary dysmenorrhea. Hormonal contraceptives treat primary dysmenorrhea by causing regression of the endometrium, shortening the time of endometrial proliferation, and limiting the secretory activity of endometrial glands. The most commonly studied hormonal contraceptive method for this indication is the combined oral contraceptive (COC) pill, but other methods, including the contraceptive patch, contraceptive ring, medroxyprogesterone injection, and levonorgestrel-releasing intrauterine system, are also endorsed by practice guidelines because of their similar mechanisms of action.3
A systematic review of 10 randomized, controlled trials concluded that low- to medium-dose (i.e., 20-35 mcg ethinyl estradiol) COCs are superior to placebo for treatment of primary dysmenorrhea pain and reduce the number of days absent from school and work.15 One study in this review also suggested that third-generation pills (containing desogestrel, etonogestrel, and norgestimate) may be more effective than second-generation pills (containing levonorgestrel and norethindrone) in providing pain relief, but this finding was not observed in the other studies comparing third-generation pills with first- and second-generation pills.15 Not surprisingly, COCs were more likely than placebo to cause nausea, headache, and weight gain, but withdrawal from therapy did not differ between groups.15 It must be remembered that rare side effects of hormonal contraception include vascular events such as venous thromboembolism, myocardial infarction, and stroke; thus, patients must be screened for additive risk factors before beginning therapy. For information about conditions that preclude use of hormonal contraception, see the CDC’s report, “U.S. Medical Eligibility Criteria for Contraceptive Use, 2010” (www.cdc.gov/mmwr/pdf/rr/rr59e0528.pdf).
Traditional dosing strategies for COCs utilize placebo pills to stimulate a chemical withdrawal bleed. This withdrawal bleed may still produce PGs and pain, so continuous-dosing contraceptive strategies have been studied as a way to improve response rates. In a small study of 38 patients, a low-dose monophasic COC was randomly dosed in a traditional 21-day hormone/7-day placebo pattern (usual dosing) or a 28-day hormone pattern (continuous dosing).16 Reported pain relief was statistically greater in the continuous-dosing versus usual-dosing group 1 month after starting therapy, but both dosing strategies showed equivalent reductions in pain after 6 months of therapy.16 This finding may be explained by the fact that several COC cycles are required to achieve maximal endometrial regression. Thus, continuous dosing may perhaps be best recommended at the start of therapy, with long-term continuation if the patient prefers not to experience withdrawal bleeding. Patients selecting continuous-dosing methods may be more likely to experience breakthrough spotting than those using more traditional dosing methods, but the continuous-dosing method is otherwise safe for long-term use.15
Upon initiation of hormonal contraception, patients should be cautioned that breakthrough bleeding and spotting might be common during the first two to three cycles of use. They should also be advised that maximal dysmenorrhea pain relief might not be experienced for a few months, although some relief likely will be noted in the first or second cycle. As with all hormonal contraception, patients should be cautioned to immediately discontinue use and contact a physician should any signs or symptoms of vascular events occur. It may be helpful to teach patients the mnemonic ACHES: abdominal pain, chest pain, headache, eye/vision changes, and severe leg pain.
Alternative and Emerging Therapies
Many patients may prefer to use more “natural” methods for treating dysmenorrhea pain, and the literature supports use of several dietary, herbal, and alternative medicine methods.
Although a wide variety of herbal and dietary supplements have been purported to help improve dysmenorrhea pain (including vitamin B1, vitamin B6, vitamin E, magnesium, and fish oil), systematic reviews have concluded that the only supplements with any reasonable evidence supporting effectiveness are vitamin B1 (B1 deficiency is thought to promote muscle cramping), magnesium (supplementation is thought to reduce PG levels), and Chinese herbal remedies.17,18 Unfortunately, evidence is less clear regarding optimal dosing and preparation for most of these products. Vitamin B1 (thiamine) dosed at 100 mg orally once daily was used in one large, well-controlled study; thus, this dosage is the most reasonable to recommend to most patients.17 The Chinese herbal preparations that have showed efficacy have generally been multi-ingredient products that are intended to “regulate Qi and blood,” “reinforce Chi and blood,” “warm the interior,” or “tonify the kidney and liver.”18 Interestingly, some data suggest that such herbal preparations may even be superior to NSAIDs and COCs for relieving dysmenorrhea pain, but larger studies are needed to confirm these trends.18 Despite the overall paucity of large, well-designed studies of vitamin B1, magnesium, and Chinese herbal preparations for dysmenorrhea, the risk of side effects from each product is small, and so it may be reasonable to allow patients to try such remedies, if desired.
More recently, vitamin D supplementation has been studied as a dysmenorrhea treatment based on the observation that higher vitamin D levels lead to reductions in PG synthesis.19 In a small, randomized, placebo-controlled study of patients with baseline 25(OH)D (25-hydroxyvitamin D) levels less than 45 ng/mL, a single dose of 300,000 IU of vitamin D administered 5 days prior to menses significantly reduced primary dysmenorrhea pain and reduced the percentage of patients requiring NSAID rescue therapy.19 Although vitamin D may be a promising therapy for dysmenorrhea, the unknown impact of such supplementation in patients with more normal baseline 25(OH)D levels and the lack of knowledge regarding duration of effect after a single dose (if 300,000 IU were given monthly, this would far exceed the safe limits set by the Institute of Medicine) suggest that vitamin D should be further studied for this indication.20
Various acupuncture methods have also been studied as a remedy for primary dysmenorrhea. Acupuncture is known to increase levels of endorphins, serotonin, and acetylcholine in the central nervous system. Systematic reviews have suggested that various acupuncture methods may be more effective than placebo, herbal remedies, and NSAIDs for treating dysmenorrhea pain.21,22 Thus, patients interested in nonpharmacologic methods of treatment may want to explore this possible treatment.
