US Pharm. 2009;34(5):26-34.
Chronic pain and depression are highly prevalent conditions whose symptoms overlap. A large number of population surveys have found a positive association between depression and chronic pain.1 A study analyzing data from the National Health and Nutrition Examination Survey III concluded that depression affects 7% to 10% of the U.S. population and that up to 90% of these patients have reported pain symptoms.2
Pathophysiology
The link between depression and pain may be both psychological and biological. The biological basis for depression has focused on dysregulation of the neurotransmitters serotonin (5-hydroxytryptamine, or 5-HT), norepinephrine (NE), and dopamine.
NE and 5-HT have been implicated in the underlying pathophysiology of chronic pain.3 Pain originates in the primary sensory neurons and terminates in the dorsal horn of the spinal cord. In the dorsal horn, pain signals activate many brain structures through the ascending pain pathway; the final result is pain manifesting clinically. In the spinal cord, the descending fibers originating in the brainstem suppress pain neurotransmission; this suppression functions as a homeostatic regulator. The neurotransmitters released by the descending fibers are 5-HT and NE. A dysfunctional 5-HT or NE system is likely to have a dysfunctional descending 5-HT or NE pathway, which explains comorbid pain symptoms in patients with depression.4,5
Patients with chronic pain are prone to depression because of the continuous disease burden imposed by the pain. Because of the reciprocal nature of depression and pain, the presence of both conditions in a patient may lead to an increased number of medical visits as well as to higher health care costs.3
Physical Pain With Depression
Major depressive disorder (MDD) is a multifaceted disease that presents with both emotional symptoms (e.g., depression, guilt, suicidal ideation) and physical symptoms (e.g., sleep disruption, gastrointestinal disturbance, unexplained aches and pains). Headache, neck and back pain, abdominal pain, and musculoskeletal pain are common in patients with depression.6
Antidepressants that work upon 5-HT or NE receptors--such as selective serotonin reuptake inhibitors (SSRIs) and serotonin-norepinephrine reuptake inhibitors (SNRIs)--have been examined in several randomized, double-blind studies of the treatment of pain associated with depression. In one trial, duloxetine (an SNRI) was studied in 512 patients with MDD.7 Patients received either duloxetine 60 mg/day or placebo. The primary outcome, pain severity, was measured by visual analogue scale (VAS). Duloxetine was associated with significant improvements in back pain (P = .020) and shoulder pain (P = .021) at week 9. Two randomized, double-blind studies found, similarly, that duloxetine 60 mg once/day significantly reduced painful symptoms compared with placebo in patients with MDD.8,9
Another randomized, double-blind study compared the effectiveness of duloxetine 40 mg/day, duloxetine 80 mg/day, and paroxetine (an SSRI) 20 mg/day versus placebo in 353 patients with MDD.10 Pain was measured by VAS. Duloxetine 80 mg/day caused a significantly greater improvement in overall pain severity compared with placebo (P <.001); however, neither duloxetine 40 mg/day nor paroxetine was more effective than placebo for relieving pain. Patients receiving duloxetine 80 mg/day experienced more insomnia (19.8%) than those receiving paroxetine 20 mg/day (8.0%). The authors concluded that inhibition of both 5-HT and NE was necessary to relieve physical pain occurring with depression.
A randomized, open-label, intent-to-treat study confirmed findings that SSRIs were not effective for relieving symptoms of physical pain associated with depression.11 This study involved 573 patients with depression who were randomized to receive fluoxetine, sertraline, or paroxetine for 9 months. Pain was measured by the Patient Health Questionnaire somatic symptom module at 1, 3, 6, and 9 months. Pain symptoms improved during the first month of treatment, but plateaued with minimal resolution in subsequent months.
Another SNRI, venlafaxine, was investigated in an open-label, prospective cohort study of 186 patients with MDD who had failed treatment with an SSRI or a tricyclic antidepressant (TCA).12 Patients received venlafaxine (150 mg/day or more) for 1 year. VAS was used to measure pain. Venlafaxine (mean dose 225 mg/day) significantly improved pain symptoms in depressed patients compared with baseline. One limitation was that this was an open-label study with no placebo group; also, the study allowed the use of cyclo-oxygenase-2 inhibitors, which affected pain outcomes.
In summary, SNRIs such as duloxetine and venlafaxine have been shown to be effective for relieving physical pain occurring with depression. On the other hand, there has been a lack of evidence to prove that SSRIs are effective. See FIGURE 1 for a treatment algorithm.
