US Pharm. 2014;39(4):35-40.
ABSTRACT: Prostatitis, which affects 5% to 9% of males and occurs mostly in middle age, is classified based on signs and symptoms, with urinary urgency, frequency, and pain typical in nearly all categories. Most physicians are not familiar with prostatitis, particularly chronic prostatitis associated with chronic pelvic pain syndrome (CP/CPPS). Accordingly, patients are often misdiagnosed and receive ineffective treatment, resulting in poor quality of life. CP/CPPS is challenging to treat, as its causes are not clearly defined and the antibiotics used for therapy have low effective rates. Clinical pharmacists can contribute significantly to patient care by advising physicians and other medical professionals regarding drug efficacy, adverse drug reactions, and drug interactions, and by assisting in the selection of optimal antibiotics and/or treatment regimens for prostatitis.
Prostatitis (inflammation of the prostate gland), which occurs in 5% to 9% of males aged 18 years and older, most often develops in middle age.1 In the early 1990s, prostatitis accounted for about 1% and 8% of office visits to family practitioners and urologists, respectively.1
In most cases, patients with prostatitis experience urinary urgency, frequency, and pain, all of which significantly impact quality of life (QOL).1-8 It is believed that the QOL of patients with prostatic pain is similar to that of patients with acute myocardial infarction, unstable angina, or active Crohn’s disease.1 A 2009 survey of 556 primary care physicians in Boston, Chicago, and Los Angeles found that only 62% saw patients for prostatitis; surprisingly, 48% of physicians surveyed were not familiar with prostatitis and 16% were unfamiliar with chronic prostatitis associated with chronic pelvic pain syndrome (CP/CPPS).4 Consequently, the diagnosis and effective treatment of prostatitis, especially CP/CPPS, pose a challenge for clinicians.
Signs and Symptoms
The National Institutes of Health (NIH) classifies prostatitis into four categories based on signs and symptoms (TABLE 1).1-8 Category I (acute bacterial prostatitis [ABP]) is rare, accounting for fewer than 0.02% of prostatitis patients. Approximately 5% of prostatitis cases are category II (chronic bacterial prostatitis [CBP]). Most chronic prostatitis patients (90%-95%) fall into category III (CP/CPPS), which is subdivided into IIIA (inflammation) and IIIB (no inflammation). Category IV (asymptomatic inflammatory prostatitis) is accidentally discovered during a physical examination or office visit for other genitourinary disorders (e.g., benign prostatic hyperplasia, prostate cancer, infertility, overactive bladder, and elevated prostate-specific antigen level).1-8,12

ABP is characterized by the acute onset of frequency, urgency (irritative and obstructive voiding), and severe pelvic pain (perineum, suprapubic area, and external genitalia), which are caused by inflammation of the prostate. In addition, patients may have bacteremia (fever, chills, and rigors) and possibly signs of sepsis. If ABP is not treated properly, about 5% of patients progress to CBP.1-8
CBP is typically associated with recurrent urinary tract infections (UTIs) with mild-to-moderate pelvic pain symptoms. Symptoms of CBP differ from those of ABP, as the levels of pain and systemic infection are milder (low-grade fever with back or pelvic discomfort) and may occur off and on.1-8
CP/CPPS is characterized by irritative urination (frequency and urgency) and chronic pelvic pain, with no evidence of UTI for at least 3 months in the previous 6 months. Chronic pelvic pain is the hallmark of CP/CPPS.4 In addition, in inflammatory CP/CPPS (category IIIA), leukocytes are found in expressed prostatic secretions (EPS), post–prostatic massage urine, or semen.1-8
Pathophysiology
