US Pharm. 2020;45(7/8):HS2-HS8.
ABSTRACT: Vaping has become an increasing problem because of the associated lung injury. More middle school and high school students are using electronic cigarettes (e-cigarettes), which has led to adverse events in this population. Vitamin E acetate, a thickening agent used in some e-cigarettes, has been identified as a potential cause of associated lung injury. Bronchoalveolar-lavage samples have revealed the presence of tetrahydrocannabinol and nicotine in patients presenting with lung injury. Patients often present with respiratory, gastrointestinal, and constitutional symptoms. The management of e-cigarette, or vaping, product use–associated lung injury includes corticosteroids and antimicrobial coverage for community-acquired pneumonia. Pharmacists should recommend vaping and tobacco cessation, as well as influenza and pneumococcal vaccinations, to patients who use e-cigarettes.
Vaping has increased in popularity, particularly among middle school– and high school–aged students, since its entrance into the U.S. consumer market. Consequently, the incidence of vaping-associated lung injury has become more common. Many inhaled environmental agents have been known to cause acute and subacute parenchymal lung injuries. The severity of symptoms secondary to inhalation of these chemical agents is often correlated with the type and amount of the agent.¹ Manifestations of parenchymal lung injury range from respiratory tract discomfort to respiratory failure and subsequent death. Acute chemical injuries to the lungs share a common pathophysiologic pathway that begins with acute inflammation leading to airway edema with epithelial sloughing, alveolar edema, and, eventually, hypoxia.² The emergence of electronic cigarette (e-cigarette) use has added to the list of common causes of acute lung disease.
E-cigarettes deliver the active ingredient to the lungs by heating a liquid vehicle to produce an aerosol, which is then inhaled. The liquid vehicle may contain a number of substances, including tetrahydrocannabinol (THC), cannabinoid, nicotine, flavorings, and other additives.3
Public policy regarding the safety of e-cigarettes has lagged behind the rapid rise in use, which has led to an increased incidence of adverse effects in adolescents. As of November 5, 2019, there were 2,051 cases of e-cigarette, or vaping, product use–associated lung injury (EVALI) reported across 49 states, and 39 deaths were confirmed among these cases. The rapid spike in EVALI cases prompted an investigation by the CDC and the FDA to determine the risk factors and source of this lung disease.3
In this investigation, an examination of bronchoalveolar lavage (BAL) fluid samples of 29 patients presenting with EVALI detected the presence of vitamin E acetate, a common thickening agent for the solutions contained in e-cigarettes. Furthermore, THC and nicotine were found in 82% and 62% of samples, respectively. The finding of a correlation between vitamin E acetate and subsequent lung injury constitutes the first time a potential causative agent of concern has been identified in patients presenting with EVALI. Although vitamin E acetate is an agent of concern, evidence remains insufficient to rule out the contribution of other chemicals that may lead to EVALI.³ Other potential causative substances and product sources are being investigated.
Epidemiology
Since the introduction of e-cigarettes to the U.S. consumer market in 2007, the use of these products has escalated in high school–aged adolescents, with a reported rate of 20.8% in 2018.4,5 The use of e-cigarettes among U.S. adults is significantly lower than in the younger population. In 2018, only 3.2% of adults reported the use of e-cigarettes, but those aged 18 to 24 years had an increased prevalence of 7.6%, and those aged 25 to 34 years of age had a rate of 5.4%.6
As of September 24, 2019, the CDC had received 805 case reports of suspected e-cigarette–induced lung disease. The CDC, FDA, and state and local health departments are investigating this outbreak and assessing the available information to better understand who is at risk. An analysis of the reports found that 69% of patients were male, the median age was 23 years, and 16.2% of patients were younger than 18 years. Among these cases, there have been 12 confirmed deaths spanning 10 states. Information regarding the inhaled substance was given in 514 case reports; 76.9% of cases involved THC-containing products, 56.8% involved nicotine-containing products, 36% involved THC-containing products exclusively, and 16.0% involved nicotine-containing products exclusively.7 Additionally, 771 cases (96%) reported basic patient data, such as demographics, dates of symptom onset, and hospitalization. Ninety-one percent of patients were hospitalized. The median duration of symptom onset before hospitalization was 6 days, with a range of 0 to 158 days.7
