US Pharm.
2008;33(8):49.
Androgenic
alopecia, or male pattern baldness, is the most common form of alopecia,
occurring in more than 50% of men. The prevalence increases with age, and
approximately 80% of Caucasian males experience the condition by age 70. The
incidence varies by race, and Caucasian males are affected more often than
males of other races. 1 Hair loss is gradual for most men,
occurring over a 15- to 25-year period; however, some men experience complete
hair loss within a period of five years or less.2 Hair loss usually
occurs in patterns as defined by the Hamilton-Norwood scale of male pattern
baldness, starting with recession of the frontal hairline and proceeding with
progressive thinning and loss of the hair on the vertex of the scalp. These
two regions of hair loss eventually meet, leading to baldness of the top of
the scalp. This is the typical progression of hair loss; however, variations
may occur.3 The pattern typically resembles an "M" in shape.4
Pathophysiology of Alopecia
Androgenic alopecia
is a hereditary condition. There is debate over the specific gene that causes
it; however, some believe that androgenic alopecia is passed down from father
to son.3 The condition is directly linked to androgens,
particularly dihydrotestosterone (DHT). DHT is the most abundant androgen in
the skin and is converted from testosterone by alpha reductase.5
The impact of androgen on hair is site specific. For example, hair located on
the chest, pubic area, and beard area reacts positively to the presence of
androgens by producing terminal--or thick, pigmented--hair. In contrast, hair
follicles located on the head are reduced in size and produce nonpigmented
vellus hairs in response to androgens. Over time, the hair growth cycle is
affected by androgens.
The hair growth cycle involves
three phases: the anagen, catagen, and telogen phases. The anagen, or hair
growth phase, is the first phase of hair growth, lasting between two to six
years. This phase is followed by the catagen phase, a transition phase lasting
approximately one to two weeks, and subsequently by the telogen, or resting,
phase lasting five to six weeks. After the telogen phase, the anagen phase
starts again.3 Men with alopecia have a shorter anagen phase and a
longer telogen phase, which eventually results in very short hair that fails
to emerge from the follicle, thereby causing baldness.2,3
Psychological Impact of
Androgenic Alopecia
Alopecia not only affects men
physically, but can also have a psychological impact on those afflicted by the
condition. Men often associate hair with attractiveness and virility. Once
hair loss occurs, men can lose confidence and satisfaction with their physical
appearance. Studies have shown that factors such as young age, no involvement
in a romantic relationship, and preexisting, poor self-esteem contribute to a
greater risk of a psychological impact from hair loss.1 It has also
been noted that males experiencing hair loss have a greater incidence of
depression and anxiety than those who do not experience this condition. There
are limitations to this finding, as stressful life events could also lead to
psychological manifestations or even alopecia.6 The psychological
effects of the condition need to be taken into account by practitioners as
they counsel men with androgenic alopecia and educate them on available
treatment options.
There are currently two
FDA-approved pharmacologic treatments for androgenic alopecia: minoxidil and
finasteride.
