US Pharm. 2007;32(6):34-44.
Urinary incontinence (UI) is best defined as a variety of abnormalities of the urinary tract that result in the inability to experience normal micturition, leading to involuntary loss of bladder control. Approximately 40% of community-dwelling elderly and hospitalized patients are afflicted with this disorder.1-3 Sixty percent of nursing home patients experience this disorder, and it is often the reason for their admittance.4 Due to the social stigma associated with UI, the actual incidence in the community may be underestimated. Urinary incontinence may be associated with significant medical complications, creating issues for both patients and caregivers.1,5,6 The economic impact that results from UI influences both direct and indirect costs, placing a significant burden on the health care system.7,8
CAUSES OF URINARY INCONTINENCE
The causes of urinary incontinence are numerous and may involve age-related
changes in the bladder and urethra, including declines in outlet resistance in
females and prostate enlargement in males. Causes may also be secondary to a
variety of cerebral insults or diseases, including cerebrovascular disease
(strokes), spinal cord lesions, Alzheimer's disease, and Parkinson's disease.
Reversible causes are numerous and include urinary tract infections, stool
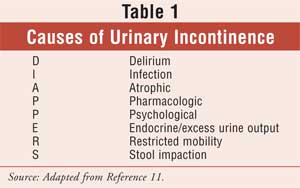
impaction, and numerous other triggers, described by the acronym DIAPPERS in
Table 1.9-14 Numerous medication and medication classes can
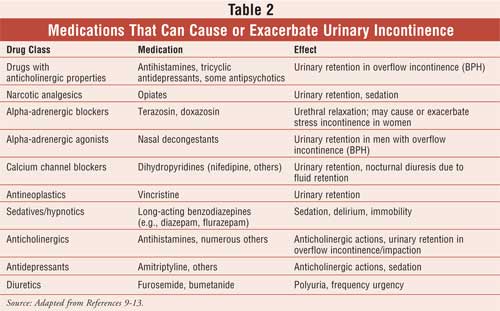
cause or exacerbate an existing incontinence and are described in Table 2
.12,13 Pharmacists should be aware of these potential disease–drug
interactions when monitoring their patients' drug therapy.
OVERVIEW OF THE ANATOMY AND PHYSIOLOGY OF THE
BLADDER
A basic understanding of the bladder's anatomy and pathophysiology is
important in order to understand the various UI types and their
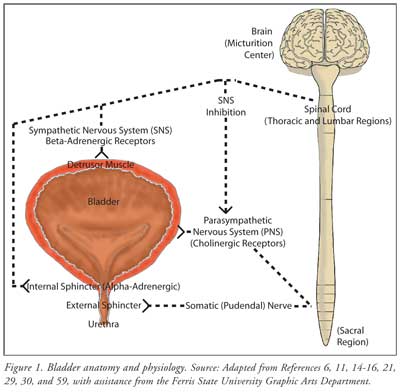
pharmacological management.11,14 Figure 1 describes the
anatomy and physiology associated with bladder function, including the
detrusor muscle in the bladder body and the internal and external sphincters,
along with their nervous system connections. The bladder fills when
sympathetic nervous system control results in a relaxed detrusor muscle and
closed sphincters at the bladder outlet. When the bladder reaches a certain
volume (200-400 mL), signals move from the spinal cord to brain centers,
resulting in the sensation of urge. During voiding, the parasympathetic
nervous system releases acetylcholine, which acts on the bladder detrusor
smooth muscle to create contractions, and, concurrently, the sympathetic
nervous system and somatic systems allow the sphincters to open. The result of
these complex processes is the release of urine (micturition) from the bladder.
14-18
DIAGNOSIS AND TREATMENT OVERVIEW
The evaluation and diagnosis of UI requires a complete medical examination to
rule out reversible causes and may require the participation of physicians
trained in urological evaluation.13 Correct diagnosis is important
since drugs utilized for one incontinence type may exacerbate other types.
18,19 From a pharmacist's perspective, a good understanding of the
medications used to treat incontinence and the medications (Table 2)
that may exacerbate this disorder are important.9-13 In addition,
since many urinary incontinent patients are elderly, drug use can be
challenging and requires an understanding of the pharmacokinetic and
pharmacodynamic changes that occur in this population.20
CLASSIFICATION OF INCONTINENCE
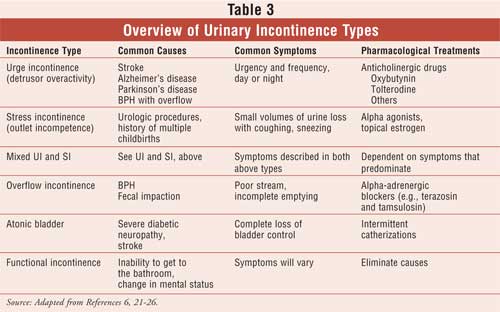
The various types of UI include those listed in Table 3, although many
patients may experience mixed etiologies. This article will provide an
overview of the three major types of UI and their treatments, including urge,
stress, and overflow types.6,13,21-26
URGE INCONTINENCE
Urge UI is the most common cause of incontinence in the elderly and is best
described by features of urgency that result in loss of urine. Urge
incontinence is often referred to as overactive bladder (OAB), although an
important distinction needs to be made from OAB since approximately two thirds
of patients with OAB are not incontinent of urine.13,27-30 The
pathophysiology is primarily due to detrusor muscle overactivity, resulting in
uncontrolled, uninhibited, or involuntary bladder muscle contractions.