The causative agent leading to the current outbreak of EVALI remains unknown. However, the most recent national data indicate that the majority of lung-injury patients used THC-containing products with or without nicotine-containing products. It is important to note that some EVALI patients report the use of nicotine-containing products exclusively. Regardless, vaping products should never be used by adolescents, young adults, and pregnant women. Adults who previously did not use tobacco products should also avoid the use of e-cigarettes.7
Clinical Presentation
Although the causes of EVALI remain unknown, case reports from 49 state health departments have aided the CDC in supplying healthcare providers with interim guidance for the evaluation and care of patients with suspected EVALI. The interim guidance includes the initial clinical evaluation; criteria for hospital admission and treatment; treatment follow-up; considerations for patients at high risk; and clinical and public-health recommendations.8
Data obtained from case reports and medical charts revealed that 95% of patients with a diagnosis of EVALI presented with respiratory symptoms, 77% presented with gastrointestinal symptoms, and 85% presented with constitutional symptoms. Respiratory symptoms most frequently manifested as chest pain and shortness of breath. Common gastrointestinal symptoms included abdominal pain, nausea, vomiting, and diarrhea. The most common constitutional symptoms were fever, chills, and weight loss.8
In addition to the subjective symptoms typically seen in patients with EVALI, distinct objective criteria in the form of vital signs, laboratory measurements, and imaging have also been found to occur in the majority of cases. Patients most often presented with hypoxemia, tachypnea, tachycardia, leukocytosis, elevated inflammatory markers, and abdominal computed tomography positive for bilateral ground-glass opacities in the lung bases.9
Treatment
The backbone of EVALI management is the use of corticosteroids and antimicrobial coverage for community-acquired pneumonia. The provider should assess the patient’s oxygenation status, encourage cessation of vaping and tobacco use, evaluate and treat for pneumonia if appropriate, and closely follow the patient during and after the treatment course. Hospital admission of patients presenting with suspected EVALI is at the discretion of the provider; however, the CDC has issued interim guidance to aid the provider with the decision.8
The CDC advises providers to strongly consider admission to a medical ward or ICU when a patient presents with respiratory distress, has comorbidities that interfere with pulmonary reserve, or has an oxygen-saturation level of less than 95%.8 Patients treated in the medical ward should receive empirical antibiotics targeted toward community-acquired pneumonia and should also undergo a short course of corticosteroids. An appropriate corticosteroid regimen may include IV methylprednisolone 1 mg/kg for 1 to 2 days followed by oral prednisone, depending on the clinical course. Clinical improvement should be expected within 1 to 3 days. Close attention should be paid to proper weaning of corticosteroids during the course of inpatient treatment, as patients have been found to relapse after a rapid wean. The patient should be monitored for secondary infectious complications and the development of a pneumothorax and/or pneumomediastinum. If there is no improvement or the patient’s condition worsens during the treatment course, the provider should reconsider the differential diagnosis and order a bronchoscopy with BAL. Postdischarge follow-up includes reassessment of symptoms and continued e-cigarette use, oxygenation status, pulmonary-function tests, and a chest x-ray within 4 to 6 weeks.9
Outpatient management of suspected EVALI may be considered on a case-by-case basis when there is evidence of a less severe injury. Outpatients should have an oxygen saturation higher than 95%, have access to care and support systems, and be advised to seek emergency medical care if symptoms worsen. Initiation of empirical corticosteroids, such as prednisone 40 mg to 60 mg by mouth daily for 5 to 10 days, should be considered. Follow-up should be initiated within 24 to 48 hours in order to assess for and manage disease progression, as well as reemphasize the importance of cessation of vaping products.8
Cessation services and preventive care should not be overlooked in the counseling of patients with a history of EVALI and those who are risk for developing the disease. Providers should offer education and cessation assistance to patients with nicotine addiction, and patients with marijuana-use disorders should be referred to a professional. The CDC also recommends that patients receive routine influenza vaccinations and consider receiving the pneumococcal vaccine.3
Vaping Case Reports
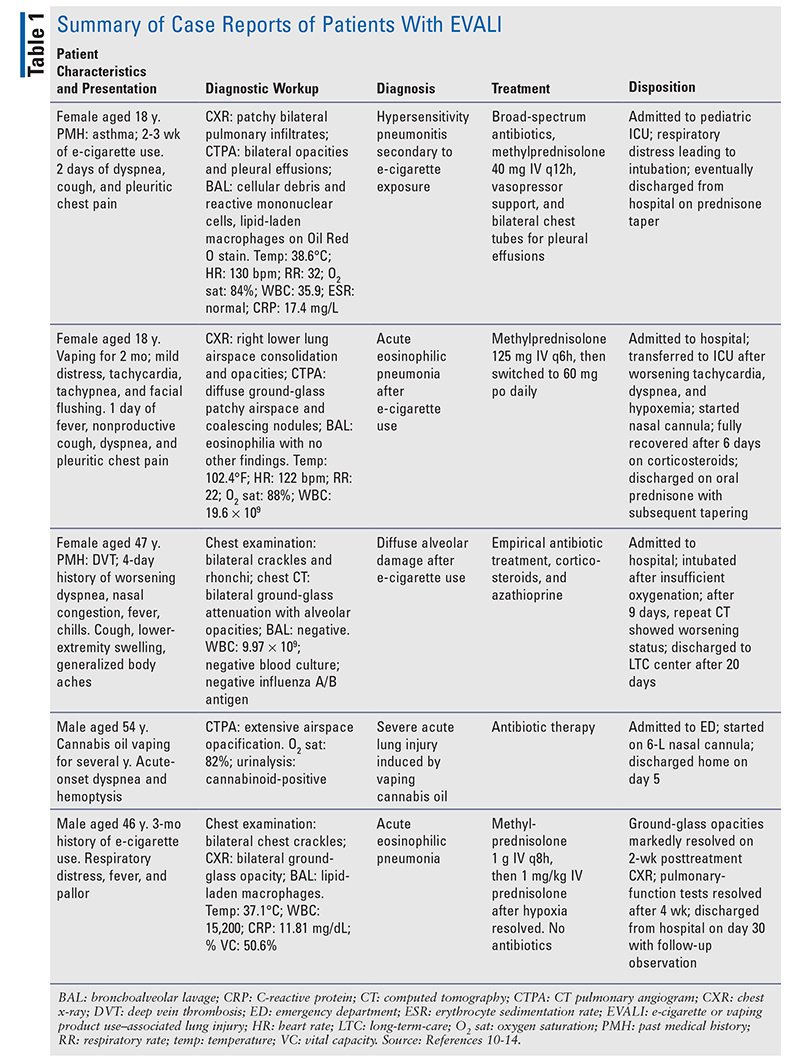
There are numerous case reports of patients with EVALI, some of which are summarized in TABLE 1.10-14 The duration of vaping does not appear to correlate with disposition. Patients in these reports presented with a combination of respiratory, gastrointestinal, and constitutional symptoms. Most patients exhibited bilateral ground-glass opacities upon computed tomography pulmonary angiogram. Patients were admitted and treated with corticosteroids and/or antibiotics, and most were discharged home on a prednisone taper.