Minoxidil
Minoxidil topical solution was the
first of the FDA-approved treatments for androgenic alopecia to be placed on
the market. This product was first used in the oral form for the treatment of
hypertension and was found to have a side effect of hypertrichosis.7
The exact mechanism of action in hair growth is unclear; however, the proposed
mechanisms of action are normalization of hair follicle structure, conversion
of vellus hairs to terminal hairs, and amplification of the number of hair
follicles in the anagen phase of hair growth.8 It is clear that the
mechanism is not due to vasodilation, as hair continues to grow in the absence
of blood flow.9
Minoxidil is available in a 2%
and 5% topical solution and a 5% foam, all of which are available to consumers
OTC. Numerous studies have been conducted indicating that minoxidil causes
regrowth of hair. Two double-blind, placebo-controlled trials studied hair
weight measurements and found that both strengths of minoxidil were more
effective than placebo in growing hair. Over a period of two years, hair loss
subsided in patients treated with both strengths of minoxidil while hair loss
continued in those treated with placebo. Subsequently, discontinuation of the
product resulted in hair loss in the treatment group that matched hair loss in
the placebo group within a period of six months.7 Additional
studies have proven that the 5% solution is superior in efficacy to the 2%
solution in males with androgenic alopecia.9
A one-year safety trial was
conducted in over 20,000 patients using topical minoxidil and assessed
patients for an increased risk of hospitalization or death. At the end of the
trial it was found that topical minoxidil did not cause an increased risk of
hospitalization or death in these patients. The study looked specifically at
cardiovascular events and found no significant difference in patients using
minoxidil compared to those in the control group. In addition, more than 90%
of patients in the trial at six, nine, and 12 months felt that the product was
working to reduce hair loss.10
Minoxidil is helpful in males
experiencing frontal or vertex alopecia or thinning of hair. The growth is not
due to the formation of new hair, but to the transformation of existing hair
from miniaturized hair follicles to terminal hair. After that point, there is
stabilization of the hair that has grown.7
Although minoxidil promotes
hair growth in men, the therapy does have the potential to create compliance
issues. A patient must continue use of the product indefinitely or hair loss
will occur. Men using minoxidil should be encouraged to use the 5% solution if
tolerated. Patients should be counseled to apply the solution directly to the
scalp to maximize effectiveness and minimize cosmetic issues such as hair
oiliness. If applied directly to the hair, the product leaves a sticky
residue. Patients should also be told that response to treatment is variable.8
Those experiencing pruritus of the scalp should be given a topical
corticosteroid to use along with minoxidil.9 Patients should not
expect results until four to six months into therapy and should not look for
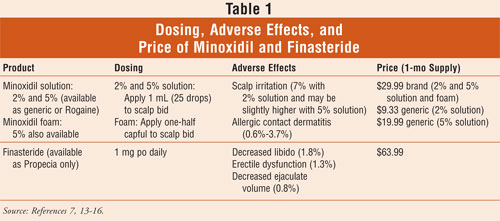
maximum results until at least one year of use. Dosing, adverse events, and
pricing of minoxidil are included in TABLE 1.
Overall, minoxidil appears to
be a safe and effective therapy in select patients with androgenic alopecia;
however, patients should realize that efficacy depends on continued use of the
product.
Finasteride
A second product,
indicated for the treatment of androgenic alopecia, is oral finasteride.
Finasteride is an inhibitor of the type II 5-alpha reductase isoenzyme, which
is predominant in the scalp hair root sheath.8 By blocking 5-alpha
reductase, finasteride inhibits the conversion of testosterone to DHT.7
As discussed earlier, DHT can lead to hair loss in the scalp. Finasteride 1 mg
has been found to reduce DHT levels in the scalp by over 60%.5
Clinical trials have proven
the efficacy of finasteride. Several of the trials, treating men with vertex
and midscalp hair loss, found a significant increase in hair count in patients
treated with finasteride 1 mg versus placebo. Hair growth occurred over the
first year, and the drug must be continued indefinitely to maintain new hair.1,7
Evidence of hair growth can become apparent within the first four months of
treatment; however, patients should continue treatment for at least 24 months
before deciding upon its effectiveness.2 Studies have concluded
that finasteride stabilizes hair loss in 80% of patients with vertex hair loss
and in 70% of patients with frontal hair loss. In addition, hair regrowth
occurs in 37% of men with frontal hair loss and in 61% of men with vertex hair
loss.7 Dosing, adverse effects, and pricing are included in TABLE
1.
