US Pharm. 2013;38(8):HS4-HS8.
ABSTRACT: Calculi in the urinary tract vary in composition, size, and location. Most kidney stones are composed of calcium oxalate or calcium phosphate, and they may lead to urinary obstruction, flank pain, hematuria, and an increased infection predisposition. Acute kidney stone management requires a thorough patient history, definitive diagnosis, supportive care, and possible surgical intervention. Long-term management focuses on addressing the underlying etiologies and preventing complications of treatment. Pharmacists should continue to identify drug-induced causes, promote public health information, and counsel patients on methods to avoid recurrent stone formation.
Urolithiasis, or stone formation in the urinary tract, includes ureterolithiasis (ureteral calculi) and nephrolithiasis (renal/kidney calculi).1,2 Stones form when substances in the urine become highly concentrated and/or insoluble.1 They vary in size and location; while small stones usually pass with minimal pain sensation, larger stones may remain in the ureter or bladder, obstruct urine outflow, and cause severe pain (secondary to muscular dilation and spasms) and/or bleeding.1
Epidemiology
Based on survey data from 2001 to 2008 in the United States, the lifetime prevalence of at least one kidney stone is up to 12% in males and 7% in females.3 Data from 2002 to 2007 demonstrated that there were more than 300,000 emergency room visits per year for upper urinary tract stones.3 The majority of these cases were in Caucasian males between 45 and 54 years of age who lived in the Southern region of the U.S.—a locale colloquially named the “kidney stone belt.”3,4
Pathophysiology/Etiology
There are four predominant types of renal calculi: calcium (80%), uric acid (9%), struvite (<10%), and cystine (<1%) stones.4 These may be found in the kidney (staghorn [pelvic and ≥2 calyces] or non-staghorn [calyceal or pelvic]), ureter (proximal, middle, or distal), and urinary bladder.4 Kidney stones may develop due to an overgrowth on sites of Randall’s plaque (e.g., calcium oxalate), growth on plugs from dilated ducts of Bellini (e.g., apatite), or in free solution (e.g., cystinuria).4
Inadequate fluid intake and/or urine output promotes urine concentration, and both have been identified as the most important factors promoting kidney stone production.1,2 Calcium stones may form secondary to idiopathic hypercalciuria, resorptive hypercalciuria, hyperoxaluria, hypocitraturia, and/or hyperuricosuria.5 Hyperoxaluria can occur with a high dietary intake of oxalate-containing foods (e.g., spinach, rhubarb, nuts, wheat bran).1,5 High intake of purine, a breakdown product of animal protein (e.g., eggs, fish), may lead to hypocitraturia or hyperuricosuria.1,5 Calcium intake from food is not implicated in calcium stone formation; it binds intestinal oxalate and decreases calcium oxalate stone development.1,5
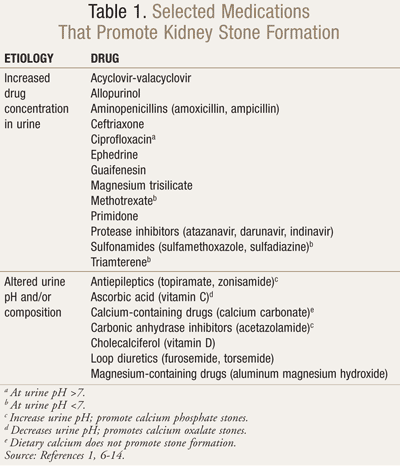
Urine pH <5.5 promotes uric acid stone formation regardless of seemingly normal 24-hour uric acid levels.5 An autosomal recessive genetic disorder leads to cystine stones, typically at a urine concentration ≥300 mg/L.5 Struvite (magnesium ammonium phosphate) stones could develop in patients with urine pH >7 and chronic genitourinary tract infections by urease-producing bacteria (e.g., Proteus, Pseudomonas, and Klebsiella species).5 Calcium phosphate stones may also form as a result of urine alkalinization (pH >6.5) for calcium (i.e., hypocitraturia), uric acid, and cystine stones.4,5 In addition, several drugs could crystallize in the kidney to form stones, due to supersaturation and/or insolubility at a certain urine pH (TABLE 1).1,6-14
Diagnosis