30-32a Clinically, patients describe a sudden desire to urinate, which
is difficult to defer and results in leakage of urine occurring at various
times during the day and night.6,29,30 The causes may be secondary
to neurogenic insults including stroke, trauma, and neurologic diseases in
addition to reversible causes such as drugs and infections.6,9-13,26
The treatments for urge incontinence include bladder training, behavioral
treatments, pelvic floor exercises (Kegels), pads for temporary support,
surgery, and pharmacotherapy.33-35
Pharmacotherapy with Anticholinergic Drugs
The basis of pharmacotherapy for urge incontinence is controlling bladder
detrusor muscle overactivity with drugs that act as acetylcholine
(cholinergic) antagonists on bladder muscarinic (M) receptors, specifically
the M2 and M3. The result is a reduction in urgency symptoms and improvements
in bladder control.36 Since muscarinic receptors are also located
in other organ systems throughout the body and since these antimuscarinic
drugs are nonselective, numerous other effects and/or adverse effects may
result from their use.
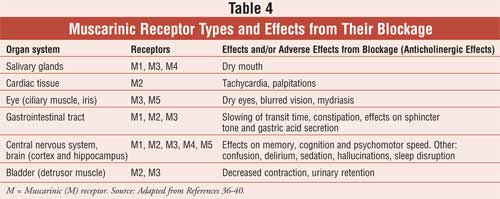
Anticholinergic or antimuscarinic side effects include both peripheral and central adverse reactions as noted in Table 4 . These adverse effects, including confusion, delirium, constipation, and urinary retention, are especially concerning in the more vulnerable elderly population.11,36,41,42 Contraindications to these agents are documented elsewhere and include narrow-angle glaucoma, urinary/gastric obstructive disorders, and dementia. Drug interactions include other anticholinergic drugs, acetylcholinesterase inhibitors, and numerous other agents. All of the antimuscarinic agents discussed below, except trospium, are metabolized by the cytochrome P-450 (CYP-450) system, and inhibitors of this system can potentiate their side-effect profiles.11,36 Monitoring for anticholinergic side effects, drug interactions, and potential contraindications, especially in patients who are taking multiple drugs with these properties, is essential for all health care professionals, including pharmacists.
A number of anticholinergic drugs are utilized in the treatment of urge incontinence. Older agents such as propantheline, dicyclomine, and flavoxate are still available but are rarely used because of their questionable efficacy and side-effect profiles. The tricyclic antidepressants (TCAs), including imipramine, have been used in urge incontinence and may have a role in mixed forms of stress and urge due to their duel antimuscarinic and alpha-adrenergic properties.36,43-47 The major agents used today in the treatment of urge incontinence include oxybutynin chloride (OBC), tolterodine (TD), and the three newest agents--trospium chloride (TC), darifenacin (DAF), and solifenacin (SFA)--along with a transdermal formulation of oxybutynin. Results from two large meta-analyses reported similar clinical efficacy among the available agents measured as reductions in episodes of urgency, frequency, daily micturitions, nocturnal awakenings, increase volume per void, and patient satisfaction. 44,48 Comparison trials with these agents are limited, however, and will be discussed below in a brief overview.49-51
Oxybutynin chloride: Oxybutynin chloride is the oldest of the agents presently used for urge incontinence and is available in regular-release and long-acting oral products, in addition to a patch formulation. Clinical trials indicate similar efficacy to other agents in the class, and the significance of OBC's proposed additional muscle relaxation properties is not clear.52 The oral extended-release formulation and patch formulations may offer improved tolerability, due to less formation of the active metabolite desethyloxybutynin (DES).53-57 The adverse effects include dose-related anticholinergic effects as described above, along with the addition of erythema and pruritus associated with the transdermal patch. Dry mouth with this agent is reported to be as high as 50% to 70% and may be due to greater binding to the parotid gland.52-57 Drug interactions include the expected additive side effects when used with other anticholinergic agents. In addition, CYP-450 2D6 and 3A4 pathway inhibitors (e.g., fluconazole and erythromycin) may potentiate its side-effect profile. Monitoring and reviewing patients' medication profiles are important when oxybutynin is prescribed with other agents.36,52,57
Tolterodine (Detrol, Detrol LA): Tolterodine is available in both regular-release tablets and long-acting products, and they may offer improved tolerability versus regular-release oxybutynin, with similar efficacy and tolerability to the other available agents.57,58 Tolterodine's bioavailability (BA) and elimination is dependent on the CYP-450 2D6 metabolism phenotype, with extensive metabolizers having a lower BA and less excretion of drug in the urine. Drug interactions are similar to those reported with oxybutynin, above.59-62
Newer Agents Released in 2004
The search for drugs with improved tolerability led to the development,
approval, and release of three new drugs in 2004.49-51 Although
these agents appear equal in efficacy to oxybutynin and tolterodine, they may
have some individual advantages in pharmacokinetic profile and/or
tolerability, although there is limited evidence to support a clinical
advantage.63-65
Trospium (Sanctura): The first of the new agents, trospium chloride, has a quaternary amine structure resulting in a hydrophilic molecule, with less blood–brain barrier penetration and a reduced potential for central nervous system (CNS) side effects. The BA of this agent is poor, and administration should be on an empty stomach. Hepatic metabolism is limited, resulting in lack of CYP-450 involvement and associated drug interactions. Elimination is via tubular secretion, requiring dose adjustments in patients with creatinine clearances (CrCl) less than 30 mL/minute.51,64,65 Trospium's efficacy is reported to be similar to other drugs in the class, although it may be better tolerated in some patients. 64,66-68 Drug interactions may involve competition for tubular secretion (metformin, digoxin) and drugs with an additive anticholinergic side-effect profile. Contraindications are similar to those of other anticholinergic agents as discussed above.51,69-71
Solifenacin (VESIcare): Solifenacin, another of the new antimuscarinics, is dosed once daily and has excellent BA. Solifenacin is metabolized primarily by CYP-450 3A4 and has some dependence on renal clearance, requiring adjustments in patients with a CrCl less than 30 mL/minute.50,72 Efficacy is reported to be similar to that of the other available anticholinergic agents,72-76 with one trial reporting fewer micturitions per 24 hours with solifenacin versus tolterodine.63 Adverse effects are similar to those of the other agents, along with reports of prolonged QTc intervals at higher doses, suggesting caution with patients at risk for this adverse event. As noted with oxybutynin and other drugs mentioned above, drug interactions, adverse effects, and contraindications are similar along with the added QTc concern. 50,77
Darifenacin (Enablex): Darifenacin, the third of the newer agents released in 2004, is also dosed once daily. Darifenacin's BA is poor and depends on the CYP-450 2D6 metabolism phenotype. Extensive metabolizers have a lower BA and more dependence on the CYP-450 3A4 pathway. Dosage adjustments are recommended in patients with hepatic impairment, and caution is suggested in patients with renal disease. 49,78
Darifenacin is reported to have a higher affinity for bladder M3 receptors, suggesting greater selectivity and tolerability, although clinical evidence of this advantage is lacking.78-80 Clinical trials with darifenacin have reported similar efficacy to the other agents, with improved tolerability versus oxybutynin.81-84 As noted with oxybutynin and the others above, metabolism involves the CYP-450 system, and adverse effects and contraindication are also similar.49
Summary
The anticholinergic/muscarinic drugs described above have a role in the
management of urge incontinence. Doses should be started low, especially in
elderly patients, and titrated slowly with careful monitoring for adverse
effects and drug interactions. An adequate trial of four to eight weeks is
recommended, with no clear advantage in terms of efficacy between the five
agents discussed, although tolerability differences may exist.47,48,81,82
Patient counseling when dispensing these agents should include a review of
potential side effects, drug interactions, and education on dosing and onset
of action.