The FDA has approved the use
of finasteride for patients ages 18 to 41 with mild to moderate male pattern
baldness in the midfrontal area. Patients should be made aware that no change
in hair density indicates that the product is effective because it has
maintained existing hair. In patients with androgenic alopecia, hair loss will
continue over time. Caution should be taken by women of childbearing age when
handling the product due to the potential to cause abnormalities of the
external genitalia in a developing male fetus.11 There are no
studies evaluating the semen of men taking finasteride and the potential for
problems to a fetus; however, the product is labeled as Pregnancy Category X.9
A randomized, open,
comparative trial in 65 patients with androgenic alopecia compared minoxidil
5% to finasteride 1 mg. The study was carried out over a period of 52 weeks in
males ranging from 18 to 50 years. At the end of the study, 80% of patients in
the finasteride group experienced some form of hair growth in comparison to
52% of patients in the minoxidil group (P <0.05). Both products were
well tolerated and showed efficacy in some patients.11 One open,
randomized, parallel-group study was conducted in 100 males with androgenic
alopecia and compared finasteride 1 mg, topical 2% minoxidil, and 2%
ketoconazole shampoo alone and all three in combination. It was found that
finasteride alone or in combination with the other agents provided a
statistically significant improvement in hair growth over minoxidil alone.12
Other Therapies
Several other drugs
have been evaluated for their potential role in patients with androgenic
alopecia; however, the sample sizes are very small and outcomes are varied.
Small studies have been conducted using a combination of topical tretinoin
with topical minoxidil, and these resulted in some hair growth. A drawback to
this combination is that the two products cannot be compounded into one
solution due to incompatibility; therefore, they must be applied at different
times of the day. In addition, some patients cannot tolerate the irritation
experienced with topical tretinoin, which has not been widely studied to
decide whether or not this is an appropriate therapy. Spironolactone 200 mg
daily has been studied in a few small trials due to its mild antiandrogenic
effect and has shown some clinical benefit; however, it is not widely used.7
In addition to pharmacologic
treatment, there are several nonpharmacologic options for men experiencing
androgenic alopecia. These include hair transplantation, scalp reduction
surgery, and cosmetic aids such as wigs and hairpieces.1
To date, there are no other
approved products for androgenic alopecia; however, minoxidil and finasteride
are safe, effective treatments for the condition. The results with these
products are not miraculous; still, they have been shown to reduce the
progression of hair loss and promote hair growth in men. Men need to be
counseled on realistic expectations of the products and must realize that
results vary from patient to patient. They must also be committed to a
lifetime of treatment, as discontinuation of both products results in a
continued progression of hair loss.
REFERENCES
1. Stough D, Stenn K, Haber R, et al. Psychological effect, pathophysiology, and management of androgenic alopecia in men. Mayo Clin Proc. 2005;80:1316-1322.
2. Sinclair R. Fortnightly review: male pattern androgenetic alopecia. BMJ. 1998;317:865-869.
3. Ellis JA, Sinclair R, Harrap SB. Androgenic alopecia: pathogenesis and potential for therapy. Expert Rev Mol Med.2002:1-11.
4. Springer K, Brown M, Stulberg DL. Common hair loss disorders. Am Fam Physician. 2003;68:93-102.
5. Otberg N, Finner A, Shapiro J. Androgenic alopecia. Endocrinol Metab Clin North Am. 2007;36:379-398.
6. Hunt N, McHale S. The psychological impact of alopecia. BMJ. 2005;331:951-953.
7. Bolduc C, Shapiro J. Management
of androgenic alopecia. Am J Clin
Dermatol.
2000;3:151-158.
8. Sinclair, RD. Management of male pattern hair loss. Cutis. 2001;68:35-40.
9. Haber RS. Pharmacologic management of pattern hair loss. Facial Plast Surg Clin North Am. 2004:181-189.
10. Shapiro J. Safety of topical minoxidil solution: a one-year, prospective, observational study. J Cutan Med Surg. 2003;7:322-329.
11. Arca E, Acikgoz G, Tastan HB, et al. An open, randomized, comparative study of oral Finasteride and 5% Topical Minoxidil in male androgenic alopecia. Dermatology. 2004;209:117-125.
12. Khandpur S, Suman M, Reddy BS. Comparative efficacy of various treatment regimens for androgenetic alopecia in men. J Dermatol. 2002;29:489-498.
13. www.drugstore.com. Accessed March 26, 2008.
14. www.rogaine.com/Mens/DrugFacts. Accessed June 12, 2008.
15. www.propecia.com/finasteride/propecia/consumer/product-information/pi. Accessed June 12, 2008.
16. Micromedex. Vol. 122.
Montvale, NJ: Thomson Healthcare; 1974-2008.
To comment on this article, contact
rdavidson@jobson.com.