Staghorn, or small nonobstructing, stones may only produce mild-to-moderate symptoms (e.g., malaise).15 Otherwise, patients with acute kidney stones most likely seek medical attention after experiencing severe flank pain (radiating inferiorly and anteriorly), nausea and vomiting, and/or hematuria.15,16
Confirmatory tests are typically performed to differentiate nephrolithiasis from infections, malignancies, and/or musculoskeletal inflammation or spasms.17 Following a thorough patient history and physical examination, diagnostic imaging is required for confirming renal calculi.1,16 In pregnant patients, an ultrasound examination is indicated to avert radiation exposure.1,16 If patients previously had radiopaque calculi, a plain-film radiography could be used.16 Otherwise, noncontrast computerized tomography (NCCT) is most preferred because of its high sensitivity and specificity.1 NCCT also avoids the risk of contrast-induced kidney damage, particularly with serum creatinine levels >2 mg/dL.15 In all other patients, if NCCT is unavailable, intravenous urography (IVU) could be used to detect stones.1,15,16
A biochemical workup should be performed in addition to imaging.1,15 Basic analyses include a urinary sediment or dipstick test out of a spot urine sample (for red and white blood cells, nitrite, and urine pH); urine culture or microscopy; serum blood sample (for creatinine, uric acid, ionized calcium, parathyroid hormone, sodium, potassium, and phosphorous); CBC count with differential (for C-reactive protein); and coagulation test (for international normalized ratio [INR] and partial thromboplastin time [PTT]).1,15 After collecting and straining urine, stone analysis should be performed in first-time calculi formers using validated procedures (e.g., x-ray diffraction or infrared spectroscopy) and be repeated in most recurrent stone formers.1,15
Management
Acute kidney stone management involves a combination of supportive care and pharmacotherapy.1,15 If patients present to the emergency department with signs and symptoms consistent with infection (e.g., fever, chills, pyuria), empiric antibiotics should cover gram-positive and -negative organisms, while considering local antibiogram data.17 Specialty consultations are indicated for patients at age extremes or with debilitation, stones >5 mm in diameter, multiple stones, hydronephrosis, pregnancy, refractory pain or nausea, or clinical emergencies (e.g., urosepsis, anuria, renal failure).16,17
Regardless of the stone type diagnosed, patients should receive oral or IV hydration with at least 2 L of fluid.15,17 Normal saline (NS; sodium chloride 0.9%) could promote calcium-based stone formation; therefore, dextrose 5% in water (D5W) with ½ NS may be preferable.17
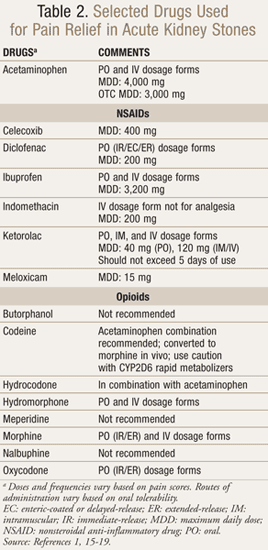
In patients with small stones and mild pain, acetaminophen is appropriate.15 For effective alleviation of moderate-to-severe pain, nonsteroidal anti-inflammatory drugs (NSAIDs) or opioid medications should be provided immediately (TABLE 2).1 Each drug class has advantages and disadvantages, and pain management should consider the patient’s renal function, ability to tolerate side effects, and dosing intervals.1,17
Most calculi <10 mm in diameter exit the kidney and travel through the ureter.1,15,16 While spontaneous passage in urine depends on location and size, ureteral stones <5 mm should pass within 6 weeks of diagnosis.1,16 Stones between 5 and 7 mm have about a 50% chance of being passed, while stones >7 mm might require urologic interventions.15 In select patients, 2- to 4-week trials of conservative management (i.e., watchful waiting/observation) may be attempted, and medical expulsive therapy (MET) could be utilized in individuals comfortable with hastening ureteral stone (<10 mm) passage by several days (TABLE 3).1,15-28 These patients must also have well-controlled pain, preserved renal function, and no systemic infection.