STRESS INCONTINENCE
Stress incontinence, the most common form of UI in elderly women, is primarily
a problem with bladder sphincter function, resulting in urine leakage at
inappropriate times. Risk factors are numerous, including aging changes,
multiple childbirths, medications (Table 2), obesity, trauma, or
neurogenic problems.13,26,59,85-88 Although the economic costs are
high, the social costs and impact on elderly women are also significant.
89-91 Clinically, patients describe involuntary loss of urine triggered
by coughing, sneezing, or rising quickly. Patients with pure stress
incontinence may lack urgency and nocturia, although many of these patients
may have mixed forms of incontinence with features of urgency.87,88,92,93
The treatment of stress incontinence includes temporary pads for social
situations, behavioral interventions, and Kegel exercises, along with a number
of surgical options.37,94-99 There are limited agents and data
available for the pharmacological management of pure stress incontinence.
Unlabeled uses of alpha-adrenergic agonists (e.g., pseudoephedrine and
phenylephrine) are based on the urethral smooth-muscle response to alpha
stimulation, resulting in improved control of the internal sphincter and
reduced urine loss. Lack of proven efficacy with these agents and concerns
with adverse effects including insomnia, anxiety, hypertension, arrhythmias,
and stroke limits their utility.13,26,59,100-102 The tricyclic
antidepressant imipramine has been used in the treatment of stress or mixed
incontinence, due to its alpha-adrenergic agonist and anticholinergic
properties. Low doses may provide relief in some patients, especially if they
have a mixed disorder with stress and urgency symptoms. With its
anticholinergic side-effect profile, the use of this agent in elderly patients
may be problematic.11,26,36,85,86
Although the loss of estrogen in postmenopausal woman may contribute to symptoms of stress incontinence due to estrogen's influence on sphincter control, there is limited clinical evidence to support its use. Estrogen therapy has been proposed to increase urethral resistance via stimulation of estrogen receptors. If treatment is utilized, local or topical treatments are recommended and include creams (Table 3), vaginal tablets, or elastomer ring formulations, which may be beneficial for improving irritative symptoms and discomfort in some patients. 85,86,103-107 The use of oral estrogen for stress incontinence is not recommended due lack of benefit, along with the small but significant increased risk of other comorbidities.108
The most recent agent being evaluated for stress incontinence is duloxetine, a dual norepinephrine (NE) and serotonin (5HT) reuptake blocker approved for depression. The proposed mechanism in this disorder is through duloxetine's action on receptors in the Onuf's nucleus of the spinal cord and on the pudendal bladder nerve to improve urethral sphincter muscle contractions and tone.109
OVERFLOW INCONTINENCE
Overflow incontinence is best described as an overfilled bladder secondary to
obstruction, bladder prolapse, or alignment problems, most commonly seen in
males with benign prostatic hyperplasia (BPH).11,13,26,110 Men with
BPH are usually found to have an enlarged prostate on physical exam, and in
some cases further evaluation may be warranted to rule out prostate cancer or
other obstructions.110 The clinical presentation of overflow
incontinence is described by lower urinary tract symptoms including difficulty
starting urination and/or having a weak urine stream, a sense of incomplete
emptying, nocturia, and dribbling. A complicating feature of this disorder is
that two thirds of patients with BPH may also have urgency symptoms, which can
make diagnosis and treatment challenging.110,111 Treatment for
overflow incontinence caused by BPH may include watchful waiting and
eliminating potential triggers, including alcohol and/or caffeine. Surgical
options may include transurethral resection of the prostate (TURP) or newer,
less-invasive laser procedures.110,112-116
Drug therapy for the treatment of overflow incontinence secondary to BPH includes the peripheral alpha-adrenergic blockers and the 5-alpha-reductase inhibitors.110,112 The alpha-adrenergic blockers include the older, nonselective agents originally approved to treat hypertension and the newer prostate selective agents, which have minimal effects on blood pressure.117-119 The alpha-adrenergic blockers are usually the choice therapy in early mild disease due to their faster onset of action, usually within six weeks.118-121 Their mechanism of action is through their blocking action at the prostate alpha 1A receptor, resulting in improved urine outflow and relief of the symptoms described above.