Because ABP is an uncommon complication of UTI, urinary tract pathogens are typically the cause of category I prostatitis. Escherichia coli is most common, followed by Proteus, Klebsiella, and Pseudomonas species. Enterococci and Staphylococcus aureus are also found, but anaerobes are rare in ABP.1,2,5-7,9
CBP is associated with recurrent UTI, urethritis, bacteriospermia, and epididymitis. The most common gram-negative bacterium detected is E coli (65%-80% of infections), followed by Klebsiella and Pseudomonas species.9 Gram-positive enterococcus is also found in CBP, but only transiently. Based on experiments in animals, it appears that pathogens form colonies in the prostate, with special growth conditions leading to off-and-on episodes.1,3,9
The causes of CP/CPPS are not clearly understood or defined. Many hypotheses have been proposed to explain CP/CPPS pathology, but no single theory can adequately explain all CP/CPPS symptoms. A microorganism-based etiology has been proposed, but is controversial (infection detected only in 8% of patients).3 It is believed that Lactobacillus and Corynebacterium species and diphtheroids may be linked to inflammatory prostatitis (category IIIA). Coagulase-negative Staphylococcus, Chlamydia, and Ureaplasma species and anaerobes have been found localized to the prostate, but it is unclear whether these bacteria cause CP/CPPS.1,9 According to recent molecular biologic research, hidden bacterial infection of the prostate could be a cause of prostatitis. It is also theorized that interactions between psychological factors and immune-, neurologic-, and endocrine-system dysfunction may contribute to CP/CPPS exacerbation.1-3,5-7,10,11
• One study found that increased stress in men with CP/CPPS resulted in increased pain and disability at 12 months.10 Psychological stress and depression may influence cytokine production in the pelvis, leading to CP/CPPS inflammation.6
• Abnormal outflow tract and intraprostatic ductal reflux may cause painful urination.
• Chemical reactions may result in prostate duct obstruction, causing pain (e.g., reflux of Tamm-Horsfall mucoprotein or urate into prostate ducts).
• Immunologic dysfunction is a possibility and could be due to defects in androgen receptors. In one study, subjects with CP/CPPS had abnormal levels of adrenocortical hormone compared with healthy subjects. However, this does not prove that hormone abnormalities cause CP/CPPS; rather, they may cause its effects.11
• Neurogenic inflammation in the perineum and pelvis could be responsible for CP/CPPS.1-3
• An imbalance of cytokines may be linked to the development of pelvic inflammation and pain in CP/CPPS.6
• An increase of nerve growth factor due to abnormal nervous-system functioning has been correlated with the development of CP/CPPS symptoms.6
• Finally, myofascial pain syndrome, which typically is caused by repetitive muscle contraction due to repetitive motions (job-related) or stress, may contribute to the chronic pain of CP/CPPS.6
Diagnosis
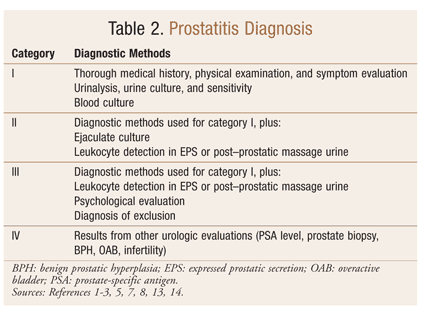
Prostatitis diagnosis (TABLE 2) includes a thorough medical history and physical examination, followed by symptom evaluation and laboratory work (urinalysis and urine culture, ejaculate culture, and leukocyte detection in EPS or post–prostatic massage urine).1-3,5,7,8
In the past, urine culture was performed via the Meares-Stamey segmented quantitative culture technique (four-glass testing, in which samples from initial urine, midstream urine, EPS, and post–prostatic massage urine are collected and cultured). However, owing to difficulties in implementation (time-consuming and difficult application in microbiology laboratories), the four-glass method has been replaced by two-glass testing, which is considered clinically equivalent.2,5,7 In two-glass testing, only midstream urine and post–prostatic massage urine samples are collected and cultured.