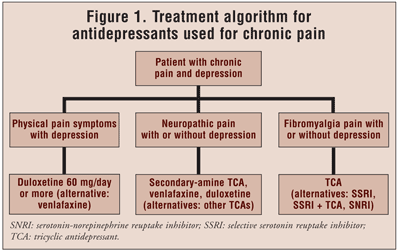
Neuropathic Pain
Two common forms of chronic neuropathic pain are diabetic neuropathy and postherpetic neuralgia, which account for 3 million and 1 million people, respectively, in the United States.13,14 Current evidence-based guidelines recommend the use of TCAs and SNRIs as first-line options for the treatment of neuropathic pain (TABLE 1).15,16 When a TCA is used, secondary-amine TCAs (nortriptyline, desipramine) are preferred because they are better tolerated than tertiary-amine TCAs (amitriptyline, imipramine) and have comparable analgesic efficacy.15 Both nortriptyline and desipramine can be initiated at a starting dose of 10 mg to 25 mg/day (taken at night) and increased by 10 mg to 25 mg/day every 3 to 7 days as tolerated. The maximum dosage of nortriptyline and desipramine is 75 mg to 150 mg/day. If the blood level is less than 100 ng/mL, however, titration can be continued as needed. It may take 6 to 8 weeks to see the full effect of the medication.17
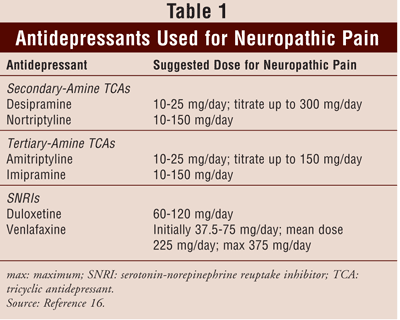
A meta-analysis evaluating the effectiveness and safety of antidepressants for neuropathic pain found that TCAs and venlafaxine had a number needed to treat of 3.6 and 3.1, respectively, for achieving at least moderate pain relief.18 Although the effectiveness of TCAs and SNRIs appeared to be comparable, TCAs were associated with more adverse effects than SNRIs. For amitriptyline, the number needed to harm for major adverse events was 28; for venlafaxine it was 16.18 Another randomized, double-blind, placebo-controlled study of 40 patients with polyneuropathy found, similarly, that both venlafaxine and imipramine were effective for pain relief compared with placebo, with no significant difference between the two treatment groups.19
Duloxetine received FDA approval for the treatment of diabetic neuropathy in 2004.16 In a randomized, double-blind, placebo-controlled study investigating the effectiveness and safety of duloxetine for this indication, 348 patients were randomly assigned to receive duloxetine 60 mg/day, duloxetine 120 mg/day, or placebo for 12 weeks.20 The primary outcome measure was the collection of weekly mean scores of 24-hour pain. Duloxetine treatment was associated with improved pain scores compared with placebo (P <.001). No significant difference in efficacy measures was found between the two duloxetine groups. The discontinuation rate due to adverse events was highest in the duloxetine 120-mg/day group (12.1%), followed by the 60-mg/day group (4.3%) and the placebo group (2.6%). When duloxetine is used for diabetic neuropathy, therefore, a daily dose of 60 mg is preferable to 120 mg owing to its comparable efficacy and fewer adverse events.
In summary, antidepressants such as TCAs and SNRIs are first-line agents for the treatment of neuropathic pain (FIGURE 1). When a TCA is used for neuropathic pain, a secondary amine is preferable to a tertiary amine. It is recommended to initiate the TCA at a low dose and slowly titrate up to the maximally effective dose.
Fibromyalgia
Fibromyalgia is a musculoskeletal condition characterized by widespread pain and muscle tenderness that is often accompanied by fatigue, sleep disturbances, and depression. It affects approximately 2% of the population.21 Current American Pain Society guidelines cite strong evidence for the efficacy of TCAs such as amitriptyline for fibromyalgia pain and modest evidence for the efficacy of SSRIs (fluoxetine alone or in combination with a TCA) and SNRIs (duloxetine and venlafaxine) for relieving fibromyalgia pain and improving quality of life.22,23 Currently, duloxetine is the only antidepressant with FDA approval for the treatment of fibromyalgia.16
Evaluation and Monitoring
The first step in determining whether an antidepressant may be effective for a patient with chronic pain should be an assessment of the etiology of the pain (FIGURE 2). In clinical practice, an evaluation of the patient's medical history may reveal diagnoses such as depression, diabetes, arthritis, or fibromyalgia as potential sources of the pain. If the etiology remains unknown, a physician or pain specialist should be consulted. A thorough review of the patient's medication history (in the chart or during a visit with the patient) is useful for excluding agents that were tried unsuccessfully in the past.