These agents are metabolized by the hepatic CYP-450 system, and monitoring for drug interactions is necessary. Adverse effects include dizziness, peripheral edema, sedation, ejaculatory dysfunction, flulike symptoms, headaches, and gastrointestinal effects. Contraindications include heart failure, hypotension, and the potential to exacerbate stress incontinence in females. Individual differences between these agents include selectivity, dosing frequency, and cost issues related to brand name products. The selective agents may be better choices in patients without hypertension or in elderly patients at risk for orthostatic hypotension. The nonselective agents may be appropriate choices in younger BPH patients with concurrent hypertension. Patient counseling when dispensing these agents should include a review of potential side effects, especially effects on blood pressure, sedation, and dizziness.110,112,118
Alpha-Reductase Inhibitors
The alpha-reductase inhibitors finasteride and dutasteride are also used to
treat overflow incontinence secondary to BPH. Their mechanism of action is
through inhibiting the conversion of testosterone to dihydrotestosterone,
resulting in reduced androgenic prostate stimulation and leading to reduced
gland size and improved urine outflow. Although usually considered a
second-line therapy, they may be used first-line in patients with
contraindications to the alpha-blockers (hypotension or heart failure) or in
combination with alpha-blockers in progressive or more moderate-to-severe
disease, e.g., large glands. Although effective in treating the symptoms of
BPH, their side-effect profile (e.g., decreased libido, impotence, dry sex,
and gynecomastia) and slower onset of action, which may take up to six to 12
months, limits their use in mild disease.110,112,118,122-124
Similar efficacy for both agents has been reported in clinical trials, and
potential differences in selectivity for reductase enzymes have not been
demonstrated clinically.125 Although these agents are not indicated
in women, they are classified as pregnancy category X, and avoidance of drug
contact by any potential route (handling broken tablets, semen transfer) with
pregnant women or those seeking to become pregnant is recommended.
110,112,118
Other therapies that have been utilized in the management of overflow incontinence secondary to BPH include the herbal saw palmetto, which is reported to have 5-alpha-reductase inhibitor activity and the cholinergic agonist bethanechol, which may be used for acute management after TURP procedures11,36 Investigational therapies include combinations of anticholinergic drugs with alpha-blockers for patients who have urgency associated with their BPH symptoms.111,126 The use of the erectile dysfunction drugs (e.g., phosphodiesterase inhibitors [sildenafil] and botulinum toxin) are also being evaluated in the treatment of overflow incontinence secondary to BPH.127,128 As noted in Table 3, other types of incontinence include functional incontinence secondary to patients' inability to reach the bathroom due to a physical disability and atonic bladder, where complete loss of bladder innervation and control secondary to various diseases or insults, including stroke or diabetes, requires the use of intermittent catheterizations. Mixed UI may occur in up to 20% to 30% of patients and, due to the various clinical presentations and symptoms, may require extensive evaluation and therapeutic trials to determine appropriate therapies.6,11,13,18,24-26,59,88
CONCLUSION
The pharmacological management of UI requires appropriate evaluation by
qualified clinicians. Pharmacists can offer educational support to patients by
questioning and monitoring the effectiveness and tolerability of the various
pharmacotherapies. Taking time to get to know your patients in the ambulatory
and clinical settings allows the pharmacist the opportunity to provide
valuable education, intervention, and recommendations to improve patient care
outcomes in the management of this complex disorder.
References
1. Wilson MM. Urinary incontinence: selected current concepts. Med Clin
North Am. 2006;90:825-836.
2. Zarowitz BJ, Ouslander JG. Management of urinary incontinence in older
persons. Geriatr Nurs. 2006:265-270.
3. Chutka DS, Takalhashi PY. Urinary incontinence in the elderly. Drugs
. 1998;56:587-595.
4. Tannenbaum C, DuBeau CE. Urinary incontinence in the nursing home:
practical approach to evaluation and management. Clin Geriatr Med.
2004;20:437-452.
5. American Medical Directors Association (AMDA) Clinical Practice Guideline:
Urinary Incontinence, 2005. Available at:
www.amda.com/info/cpg/incontinence.htm.
6. AHCPR Urinary Incontinence in Adults Guideline Update Panel. Clinical
practice guidelines: managing acute and chronic urinary incontinence. Am
Family Physician. 1996;54:1661-1671.
7. Levy R, Muller N. Urinary incontinence: economic burden and new choices in
pharmaceutical treatment. Adv Ther. 2006;23:556-573.
8. Wilson L, Brown JS, Shin GP, et al. Annual direct cost of urinary
incontinence. Obstet Gynecol. 2001;98:398-406.
9. Thom DH, van den Eeden SK, Brown JS. Evaluation of parturition and other
reproductive variables as risk factors for urinary incontinence in later life.
Obstet Gynecol. 1997;90:983-989.
10. Thom DH, Brown JS. Reproductive and hormonal risk factors for urinary
incontinence in later life: a review of the clinical and epidemiologic
literature. J Am Geriatri Soc. 1998;46:1411-1417.
11. Thompson JF. Geriatric urological disorders. In: Koda Kimble MA, Young LL,
eds. Applied Therapeutics: The Clinical Use of Drugs. 8th ed.
Vancouver, WA: Applied Therapeutics, Inc; 2005:101-1–101-33.
12. Rondorf-Klyn LM, Colling J, Simonson W. Medication use by
community-dwelling elderly with urinary incontinence. Urol Nurs.
1998;18:201-206.
13. Abrams P, Andersson KE, Brubaker L, et al; 3rd International Consultation
on Incontinence. Recommendations of the International Scientific Committee.
Evaluation and treatment of urinary incontinence, pelvic organ prolapse, and
faecal incontinence. In: Abrams P, Cardozo L, Khoury S, Wein A, eds.
Incontinence. Vol 2. Plymouth, UK: Health Publication Ltd; 2005:1589-1626.
14. Gosing, JA. The structure of the female lower urinary tract and pelvic
floor. Urol Clin North Am. 1985;12:207.