ABP is diagnosed through physical examination, symptom evaluation, urinalysis, urine culture, and sensitivity. Prostatic massage to obtain EPS should not be performed, because it could cause bacteremia; however, blood culture should be performed in order to detect bacteremia. The prostate is typically enlarged, tender, and indurate. Although the symptoms of ABP may be confused with those of UTI, ABP can be differentiated by prostatic inflammation and severe pelvic pain, in addition to the presence of pus and bacteria in the urine.5,7 Changes in the smell, color, or consistency of urine are unique clinical manifestations of ABP that differentiate it from CBP.12
In addition to the techniques used to diagnose ABP, ejaculate culture and leukocyte detection are performed to confirm CBP. CBP is diagnosed if the patient has had recurrent UTIs with a significant number of pathogenic bacteria in purulent prostatic fluid, plus the absence of a concurrent UTI or significant signs of systemic infection.7 To confirm CBP, the bacterial count of EPS and/or post–prostatic massage urine should be 10 times that of midstream urine. In addition, leukocyte or noncellular markers of inflammation (leukocyte elastase or interleukin-8) must be detected in EPS or post–prostatic massage urine.2 For counting leukocytes in the ejaculate, special stains (peroxidase stain) must be used to differentiate leukocytes from precursors of spermatozoa.2 In the case of treatment resistance, prostate imaging should be used to confirm prostatic abscess.2
CP/CPPS is diagnosed by a history of chronic pelvic pain without documented infections for at least 3 months within the previous 6 months. The hallmark symptom of CP/CPPS is pain (more intense in the anorectal and genital areas, but may occur anywhere in the pelvic area). The NIH Chronic Prostatitis Symptom Index (NIH-CPSI) must be used to validate CP/CPPS. If the total pain score is above 10, then the patient likely has CP/CPPS.1-4,7
Leukocyte detection in EPS and post–prostate massage urine is required to differentiate category IIIA prostatitis from category IIIB. A physical examination of the prostate and other pelvic structures should be conducted to exclude other disorders that can cause pelvic pain, such as prostate duct obstruction, primary bladder neck obstruction, detrusor-sphincter dyssynergia, urethritis, and prostate cancer.2,5,13 A psychological evaluation should be performed to ensure optimal treatment of CP/CPPS patients.
Treatment
Specific antibiotic treatment regimens for ABP and CBP are outlined in TABLE 3.
![]()
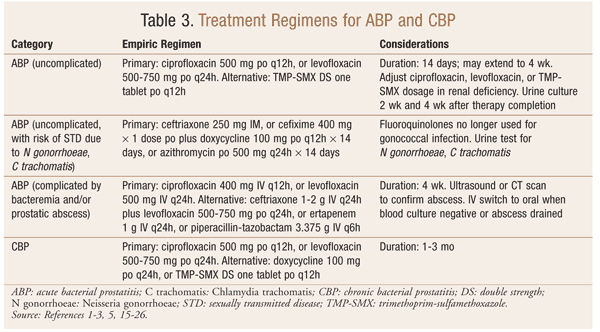
In hospitals, ABP complicated by bacteremia and/or prostatic abscess is treated aggressively with initial empirical parenteral antibiotics, e.g., a broad-spectrum penicillin with a beta-lactam inhibitor, a third-generation cephalosporin, fluoroquinolones, or an aminoglycoside combined with ampicillin. When the culture result is available, specific antibiotics should be chosen based on the culture’s sensitivity and susceptibility. After the initial parenteral treatment, further treatment with oral fluoroquinolones for 2 to 4 weeks should be administered to ensure a complete cure. For outpatients, oral fluoroquinolones (the most common regimen is ciprofloxacin 500 mg orally twice daily) may be initiated for 2 to 4 weeks.2 According to one source, the optimal treatment duration is 6 weeks.5 In the case of renal deficiency, the dosing of ciprofloxacin, levofloxacin, trimethoprim-sulfamethoxazole (TMP-SMX), ertapenem, or piperacillin-tazobactam should be adjusted based on renal clearance.15 In the case of prostatic abscess, in addition to the antibiotic treatment, prostate drainage should be performed.