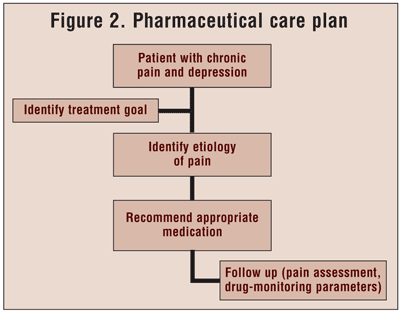
Pain Assessment: Pain assessment is a vital component of evaluating the effectiveness of pain treatment. In evaluating a patient with pain, a systematic approach is recommended to cover the basic parameters.24 Pain parameters include location, effect on function and activities of daily living, levels at rest and during activity, quality, radiation, timing, precipitating factors, and medication history. Quality can be evaluated by asking the patient to describe the pain (e.g., aching, throbbing). Timing can be described as occasional, intermittent, or constant.24 In a nonverbal patient, the emotional state and behaviors such as facial expression and moaning may indicate whether he or she is experiencing pain. A common tool for assessing severity is the VAS, which asks patients to rate their pain level from 0 (no pain) to 10 (unbearable pain).25 The Faces Pain Scale can be used to evaluate pediatric patients. Two common screening tools for depression--the Hamilton Rating Scale for Depression and the Brief Inventory of Depressive Symptoms--have questions that screen for somatic pain symptoms associated with depression.25
Adverse Effects: Prior to the initiation of antidepressant treatment for chronic pain, the potential for adverse effects caused by antidepressants should be carefully considered; the benefits should outweigh the risks. All antidepressants carry a black box warning detailing the increased risk of suicidal ideation in children and adolescents. The antidepressant dose should be started low and slowly titrated up while monitoring for adverse effects.25
The most common adverse effects associated with TCAs are sedation, dry mouth, blurred vision, constipation, urinary retention, weight gain, and orthostatic hypotension. Tertiary amines (amitriptyline, imipramine) are associated with more adverse effects than secondary amines (nortriptyline, desipramine). All TCAs can cause cardiac toxicity, including torsades de pointes and ventricular arrhythmia; they should be used cautiously, if not avoided, in patients with a history of ischemic heart disease, tachycardia, or conduction abnormalities.25 TCAs may predispose patients to falls and can cause or exacerbate cognitive impairment and gait disturbances in older patients; therefore, TCAs should not be used in the elderly population.15
SSRIs and SNRIs are better tolerated than TCAs. The most common adverse effects associated with SSRIs are sedation, agitation, insomnia, gastrointestinal effects such as nausea and diarrhea, anticholinergic effects, sexual dysfunction, and weight gain. SNRIs cause adverse effects similar to those of SSRIs, and they also can induce elevations in blood pressure.25 Nausea is the most commonly reported side effect of SNRIs, accounting for 22% to 25% of patients taking duloxetine; insomnia comes in second, occurring in 17% to 19% of patients taking duloxetine.10 Nausea and insomnia have been found to occur more often in patients taking duloxetine 40 mg/day compared with those taking paroxetine 20 mg/day.10
Monitoring Parameters: Patients taking antidepressants must be educated about the black box warnings associated with these medications. Distribution of medication guides can help ensure that patients receive necessary information. Patients should be monitored for agitation, irritability, unusual changes in behavior, mania, panic attacks, sleep disturbances, anxiety, and suicidal ideation.16 If the patient has depression, depressive symptoms should be closely monitored.
Patients taking TCAs should have their blood pressure checked periodically to screen for orthostasis and an increased risk of falling. A baseline electrocardiogram is recommended for patients with preexisting cardiac-conductivity defects. Therapeutic drug monitoring of TCA levels is not routinely performed but is recommended when a patient is elderly, nonresponsive to therapy, nonadherent, experiencing adverse effects, or having possible drug interactions. TCA serum concentrations may be increased by CYP2D6 inhibitors including fluoxetine, paroxetine, bupropion, duloxetine, and others. TCA levels may be checked at least once a week on a constant dosage when steady state is achieved and 12 hours after the last dose is administered.25 When a TCA is used in combination with fluoxetine in fibromyalgia patients, monitoring of blood levels is recommended.
Patients receiving SNRIs should be monitored for elevations in blood pressure and heart rate. Plasma drug monitoring of SSRIs and SNRIs is not recommended.25
Conclusion
Antidepressants have been proven effective for various types of chronic pain, including physical pain associated with depression, neuropathy, and fibromyalgia. Different antidepressant classes are effective for different types of pain. Pharmacists play a key role in assessing the effectiveness of antidepressants used for pain control and identifying potential risks of adverse effects and drug interactions. When a patient presents with depression and chronic pain, it is important to address both conditions effectively. It is also critical that pharmacists discuss with patients the risks and benefits of antidepressants, including the black box warnings, and to provide them with medication guides.