15. Abrams P. Describing bladder storage function: overactive bladder syndrome
and detrusor overactivity. Urology. 2003;62:28-37.
16. Elbadawi A, Diokno A, Millard R. The aging bladder: morphology and
urodynamics. World J Urol. 1998;16(suppl 1):S10-S34.
17. Chapple CR, Artibani W, Cardozo LD, et al. The role of urinary urgency and
its measurement in the overactive bladder symptom syndrome: current concepts
and future prospects. BJU Int. 2005;95:335-340.
18. Tannenbaum C, Perrin L, DuBeau C, et al. Diagnosis and management of
urinary incontinence in the older patient. Arch Phys Med Rehabil.
2001;82:134-138.
19. Scientific Committee of the First International Consultation on
Incontinence. Assessment and treatment of urinary incontinence. Lancet.
2000;355:2153-2158.
20. Linnebur SA, O'Connell MB, Wessell AM, et al. Pharmacy practice, research,
education, and advocacy for older adults. Pharmacotherapy.
2005;25:1396-1430.
21. Anderson KE, Hedlund P. Pharmacologic perspective on the physiology of the
lower urinary tract. Urology. 2002;605(suppl 1):13-21.
22.Yim PS, Peterson AS. Urinary incontinence. Postgrad Med.
1996;99:137-150.
23. Sandvik H, Hunskaar S, Vanvik A, et al. Diagnostic classification of
female urinary incontinence: an epidemiological survey corrected for validity.
J Clin Epidemiol. 1995;48:339-343.
24. Bump RC, Norton PA, Zinner NR, et al. Mixed urinary incontinence in women
with predominant stress urinary incontinence symptoms: urodynamic findings,
incontinence severity measures, and duloxetine treatment response. Obstet
Gynecol. 2003;102:76-83.
25. Viktrup L. Addressing the need for a simpler algorithm for the management
of women with urinary incontinence. Medscape General Medicine. 2005;7:3.
Available at: http://www.medscape.com/viewarticle/506898.
26. National Library of Medicine. Urinary incontinence in adults: acute and
chronic management. clinical practice guideline number 2 (1996 Update) AHCPR
Publication No. 96-0682: March 1996. Available at:
www.ncbi.nlm.nih.gov/books/bv.fcgi?rid=hstat6.chapter.9995.
27. Milsom L, Abrams P, Cardozo L, et al. How widespread are the symptoms of
an overactive bladder and how are they managed? A population-based prevalence
study. BJU Int. 2001;87:760-766.
28. Stewart WF, Van Rooyen JB, Cundiff GW, et al. Prevalence and burden of
overactive bladder in the United States. World J Urol. 2003;20:327-336.
29. Abrams P, Cardozo L, Fall M, et al. The standardization of terminology in
lower urinary tract function: report from the Standardization Sub-committee of
the International Continence Society. Am J Obstet Gynecol.
2002;187:116-126.
30. Wein AJ, Rovner ES. Definition and epidemiology of overactive bladder.
Urology. 2002;60(suppl 5A):7-12.
31. Payne CK. Epidemiology, pathophysiology, and evaluation of urinary
incontinence and overactive bladder. Urology. 1998;51(suppl 2A):3-10.
32. Fourcroy JL. Urogynecology update: incontinence. Hospital Practice.
1998;33:63-81.
32a. Resnick NM, Blaivas JG, Ostergard DR. Practical pointers on urinary
incontinence. Patient Care. 1995;29:103-131.
33.Burgio KL, Locker JL, Goode PS, et al. Behavioral vs drug treatment for
urge incontinence in older women: a randomized controlled trial. JAMA.
1998;280:2034-2035.
34.Wyman JE, Fantl JA. Bladder training in ambulatory care management of
urinary incontinence. Urol Nurs. 1991;11:11-17.
35. Wyman JF, Fantl JA, McClish DK, et al. Comparative efficacy of behavioral
interventions in the management of female urinary incontinence. Continence
Program for Women Research Group. Am J Obstet Gynecol.
1998;179:999-1007.
36. Brown JH, Palmer T. Muscarinic receptor agonist and antagonists. In:
Goodman & Gilman's The Pharmacolgical Basis of Therapeutics
. 9th ed. New York, NY: McGraw-Hill; 1996:141-160.
37. Goode PS, Burgio KL. Pharmacological treatment of lower urinary
tract dysfunction in geriatric patients. Am J Med Sci. 1997;314:262-267.
38. Yasuhiko I. Discussion: functional role of M1, M2 and M3 muscarinic
receptors in overactive bladder. Urology. 2000;55:47-49.
39. Caulfield MP, Birdsall NJ. International Union of Pharmacology. XVII.
Classification of muscarinic acetylcholine receptors. Pharmacol Rev.
1998;50:279-290.
40. Andersson KE. Antimuscarinics for treatment of overactive bladder.
Lancet Neurol. 2004;3:46-53.
41. Moore AR, O'Keeffe ST. Drug-induced cognitive impairment in the elderly.
Drugs Aging. 1999;15:15-28.
42. Power AE, McIntyre CK, Litmanovich A, et al. Cholinergic modulation of
memory in the basolateral amygdala involves activation of both M1 and M2
receptors. Behav Pharmacol. 2003;14:207-213.
43. Herbison P, Hay-Smith J, Blis G, et al. Effectiveness of anticholinergic
drugs compared with placebo in the treatment of overactive bladder: systemic
review. BMJ. 2003;326:8414.
44. Andersson KE. Antimuscarinics for treatment of overactive bladder.
Lancet Neurol. 2004;3:46-53.
45. Fischer CP, Diokno A, Lapides J. The anticholinergic effects of
dicyclomine HCl in uninhibited neurogenic bladder dysfunction. J Urol.
1978;120:328-329.
46. Briggs RS, Castleden CM, Asher MJ. The effect of flavoxate on uninhibited
detrusor contractions and urinary incontinence in the elderly. J Urol.