The drug of choice for CBP is an oral fluoroquinolone administered for 4 to 6 weeks (may be extended to 3 months).2,16 Fluoroquinolones are safe and effective for CBP because of their excellent pharmacodynamic properties, good prostate-tissue penetration, high bioavailability, equivalence between parenteral and oral forms, and low resistance.5 Norfloxacin 400 mg twice daily for 28 days, ofloxacin 400 mg daily for 14 days, and ciprofloxacin 500 mg twice daily for 28 days (used to treat CBP refractory to TMP-SMX or carbenicillin) show similar results, with cure rates from 60% to 92%.1,17-20 However, ciprofloxacin is commonly prescribed by physicians and is the drug of choice, owing to its superior cure rates.
For fluoroquinolone resistance, doxycycline or TMP-SMX should be given for 3 months. Intermittent antibiotic treatment may be used for acute symptoms of each episode, and a low-dose antibiotic may be used for suppression. The last resort for resistant CBP is radical transurethral resection of the prostate (TURP) or simple prostatectomy.2 TURP is considered if the patient has one or more of the following symptoms: urinary retention, frequent UTIs, prostatic bleeding, bladder stones with prostate enlargement, extremely slow urination, and renal damage. The most common adverse effect of TURP is erectile dysfunction.1,2
CP/CPPS treatment (TABLE 4) has low efficacy because the condition’s etiology is poorly understood and standard antibiotic therapy is not successful.3,21 The widespread use of antibiotics for CP/CPPS is based on an old assumption that infection is the main cause of this type of prostatitis. Actually, however, CP/CPPS could be caused by different factors, such as urinary, psychosocial, organ-specific, infection, and neurologic/systemic muscular tenderness (UPOINT) symptoms.22,23 Therefore, multimodal symptomatic therapy is preferred for CP/CPPS. Clinicians can use the UPOINT classification system to design an individualized therapeutic plan for each CP/CPPS patient. In a recent prospective study, multimodal treatment using the UPOINT system resulted in significant improvement in CP/CPPS patients with a minimum follow-up of 6 months and an average follow-up of 50 weeks (total NIH-CPSI score ≥6 points, 84%; ≥50% improvement in total NIH-CPSI score, 51%).3,22-25
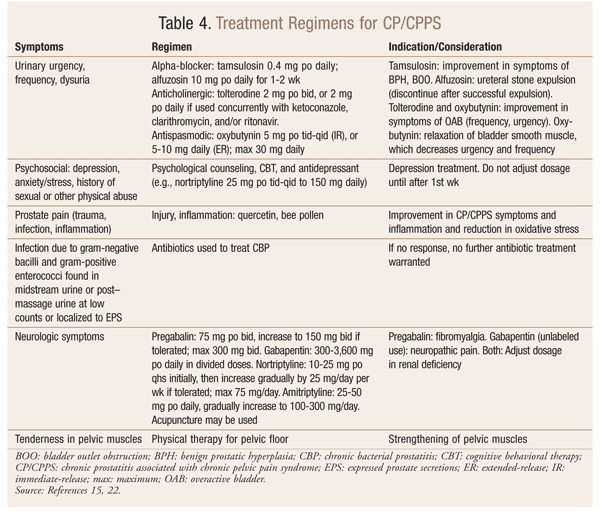
Alternative treatments for CP/CPPS include herbal products and acupuncture. In a randomized, double-blind, placebo-controlled study, Prosta-Q (a combination of zinc, quercetin, cranberry, saw palmetto, bromelain, and papain) led to a 25% or greater improvement in NIH pain and QOL symptom scores in 82% of patients.26-28 Several trials found that acupuncture significantly reduces pain and QOL scores after 5 to 6 weeks.29,30
The following therapies are not recommended for CP/CPPS: alpha-receptor blocker and antimicrobial therapy for patients with prior multiple therapies; anti-inflammatory monotherapy; alpha-reductase inhibitor monotherapy; minimally invasive therapies such as laser and transurethral needle ablation of the prostate; and invasive surgical therapies such as TURP and radical prostatectomy.1,2