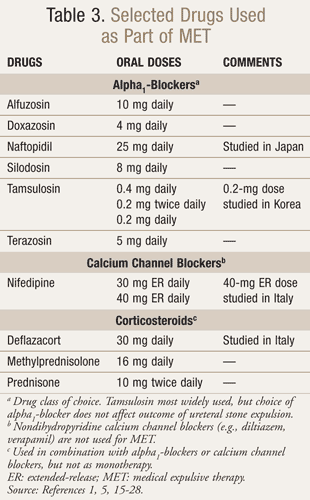
MET includes alpha1-blockers and dihydropyridine calcium channel blockers (e.g., nifedipine), which possess antispasmodic activity and relax ureteral smooth muscles.1,15,17 Corticosteroids may be used in addition to these drugs, but they are not effective as monotherapy.1,15,17 Phosphodiesterase type 5 (PDE5) inhibitors (e.g., vardenafil, sildenafil, tadalafil) demonstrate in vitro activity on ureteral smooth muscles, but their utility as MET is unknown.20
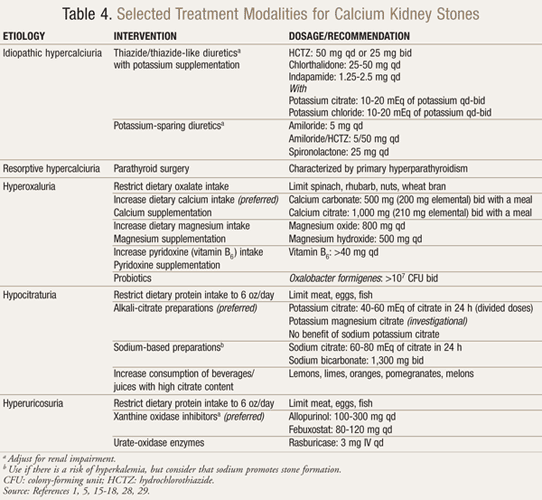
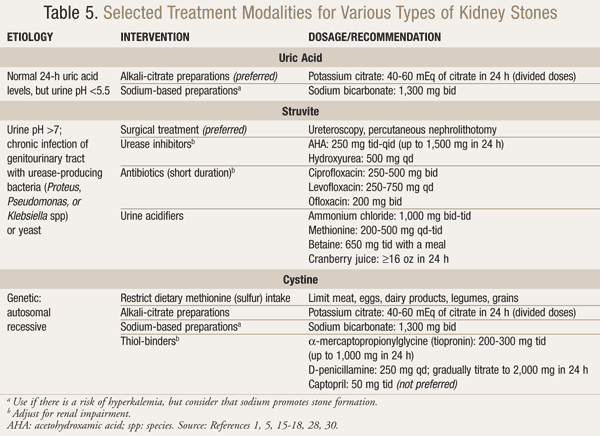
Definitive therapy, which is based on stone type and etiology, is outlined in TABLES 4 and 5.1,5,15-18,28-30
Discharge Planning and Prevention
Prior to discharge, patients should receive information about follow-up appointments in urologic clinics, potential surgical procedures, definitive therapies, and methods for preventing or minimizing kidney stone formation.15-17 They may need to modify their diets to increase fluid intake, minimize oxalate-containing food consumption, and/or limit animal protein intake.1,5 After initial treatment for renal calculi, patients might continue taking a combination of oral opioids and NSAIDs for pain control.16
Surgery
Surgical procedures are indicated for the breakdown and removal of stones that remain in the urinary tract, particularly in patients with obstruction.1,15,16 These procedures include, but are not limited to, extracorporeal shock wave lithotripsy (SWL), ureteroscopy (URS), and percutaneous nephrolithotomy (PCNL).1,2,15,16 Each depends on the size and location of the ureteral stone and is associated with various limitations and complications.1,2,15,16 Of note, patients should avoid medications that increase bleeding risk (e.g., NSAIDs, aspirin) several days prior to SWL.16
Conclusion
Following diagnostic imaging, the treatment of most acute kidney stones is hydration and pain management. Although many factors contribute to renal calculi formation, recurrence could be minimized. Along with other health care professionals, pharmacists should be aware of drug-induced causes and counsel patients to adhere to definitive therapies and recommended diets.
REFERENCES
1. Turk C, Knoll T, Petrik A, et al. Guidelines on
urolithiasis. European Association of Urology 2011.
www.uroweb.org/gls/pdf/18_Urolithiasis.pdf. Accessed April 21, 2013.
2. Preminger GM, Tiselius HG, Assimos DG, et al. 2007 guideline for the management of ureteral calculi. J Urol. 2007;178:2418-2434.
3. Urinary tract stones. In: Litwin MS, Saigal CS, eds. Urologic Diseases in America.
National Institute of Diabetes and Digestive and Kidney Diseases.
Washington, DC: U.S. Government Printing Office; 2012. NIH Pub. No.
12-7865.
4. Evan AP. Physiopathology and etiology of stone formation in the kidney and the urinary tract. Pediatr Nephrol. 2010;25:831-841.
5. Eisner BH, Goldfarb DS, Pareek G. Pharmacologic treatment of kidney stone disease. Urol Clin North Am. 2013;40:21-30.
6. Matlaga BR, Shah OD, Assimos DG. Drug-induced urinary calculi. Rev Urol. 2003;5:227-231.
7. Fogazzi GB. Crystalluria: a neglected aspect of urinary sediment analysis. Nephrol Dial Transplant. 1996;11:379-387.
8. Blossom AP, Cleary JD, Daley WP. Acyclovir-induced crystalluria. Ann Pharmacother. 2002;36:526.
9. Fogazzi GB, Cantù M, Saglimbeni L, et al. Amoxycillin, a rare but possible cause of crystalluria. Nephrol Dial Transplant. 2003;18:212-214.