1980;123:665-666.
47. Chapple CR. Muscarinic receptors antagonists in the treatment of
overactive bladder. Urology. 2000;55(suppl 5A):33-46.
48. Chapple C, Khullar V, Gabriel Z, Dooley JA. The effects of antimuscarinic
treatments in overactive bladder: a systematic review and meta-analysis.
Eur Urol. 2005;48:5-26.
49. Enablex (darifenacin). East Hanover, NJ: Novartis Pharmaceuticals
Corporation; 2004.
50. Vesicare (solifenacin) tablets. Yamanouchi Pharma America, Inc., and
GlaxoSmithKline; 2004.
51. Sanctura (trospium chloride) tablets. Lexington, MA: Indevus
Pharmaceuticals, Inc.; 2004.
52. Yarker Y, Goa KL, Fitton A. Oxybutynin: a review of its pharmacodynamic
and pharmacokinetic properties, and its therapeutic use in detrusor
instability. Drugs Aging. 1995;6:243-262.
53. Waldeck K, Larsson B, Andersson KE. Comparison of oxybutynin and its
active metabolite, N-desethyl-oxybutynin in the human detrusor and
parotid gland. J Urol. 1997;157:1093-1097.
54. Diokno AC, Appell RA, Sand PK, et al. Prospective, randomized,
double-blind study of the efficacy and tolerability of the extended-release
formulations of oxybutynin and tolterodine for overactive bladder: results of
the OPERA trial. Mayo Clin Proc. 2003;78:687-695.
55. Davila GW, Daugherty CA, Sanders SW. A short-term, multicenter, randomized
double-blind dose titration study of the efficacy and anticholinergic side
effects of transdermal compared to immediate release oral oxybutynin treatment
of patients with urge urinary incontinence. J Urol. 2001;166:140-145.
56. Barkin J, Corcos J, Radomski S, et al. A randomized, double-blind,
parallel-group comparison of controlled- and immediate- release oxybutynin
chloride in urge urinary incontinence. Clin Ther. 2004;26:1026-1036.
57. Dmochowski RR, Sand PK, Zinner NR, et al., for the Transdermal Oxybutynin
Study Group. Comparative efficacy and safety of transdermal oxybutynin and
oral tolterodine versus placebo in previously treated patients with urge and
mixed urinary incontinence. Urology. 2003;62:237-242.
58. Abrams P, Freeman R, Anderstrom C, et al. Tolterodine, a new
antimuscarinic agent: as effective but better tolerated than oxybutynin in
patients with an overactive bladder. Br J Urol. 1998;81:801-810.
59. Andersson K-E, Appell R, Cardozo L, et al. 3rd International Consultation
on Incontinence. Pharmacological treatment of urinary incontinence. In: Abrams
P, Cardozo L, Khoury S, Wein A, eds. Incontinence. Vol 2. Plymouth, UK:
Health Publication Ltd.; 2005:809-855.
60. Guay D. Clinical pharmacokinetics of drugs used to treat urge
incontinence. Clin Pharmacokinetics. 2003;42:1243-1285.
61. Nilvebrant L, Andersson KE, Gillberg PG, et al. Tolterodine: a new
bladder-selective antimuscarinic agent. Eur J Pharmacol.
1997;327:195-207.
62. Nilvebrant L, Glas G, Jonsson A, et al. The in vitro pharmacological
profile of tolterodine: a new drug for the treatment of urinary incontinence.
Neurourol Urodyn. 1994;13:433-435.
63. Chapple C, Rechberger T, Al-Shukri S, et al. Randomized, double-blind
placebo- and tolterodine-controlled trial of the once-daily antimuscarinic
agent solifenacin in patients with symptomatic overactive bladder. BJU Int
. 2004;93:303-310.
64. Pak RW, Petrou SP, Staskin DR. Trospium chloride: a quaternary amine with
unique pharmacologic properties. Curr Urol Rep. 2003;4:436-440.
65. Haab F, Stewart L, Dwyer P. Darifenacin, an M3 selective
receptor antagonist, is an effective and well-tolerated once-daily treatment
for overactive bladder. Eur Urol. 2004;45:420-429.
66. Todorova A, Vonderheid-Guth B, Dimpfel W. Effects of tolterodine, trospium
chloride, and oxybutynin on the central nervous system. J Clin Pharmacol
. 2001;41:636-644.
67. Pietzko A, Dimpfel W, Schwantes U, Topfmeier P. Influences of trospium
chloride and oxybutynin on quantitative EEG in healthy volunteers. Eur J
Clinic Pharmacol. 1994;47:337-343.
68. Madersbacher H, Stöhrer, Richter R, et al. Trospium chloride versus
oxybutynin: a randomized, double-blind, multicentre trial in the treatment of
detrusor hyper-reflexia. Br J Urol. 1995;75:452-456.
69. Beckmann-Knopp S, Rietbrock S, Weyenmeyer R, et al. Inhibitory effects of
trospium chloride on cytochrome P540 enzymes in human liver microsomes.
Pharmacol Toxicol. 1999;85:299-304.
70. Rovner ES. Trospium chloride in the management of overactive bladder.
Drugs. 2004;64:2433-2446.
71. Fusgen I, Hauri D. Trospium chloride: an effective option for medical
treatment of bladder overactivity. Int J Clin Pharmacol Ther.
2000;38:223-234.
72. Brunton S, Kuritzky L. Recent developments in the management of overactive
bladder: focus on the efficacy and tolerability of once daily solifenacin
succinate 5 mg. Curr Med Res Opin. 2005;21:71-80.
73. Chapple CR, Martinez-Garcia R, Selvaggi L, et al. A comparison of the
efficacy and tolerability of solifenacin succinate and extended release
tolterodine at treating overactive bladder syndrome; results of the STAR
trial. Eur Urol. 2005;48:464-470.