REFERENCES
1. Angst F, Verra ML, Lehmann S, et al. Refined insights into the pain-depression association in chronic pain patients. Clin J Pain. 2008;24:808-816.
2. Riolo SA, Nguyen TA, Greden JF, King CA. Prevalence of depression by race/ethnicity: findings from the National Health and Nutrition Examination Survey III. Am J Public Health. 2005;95:998-1000.
3. Bair MJ, Robinson RL, Katon W, Kroenke K. Depression and pain comorbidity: a literature review. Arch Intern Med. 2003;163:2433-2445.
4. Blier P, Abbott FV. Putative mechanisms of action of antidepressant drugs in affective and anxiety disorders and pain. J Psychiatry Neurosci. 2001;26:37-43.
5. Jann MW, Slade JH. Antidepressant agents for the treatment of chronic pain and depression. Pharmacotherapy. 2007;27:1571-1587.
6. Fava M. Somatic symptoms, depression, and antidepressant treatment. J Clin Psychiatry. 2002;63:305-307.
7. Fava M, Mallinckrodt CH, Detke MJ, et al. The effect of duloxetine on painful physical symptoms in depressed patients: do improvements in these symptoms result in higher remission rates? J Clin Psychiatry. 2004;65:521-530.
8. Brannan SK, Mallinckrodt CH, Brown EB, et al. Duloxetine 60 mg once-daily in the treatment of painful physical symptoms in patients with major depressive disorder. J Psychiatr Res. 2005;39:43-53.
9. Detke MJ, Lu Y, Goldstein DJ, et al. Duloxetine, 60 mg once daily, for major depressive disorder: a randomized double-blind placebo-controlled trial. J Clin Psychiatry. 2002;63:308-315.
10. Goldstein DJ, Lu Y, Detke MJ, et al. Duloxetine in the treatment of depression: a double-blind placebo-controlled comparison with paroxetine. J Clin Psychopharmacol. 2004;24:389-399.
11. Greco T, Eckert G, Kroenke K. The outcome of physical symptoms with treatment of depression. J Gen Intern Med. 2004;19:813-818.
12. Bradley RH, Barkin RL, Jerome J, et al. Efficacy of venlafaxine for the long term treatment of chronic pain with associated major depressive disorder. Am J Ther. 2003;10:318-323.
13. Schmader KE. Epidemiology and impact on quality of life of postherpetic neuralgia and painful diabetic neuropathy. Clin J Pain. 2002;18:350-354.
14. Bowsher D. The lifetime occurrence of Herpes zoster and prevalence of postherpetic neuralgia: a retrospective survey in an elderly population. Eur J Pain. 1999;3:335-342.
15. Dworkin RH, O'Connor AB, Backonja M, et al. Pharmacologic management of neuropathic pain: evidence-based recommendations. Pain. 2007;132:237-251.
16. USP DI Volume I: Drug Information for the Healthcare Professional. 27th ed. Greenwood Village, CO: Thomson Micromedex; 2007.
17. Dworkin RH, Backonja M, Rowbotham MC, et al. Advances in neuropathic pain: diagnosis, mechanisms, and treatment recommendations. Arch Neurol. 2003;60:1524-1534.
18. Saarto T, Wiffen PJ. Antidepressants for neuropathic pain. Cochrane Database Syst Rev. 2007;4:CD005454.
19. Sindrup SH, Bach FW, Madsen C, et al. Venlafaxine versus imipramine in painful neuropathy: a randomized, controlled trial. Neurology. 2003;60:1284-1289.
20. Raskin J, Pritchett YL, Wang F, et al. A double-blind, randomized multicenter trial comparing duloxetine with placebo in the management of diabetic peripheral neuropathic pain. Pain Med. 2005;6:346-356.
21. Arnold LM, Keck PE, Welge JA. Antidepressant treatment of fibromyalgia. A meta-analysis and review. Psychosomatics. 2000;41:104-113.
22. Burckhardt CS, Goldberg D, Crofford L, et al. Guideline for the Management of Fibromyalgia Syndrome Pain in Adults and Children. Glenview, IL: American Pain Society; 2005.
23. Goldenberg DL, Burckhardt C, Crofford L. Management of fibromyalgia syndrome. JAMA. 2004;292:2388-2395.
24. Registered Nurses' Association of Ontario. Assessment and Management of Pain: Supplement. Toronto, ON: Registered Nurses' Association of Ontario; 2007.
25. Koda-Kimble MA, Young LY, Kradjan WA, et al. Applied Therapeutics: The Clinical Use of Drugs. 8th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2000:9-1-39.
To comment on this article, contact rdavidson@jobson.com.