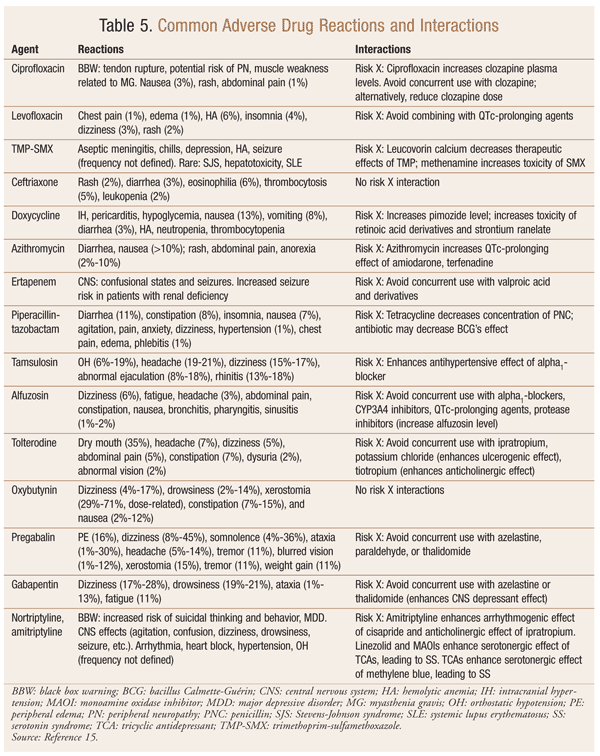
Pharmacists can contribute significantly to patient care by helping physicians select the best antibiotic regimen (optimal efficacy and fewest adverse effects) and by assisting them to develop individualized treatment plans based on the patient’s CP/CPPS symptoms. Pharmacists can also review the profiles of polypharmacy patients to minimize serious interactions (risk X) between drugs used for CP/CPPS and those indicated for other concurrent conditions, such as diabetes and cardiovascular disease. TABLE 5 gives examples of common adverse drug reactions and interactions.15 Detailed information about other adverse drug reactions and interactions is found in pharmacy references such as Lexicomp Online and Micromedex.
Conclusion
Prostatitis is a common disorder in males aged 18 years and older. Prostatitis is classified into four categories by the NIH, and each category presents with different signs and symptoms. Although the causes of ABP and CBP are clearly identified, the etiology of CP/CPPS is unclear, which leads to ineffective treatment and results in poor QOL for related patients. Since most physicians are not familiar with CP/CPPS, the treatment of CP/CPPS becomes a challenge for clinicians. Consequently, clinical pharmacists can significantly contribute to developing an optimal treatment regimen for each CP/CPPS patient by collaborating with and/or consulting physicians or medical teams about drug efficacy, adverse drug reactions, and drug interactions.
REFERENCES
1. Nickel JC, Weidner W. Chronic prostatitis: current concepts and
antimicrobial therapy. www.medscape.com/viewarticle/412693_4. Accessed
January 2, 2013.
2. Wagenlehner FM, Naber KG, Bschleipfer T, et al. Prostatitis and male pelvic pain syndrome: diagnosis and treatment. Dtsch Arztebl Int. 2009;106:175-183.
3. Lee KS, Choi JD. Chronic prostatitis: approaches for best management. Korean J Urol. 2012;53:69-77.
4. Calhoun EA, Clemens JQ, Litwin MS, et al. Primary care physician
practices in the diagnosis, treatment and management of men with chronic
prostatitis/chronic pelvic pain syndrome. Prostate Cancer Prostatic Dis. 2009;12:288-295.
5. Sharp VJ, Takacs EB, Powell CR. Prostatitis: diagnosis and treatment. Am Fam Physician. 2010;82:397-406.
6. Watson RA. Chronic pelvic pain in men. http://emedicine.medscape.com/article/437745-overview. Accessed October 6, 2013.
7. ACP Medicine. Prostatitis. http://enotes.tripod.com/prostatitis.htm. Accessed August 11, 2013.
8. Nickel JC. Prostatitis. Can Urol Assoc J. 2011;5:306-315.
9. Nickel JC. Chronic prostatitis: an infectious disease? www.medscape.com/viewarticle/410204. Accessed March 9, 2013.
10. Ulrich PM, Turner JA, Ciol M, Berger R. Stress is associated with
subsequent pain and disability among men with nonbacterial
prostatitis/pelvic pain. Ann Behav Med. 2005;30:112-118.