74. Chapple C, Wyndaele JJ, Gronen S, for the Solifenacin Study Group.
Solifenacin provided statistically significant and clinically relevant
reductions in urgency, a defining symptom of overactive bladder. Neurourol
Urodyn. 2004;23:316.
75. Haab F, Cardozo L, Chapple C, Ridder AM, for the Solifenacin Study Group.
Long-term open-label solifenacin treatment associated with persistence with
therapy in patients with overactive bladder syndrome. Eur Urol.
2005;47:376-384.
76. Cardozo L, Lisec M, Millard R, et al. Randomized, double-blind
placebo-controlled trial of the once-daily antimuscarinic agent solifenacin
succinate in patients with overactive bladder. J Urol.
2004;172:1919-1924.
77. Ikeda K, Kobayashi S, Suzuki M, et al. M(3) receptor antagonist by the
novel antimuscarinic agent solifenacin in the urinary bladder and salivary
gland. Arch Pharmacol. 2002;366:97-103.
78. Chapple CR. Darifenacin: a novel M3 muscarinic selective receptor
antagonist for the treatment of overactive bladder. Expert Opin Invest Drugs
. 2004;13:1493-1500.
79. Newgreen DT, Anderson DW, Carter AJ. Darifenacin: a novel
bladder-selective agent for the treatment of urge incontinence. Neurourol
Urodyn. 1995;14:95-99.
80. Braverman AS, Ruggieri MR, Pontari MA. The M2 muscarinic receptor subtype
medicates cholinergic bladder contractions in patients with neurogenic bladder
dysfunction. J Urol. 2001;165:36.
81. Haab F, Stewart L, Dwyer P. Darifenacin. an M3 selective receptor
antagonist, is an effective and well-tolerated once-daily treatment for
overactive bladder. Eur Urol. 2004;45:420-429.
82. Foote JE. Darifenacin. A highly M3 selective muscarinic receptor
antagonist, is effective and well tolerated by elderly patients with
overactive bladder. J Am Geriatr Soc. 2004;52:S126.
83. Zinner N, Tuttle JB, Marks L. Efficacy and tolerability of darifenacin, a
muscarinic M3 selective receptor antagonist, compared with oxybutynin in the
treatment of patients with overactive bladder. International Continence
Society Meeting; 2004. Paris, France.
84. Khullar V. Darifenacin, an M3 selective receptor antagonist, reduces the
frequency of nocturnal awaking, and important symptom of overactive bladder.
J Urol. 2004;171(suppl 4):131. Abstract 491.
85. Mushkat Y, Bukovsky I, Langer R. Female urinary stress incontinence--does
it have familial prevalence? Am J Obstet Gynecol. 1996;174:617-619.
86. Brown JS, Grady D, Ouslander JG, et al. Prevalence of urinary incontinence
and associated risk factors in postmenopausal women. Heart &
Estrogen/Progestin Replacement Study (HERS) Research Group. Obstet
Gynecol. 1999;94:66-70.
87. Viktrup L, Koke S, Burgio KL, Ouslander JG. Stress urinary incontinence in
active elderly women. South Med J. 2005;98:79-89.
88. Urinary Incontinence in Women. Bethesda, Md: National Kidney and
Urologic Diseases Clearinghouse. NIH Publication No. 02-4132. May 2002.
Available at: http://kidney.niddk.nih.gov/kudiseases/pubs/uiwomen/index.htm.
89. Wilson L, Brown JS, Shin GP, et al. Annual direct cost of urinary
incontinence. Obstet Gynecol. 2001;98:398-406.
90. Dugan E, Cohen S, Bland DR, et al. The association of depressive symptoms
and urinary incontinence among older adults. J Am Geriatr Soc.
2000;48:413-416.
91. Melville JL, Walker E, Katon W, et al. Prevalence of comorbid psychiatric
illness and its impact on symptom perception, quality of life, and functional
status in women with urinary incontinence. Am J Obstet Gynecol.
2002;187:80-87.
92. Campbell SC, Siegel SW. Female urinary incontinence, current evaluation
and management. Consultant. 1992;32:45-52.
93. Artibani W, Andersen JT, Gajewski JB, et al. Imaging and other
investigations. In: Abrams P, Cardozo L, Khoury S, Wein A, eds. Incontinence
. 2nd ed. Plymouth, UK: Health Publication Ltd.; 2002:425-477.
94. Wilson PD, Bo K, Hay-Smith J, et al. Conservative treatment in women. In:
Abrams P, Cardozo L, Khoury S, Wein A, eds. Incontinence. 2nd ed.
Plymouth, UK: Health Publication Ltd.; 2002:571-624.
95. American Academy of Family Physicians. Urinary incontinence: Kegel
exercises for your pelvic muscles. Information from your family doctor.
Available at: http://familydoctor.org/handouts/642.html.
96. Bo K, Talseth T, Holme I. Single blind, randomised controlled trial of
pelvic floor exercises, electrical stimulation, vaginal cones, and no
treatment in management of genuine stress incontinence in women. BMJ.
1999;318:487-493.
97. Ulmsten U, Falconer C, Johnson P, et al. A multicenter study of
tension-free vaginal tape (TVT) for surgical treatment of stress urinary
incontinence. Int Urogynecol J Pelvic Floor Dysfunct. 1998;9:210-213.
98. Weber AM, Walters MD. Burch procedure compared with sling for stress
urinary incontinence. A decision analysis. Obstet Gynecol.
2000;96:867-873.
99. Hilton P. Trials of surgery for stress incontinence--thoughts on the
‘Humpty Dumpty principle.' BJOG. 2002;109:1081-1088.
100. Hoffman BB, Lefkowitz RJ. Catecholamines, sympathomimetic drugs and
adrenergic receptor antagonists. In: Goodman & Gilman's The
Pharmacolgical Basis of Therapeutics. 9th ed. New York, NY: McGraw-Hill;
1996:199-248.