Conclusion
As the practice of vaping continues to grow in popularity, so will the number of cases of EVALI grow. These dangerous trends emphasize the importance of the research currently being conducted to identify and eliminate the root cause of EVALI. Current evidence suggests a correlation between vitamin E acetate and lung injury, but more studies need to be conducted to rule out other causes. Pharmacists can counsel patients on vaping cessation and preventive measures, such as the influenza and pneumococcal vaccines. Pharmacists also should educate patients about the risks associated with vaping.
REFERENCES
1. Kales SN, Christiani DC. Acute chemical emergencies. N Engl J Med. 2004;350(8):800-808.
2. Matthay MA, Zemans RL, Zimmerman GA, et al. Acute respiratory distress syndrome. Nat Rev Dis Primers. 2019;5(1):18.
3. CDC. Electronic cigarettes. www.cdc.gov/tobacco/basic_information/e-cigarettes/severe-lung-disease/need-to-know/index.html#what-is-new. Accessed November 10, 2019.
4. U.S. Department of Health and Human Services. E-Cigarette Use Among Youth and Young Adults. A Report of the Surgeon General. Atlanta, GA: U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Center for Chronic Disease Prevention and Health Promotion, Office on Smoking and Health; 2016.
5. Gentzke AS, Creamer ML, Cullen KA, et al. Vital signs: tobacco product use among middle and high school students—United States, 2011-2018. MMWR Morb Mortal Wkly Rep. 2019;68(6):157-164.
6. Dai H, Leventhal AM. Prevalence of e-cigarette use among adults in the United States, 2014-2018. JAMA. 2019;322(18):1824-1827.
7. Perrine CG, Pickens CM, Boehmer TK, et al. Characteristics of a multistate outbreak of lung injury associated with e-cigarette use, or vaping—United States, 2019. MMWR Morb Mortal Wkly Rep. 2019;68(39):860-864.
8. Siegel DA, Jatlaoui TC, Koumans EH, et al. Update: interim guidance for health care providers evaluating and caring for patients with suspected e-cigarette, or vaping, product use associated lung injury—United States, October 2019. MMWR Morb Mortal Wkly Rep. 2019;68(41):919-927.
9. Blagev DP, Harris D, Dunn AC, et al. Clinical presentation, treatment, and short-term outcomes of lung injury associated with e-cigarettes or vaping: a prospective observational cohort study. Lancet. 2019;394(10214):2073-2083.
10. Sommerfeld CG, Weiner DJ, Nowalk A, Larkin A. Hypersensitivity pneumonitis and acute respiratory distress syndrome from e-cigarette use. Pediatrics. 2018;141(6):e20163927.
11. Arter ZL, Wiggins A, Hudspath C, et al. Acute eosinophilic pneumonia following electronic cigarette use. Respir Med Case Rep. 2019;27:100825.
12. Bakre SA, Al-Farra TS, Al-Farra S. Diffuse alveolar damage and e-cigarettes: case report and review of literature. Respir Med Case Rep. 2019;28:100935.
13. He T, Oks M, Esposito M, et al. “Tree-in-bloom”: severe acute lung injury induced by vaping cannabis oil. Ann Am Thorac Soc. 2017;14(3):468-470.
14. Itoh M, Aoshiba K, Herai Y, et al. Lung injury associated with electronic cigarettes inhalation diagnosed by transbronchial lung biopsy. Respirol Case Rep. 2017;6(1):e00282.
The content contained in this article is for informational purposes only. The content is not intended to be a substitute for professional advice. Reliance on any information provided in this article is solely at your own risk.
To comment on this article, contact rdavidson@uspharmacist.com.