CONCLUSION
Primary dysmenorrhea is a common menstrual complaint that is frequently self-treated by patients of all ages. Teenagers are especially likely to experience dysmenorrhea symptoms and to select OTC therapies without adult supervision. Pharmacists should provide education to patients regarding proper drug selection and dosing, when possible, in order to optimize patient outcomes.
REFERENCES
1. American College of Obstetricians and Gynecologists. Dysmenorrhea:
frequently asked questions.
www.acog.org/~/media/For%20Patients/faq046.pdf?dmc=1&ts=20130714T1255224066.
Accessed July 10, 2013.
2. Umland EM, Weinstein LC, Buchanan EM. Menstruation-related disorders. In: DiPiro J, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. Based on: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 8th
ed. New York, NY: McGraw-Hill; 2011.
www.accesspharmacy.com.proxyhs.researchport.umd.edu/content.aspx?aid=7993589.
Accessed July 10, 2013.
3. Lefebvre G, Pinsonneault O, Antao V, et al. Primary dysmenorrhea consensus guideline. J Obstet Gynaecol Can. 2005;27:1117-1146.
4. Hall JE. Menstrual disorders and pelvic pain. In: Longo DL, Fauci AS, Kasper DL, et al, eds. Harrison’s Online. Based on: Longo DL, Fauci AS, Kasper DL, et al, eds. Harrison’s Principles of Internal Medicine. 18th
ed. New York, NY: McGraw-Hill; 2012.
www.accesspharmacy.com.proxy-hs.researchport.umd.edu/content.aspx?aID=9093327.
Accessed July 10, 2013.
5. Johnson J. Level of knowledge among adolescent girls regarding effective treatment for dysmenorrhea. J Adolesc Health Care. 1988;9:398-402.
6. Campbell MA, McGrath PJ. Use of medication by adolescents for the management of menstrual discomfort. Arch Pediatr Adolesc Med. 1997;151:905-913.
7. Klein JR, Litt IF. Epidemiology of adolescent dysmenorrhea. Pediatrics. 1981;68:661-664.
8. O’Connell K, Davis AR, Westhoff C. Self-treatment patterns among adolescent girls with dysmenorrhea. J Pediatr Adolesc Gynecol. 2006;19:285-289.
9. Chambers CT, Reid GJ, McGrath PJ, Finley GA. Self-administration of over-the-counter medication for pain among adolescents. Arch Pediatr Adolesc Med. 1997;151:449-455.
10. Purcell KJ, Taylor RN. Disorders of the female reproductive tract. In: McPhee SJ, Hammer GD. Pathophysiology of Disease. Based on: McPhee SJ, Hammer GD, eds. Pathophysiology of Disease: An Introduction to Clinical Medicine. 6th
ed. New York, NY: McGraw-Hill; 2010.
www.accesspharmacy.com.proxy-hs.researchport.umd.edu/content.aspx?aid=5372081.
Accessed July 10, 2013.
11. Blakey H, Chisholm C, Dear F, et al. Is exercise associated with primary dysmenorrhoea in young women? BJOG. 2010;117:222-224.
12. Brown J, Brown S. Exercise for dysmenorrhoea. Cochrane Database Syst Rev. 2010;(2):CD004142.
13. Daley AJ. Exercise and primary dysmenorrhoea: a comprehensive and critical review of the literature. Sports Med. 2008;38:659-670.
14. Marjoribanks J, Proctor M, Farquhar C, Derks RS. Nonsteroidal anti-inflammatory drugs for dysmenorrhoea. Cochrane Database Syst Rev. 2010;(1):CD001751.
15. Wong CL, Farquhar C, Roberts H, Proctor M. Oral contraceptive pill for primary dysmenorrhoea. Cochrane Database Syst Rev. 2009;(4):CD002120.
16. Dmitrovic R, Kunselman AR, Legro RS. Continuous compared with
cyclic oral contraceptives for the treatment of primary dysmenorrhea: a
randomized controlled trial. Obstet Gynecol. 2012;119:1143-1150.
17. Proctor ML, Murphy PA. Herbal and dietary therapies for primary and secondary dysmenorrhoea. Cochrane Database Syst Rev. 2001;(3):CD002124.
18. Zhu X, Proctor M, Bensoussan A, et al. Chinese herbal medicine for primary dysmenorrhoea. Cochrane Database Syst Rev. 2008;(2):CD005288.
19. Lasco A, Catalano A, Benvenga S. Improvement of primary
dysmenorrhea caused by a single oral dose of vitamin D: results of a
randomized, double-blind, placebo-controlled study. Arch Intern Med. 2012;172:366-367.
20. Bertone-Johnson ER, Manson JE. Vitamin D for menstrual and
pain-related disorders in women: comment on “improvement of primary
dysmenorrhea caused by a single oral dose of vitamin D.” Arch Intern Med. 2012;172:367-369.
21. Chung YC, Chen HH, Yeh ML. Acupoint stimulation intervention for
people with primary dysmenorrhea: systematic review and meta-analysis of
randomized trials. Complement Ther Med. 2012;20:353-363.
22. Cho SH, Hwang EW. Acupuncture for primary dysmenorrhoea: a systematic review. BJOG. 2010;117:509-521.
To comment on this article, contact rdavidson@uspharmacist.com.