11. Pontari MA, Ruggieri MR. Mechanisms in prostatitis/chronic pelvic pain syndrome. J Urol. 2004;172:839-845.
12. Church DL. Prostatitis. Antimicrobe [online database]. www.antimicrobe.org/e53.asp. Accessed March 4, 2014.
13. Watson RA. Chronic pelvic pain in men workup.
http://emedicine.medscape.com/article/437745-workup. Accessed October
20, 2013.
14. Watson RA. Chronic pelvic pain in men differential diagnoses.
http://emedicine.medscape.com/article/437745-differential. Accessed
October 20, 2013.
15. Lexicomp Online [subscription database]. http://online.lexi.com. Accessed March 6, 2014.
16. Cunha BA. Antibiotic Essentials. 7th ed. Burlington, MA: Jones & Bartlett; 2008.
17. Schaeffer AJ, Darras FS. The efficacy of norfloxacin in the
treatment of chronic bacterial prostatitis refractory to
trimethoprim-sulfamethoxazole and/or carbenicillin. J Urol. 1990;144;690-693.
18. Weiner W, Schiefer HG. Chronic bacterial prostatitis: therapeutic experience with ciprofloxacin. Infection. 1991;19(suppl 3):S165-S166.
19. Weidner W, Ludwig M, Brähler E, Schiefer HG. Outcome of
antibiotic therapy with ciprofloxacin in chronic bacterial prostatitis. Drugs. 1999;58(suppl 2):103-106.
20. Naber KG, Busch W, Focht J. Ciprofloxacin in the treatment of
chronic bacterial prostatitis: a prospective, non-comparative
multicentre clinical trial with long-term follow-up. The German
Prostatitis Study Group. Int J Antimicrob Agents. 2000;14:143-149.
21. Alexander RB. Treatment of chronic prostatitis. Nat Clin Pract Urol. 2004;1:2-3.
22. Shoskes DA, Nickel JC, Rackley RR, Pontari MA. Clinical
phenotyping in chronic prostatitis/chronic pelvic pain syndrome and
interstitial cystitis: a management strategy for urologic chronic pelvic
pain syndromes. Prostate Cancer Prostatic Dis. 2009;12:177-183.
23. Watson RA. Chronic pelvic pain in men treatment & management.
http://emedicine.medscape.com/article/437745-treatment. Accessed
October 20, 2013.
24. Perletti G, Magri V. α-Blockers and antibiotics, alone or in
combination, improve symptoms in men with chronic prostatitis/chronic
pelvic pain syndrome. www.medscape.com/viewarticle/750499. Accessed
August 11, 2013.
25. Stein J. Intensive therapy regimen helps men with chronic pelvic
pain syndrome. www.medscape.com/viewarticle/722735. Accessed January 25,
2013.
26. New study offers hope for chronic prostatitis treatment. Medscape Medical News. www.medscape.com/viewarticle/411511. Accessed January 2, 2013.
27. Shoskes DA, Zeitlin SI, Shahed A, Rajfer J. Quercetin in men with
category III chronic prostatitis: a preliminary prospective,
double-blind, placebo-controlled trial. Urology. 1999;54:960-963.
28. Shoskes DA. Use of the bioflavonoid quercetin in patients with longstanding chronic prostatitis. J Am Neutraceutical Assoc. 1999;2:18.
29. Honjo H, Kamoi K, Naya Y, et al. Effects of acupuncture for
chronic pelvic pain syndrome with intrapelvic venous congestion:
preliminary results. Int J Urol. 2004;11:607-612.
30. Chen R, Nickel JC. Acupuncture ameliorates symptoms in men with chronic prostatitis/chronic pelvic pain syndrome. Urology. 2003;61:1156-1159.
To comment on this article, contact rdavidson@uspharmacist.com.