101. Collset L, Lindskog M. Phenylpropanolamine in the treatment of female
stress urinary incontinence: double-blind placebocontrolled study in 24
patients. Urology. 1987;30:398-403.
102. Stier BG, Hennekens CH. Phenylpropanolamine and hemorrhagic stroke in the
Hemorrhagic Stroke Project: a reappraisal in the context of science, the Food
and Drug Administration, and the law. Ann Epidemiol. 2006;16:49-52.
103. Maloney C. Estrogen in urinary incontinence treatment: an anatomic and
physiologic approach. Urol Nurs. 1997;17:88-91.
104. Bernier F, Jenkins P. The role of vaginal estrogen in the treatment of
urogenital dysfunction in postmenopausal women. Urol Nurs.
1997;17:92-95.
105. Sartori MG, Baracat EC, Girao MJ, et al. Menopausal genuine stress
incontinence treated with conjugated estrogens plus progestogens. Int J
Gynaecol Obstet. 1995;49:165-169.
106. Makinen JI, Pitkanen YA, Salmi TA, et al. Transdermal estrogens for
female stress urinary incontinence in postmenopause. Maturitas.
1995;22:233-238.
107. Griebling TL, Nygaard IE. The role of estrogen replacement therapy in the
management of urinary incontinence and urinary tract infection in
postmenopausal women. Endocrinol Metab Clin North Am. 1997;26:347-360.
108. Hendrix SL, Cochrane BB, Nygaard IE, et al. Effects of estrogen with and
without progestin on urinary incontinence. JAMA. 2005;293:935-948.
109. Millard RJ, Moore K, Rencken R, et al. Duloxetine vs placebo in the
treatment of stress urinary incontinence: a four-continent randomized clinical
trial. BJU Int. 2004;93:311-318.
110. Lee ML. Management of benign prostatic hyperplasia. In: DiPiro JT, ed.
Pharmacotherapy: A Pathophysiologic Approach. 6th ed. New York, NY:
McGraw-Hill; 2005:1535-1546.
111. Macdiarmid S, Chen A, Tu N, et al. Effects of tamsulosin and
extended-release oxybutynin on lower urinary tract symptoms in men. Program
and abstracts of the American Urological Association 2006 Annual Meeting; May
20-25, 2006; Atlanta, Ga. Abstract 1638.
112. Walsh PC. Treatment of benign prostate hyperplasia. N Engl J Med.
1996;335;586-587.
113. Gnanapragasam VJ, Kumar V, Langton D, et al. Outcome of transurethral
prostatectomy for the palliative management of lower urinary tract symptoms in
men with prostate cancer. Int J Urol. 2006;13:711-715.
114. Mattiasson A, Wagell L, Schelin S, et al. Five-year follow-up of
feedback microwave thermotherapy versus TURP for clinical BPH: a prospective
randomized multicenter study. Urology. 2007;69:91-97.
115. Fried NM. New laser treatment approaches for benign prostatic
hyperplasia. Curr Urol Rep. 2007;8:47-52.
116. Harris JL. Treatment of postprostatectomy urinary incontinence with
behavioral methods. Clin Nurse Spec. 1997;11:159-166.
117. Saseen JJ, Carter BL. Hypertension. In: Dipiro JT.
Pharmacotherapy: A Pathophysiologic Approach. 6th ed. Connecticut:
McGraw-Hill; 2005:18-217.
118. American Urological Association Practice Guidelines Committee. AUA
guidelines on management of benign prostatic hyperplasia (2003). Chap 1:
Diagnosis and treatment recommendations. J Urol. 2003;170:530-547.
119. Oades GM, Eaton JD, Kirby RS. The clinical role of alpha-blockers in the
treatment of benign prostatic hyperplasia. Curr Urol Rep.
2000;1:97-102.
120. Noble AJ, Chess-Williams R, Couldwell C, et al. The effects of
tamsulosin, a high affinity antagonist at functional alpha 1A and alpha
1D-adrenoceptor subtypes. Br J Pharmacol. 1997;120:231-238.
121. MacDonald R, Wilt TJ. Alfuzosin for treatment of lower urinary tract
symptoms compatible with benign prostatic hyperplasia: a systematic review of
efficacy and adverse effects. Urology. 2005;66:780-788.
122. Anderson JT, Nickel JC, Marshall VR, et al. Finasteride significantly
reduces acute urinary retention and need for surgery in patients with
symptomatic benign prostatic hyperplasia. Urology. 1997;49:839-845.
123. Lepor H, Willford WO, Barry MJ, et al. The efficacy terazosin,
finasteride, or both in benign prostate hyperplasia. N Engl J Med.
1996;335:533-540.
124. Sandhu JS, Te AE. The role of 5-alpha-reductase inhibition as monotherapy
in view of the MTOPS data. Curr Urol Rep. 2004;5:274-279.
125. Nickel JC. Comparision of clinical trials with finasteride and
dutasteride. Rev Urol. 2004;6(suppl 9):S31-S39.
126. Yang Y, Zhao XF, Li HZ, et al. Efficacy and safety of combined therapy
with terazosin and tolteradine for patients with lower urinary tract symptoms
associated with benign prostatic hyperplasia: a prospective study. Chin Med
J. 2007;120:370-374.
127. Hopps CV, Mulhall J. Assessment of the impact of sildenafil citrate on
lower urinary tract symptoms (LUTS) in men with erectile dysfunction (ED).
J Urol. 2003;169:375A.
128. Chuang YC, Chiang P, Hsien K, et al. Beneficial effects of up to one year
with intraprostatic botulinum toxin type A injection on LUTS and quality of
life in BPH patients. Program and abstracts of the American Urological
Association 2006 Annual Meeting; May 20-25, 2006; Atlanta, Ga. Abstract 1436.
To comment on this article, contact editor@uspharmacist.com.