10. Rockwood N, Mandalia S, Bower M, et al.
Ritonavir-boosted atazanavir exposure is associated with an increased
rate of renal stones compared with efavirenz, ritonavir-boosted
lopinavir and ritonavir-boosted darunavir. AIDS. 2011;25:1671-1673.
11. Fogazzi GB, Garigali G, Brambilla C, et al. Ciprofloxacin crystalluria. Nephrol Dial Transplant. 2006;21:2982-2983.
12. Mallipattu SK, Ross MJ. Methotrexate in the urine. Kidney Int. 2011;80:226.
13. Roberts DM, Smith MW, McMullan BJ, et al. Acute kidney injury due to crystalluria following acute valacyclovir overdose. Kidney Int. 2011 ;79:574.
14. Auer BL, Auer D, Rodgers AL. Relative hyperoxaluria, crystalluria and haematuria after megadose ingestion of vitamin C. Eur J Clin Invest. 1998;28:695-700.
15. Wolf JS, Howes DS, Craig S, et al. Nephrolithiasis. Medscape Reference. http://emedicine.medscape.com/article/437096-overview. Accessed April 21, 2013.
16. Portis AJ, Sundaram CP. Diagnosis and initial management of kidney stones. Am Fam Physician. 2001;63:1329-1338.
17. Frassetto L, Kohlstadt I. Treatment and prevention of kidney stones: an update. Am Fam Physician. 2011;84:1234-1242.
18. Parmar MS. Kidney stones. BMJ. 2004;328:1420-1424.
19. Nuss GR, Rackley JD, Assimos DG. Adjunctive therapy to promote stone passage. Rev Urol. 2005;7:67-74.
20. Gratzke C, Uckert S, Kedia G, et al. In vitro effects
of PDE5 inhibitors sildenafil, vardenafil and tadalafil on isolated
human ureteral smooth muscle: a basic research approach. Urol Res. 2007;35:49-54.
21. Cha WH, Choi JD, Kim KH, et al. Comparison and
efficacy of low-dose and standard-dose tamsulosin and alfuzosin in
medical expulsive therapy for lower ureteral calculi: prospective,
randomized, comparative study. Korean J Urol. 2012;53:349-354.
22. Tsuzaka Y, Matsushima H, Kaneko T, et al. Naftopidil
vs. silodosin in medical expulsive therapy for ureteral stones: a
randomized controlled study in Japanese male patients. Int J Urol. 2011;18:792-795.
23. Resorlu B, Bozkurt O, Senocak C, et al. Effectiveness
of doxazosin in the management of lower ureteral stones in male and
female patients. Int Urol Nephrol. 2011;43:645-649.
24. Gurbuz MC, Polat H, Canat L, et al. Efficacy of three
different alpha 1-adrenergic blockers and hyoscine N-butylbromide for
distal ureteral stones. Int Braz J Urol. 2011;37:195-202.
25. Dellabella M, Milanese G, Muzzonigro G.
Medical-expulsive therapy for distal ureterolithiasis: randomized
prospective study on role of corticosteroids used in combination with
tamsulosin-simplified treatment regimen and health-related quality of
life. Urology. 2005;66:712-715.
26. Porpiglia F, Destefanis P, Fiori C, et al.
Effectiveness of nifedipine and deflazacort in the management of distal
ureter stones. Urology. 2000;56:579-582.
27. Borghi L, Meschi T, Amato F, et al. Nifedipine and
methyl-prednisolone in facilitating ureteral stone passage: a
randomized, double-blind, placebo-controlled study. J Urol. 1994;152:1095-1098.
28. Moe OW, Pearle MS, Sakhaee K. Pharmacotherapy of urolithiasis: evidence from clinical trials. Kidney Int. 2011;79:
385-392.
29. Siener R, Bangen U, Sidhu H, et al. The role of Oxalobacter formigenes colonization in calcium oxalate stone disease. Kidney Int. 2013;83:1144-1149.
30. Zanetti G, Paparella S, Trinchieri A, et al.
Infections and urolithiasis: current clinical evidence in prophylaxis
and antibiotic therapy. Arch Ital Urol Androl. 2008;80:5-12.
To comment on this article, contact rdavidson@uspharmacist.com.






