US Pharm. 2014;39(9):HS2-HS6.
ABSTRACT: Vasectomy is the only surgical form of male contraception being practiced worldwide. Vasectomies are safe, effective, economical, and should be approached as permanent sterilization. The entire procedure can be performed quickly and is commonly done in an outpatient office or in an ambulatory surgery center under local anesthesia. Men tend to forego vasectomies based on misinformation or concern for pain during the procedure. Therefore, evaluation and consultation for prospective vasectomy patients prior to the procedure are provided. Surgical techniques used to anesthetize and isolate the vas deferens have evolved and are directed at decreasing pain, discomfort, and complications.
Vasectomy is the only permanent form of male contraception being practiced worldwide. Aside from being permanent, vasectomy is also considered to be a safe, effective, and economical form of contraception.1 Vasectomy is a surgical procedure that involves isolation and occlusion of the vas deferens, and is typically done under local anesthesia in various outpatient settings with a quick recovery time.2,3 It has been estimated that 500,000 vasectomies are performed annually, and vasectomy is the fourth most commonly used contraceptive method in the United States.4 When compared to tubal ligation, vasectomy is equally effective, less invasive, and less expensive.3 The cost is approximately one-fourth the cost of tubal ligation.5 Vasectomy is not 100% effective, with a recanalization rate of <0.5% with the most effective techniques.6
Surgical techniques used to isolate and occlude the vas deferens differ drastically.3 The two most common techniques used are the incisional method and the no-scalpel technique.7 The incisional method uses a scalpel to make one or two incisions approximately 1 to 2 centimeters in length to approach the vas deferens. The no-scalpel technique uses a sharp, pointed, forcepslike instrument to puncture the scrotum. When the two techniques were compared, there was no difference in efficacy. However, the no-scalpel technique resulted in a shorter operation time and decreased occurrence of vasectomy-related adverse events such as intraoperative and early postoperative pain, bleeding, and infection.2 For those reasons, the no-scalpel technique is the preferred method in the U.S.3
Medications used before, during, and after vasectomy procedures can range from topical anesthetics, local anesthesia with or without oral sedation, and general anesthesia to postoperative pain control (TABLE 1).8-12 General anesthesia is reserved for patients who decline local anesthesia or when the preoperative examination suggests that local anesthesia with or without oral medication will be insufficient.8 The purpose of using pharmacologic agents for the vasectomy procedure is to reduce adverse events such as pain and increase patient comfort.
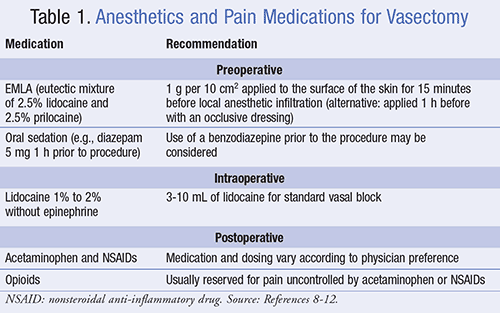
Preoperative
Prior to the procedure, a general medical history should be taken to identify contraindications and conditions that increase the risk of bleeding. A physical examination should also be performed to identify any structural abnormalities. In addition, a risk-benefit analysis should be done to determine if specific medications like anticoagulants, which may increase the risk for bleeding, should be stopped. Pharmacotherapy used in the preoperative phase is focused on decreasing pain and anxiety commonly associated with the procedure. Oral sedation may be necessary to aid in relaxing the patient.8 Drugs such as diazepam can be prescribed prior to the procedure, a common practice used in many other types of procedures. Use of a topical anesthetic before the procedure to help reduce procedural pain appears to be a matter of debate. Currently, only a few trials have reviewed this practice, with discrepant results.9,10
Additionally, the topical anesthetic cream EMLA (eutectic mixture of 2.5% lidocaine and 2.5% prilocaine) has the most data in vasectomy procedures.11 EMLA is now available in generic form in the U.S. Previously, before the availability of the generic product, cost may have been a factor in deciding upon its use.
Presently, two studies have investigated its use preoperatively.9,10 In the first study, Cooper found that EMLA decreased injection site pain when it was applied with an occlusive dressing 1 hour prior to surgery.9 In contrast, Thomas et al found that the application of EMLA 1 hour prior to surgery did not decrease pain during vasectomy.10 Although the conclusions of these studies varied, the overall designs differed as well. Therefore, it would be difficult to attest that the conclusions conflict.
Given the evidence, use of a topical anesthetic applied with an occlusive dressing 1 hour before the procedure may help to reduce injection site pain, so its use may be warranted. Current guidelines state that if the decision by the practitioner is to use this method, the cream should be applied by a health professional to prevent overapplication, which may lead to toxicity.8 Over-application of EMLA has been associated with adverse effects and toxicity.12 Dosing recommendations specifically for EMLA for use on adult male genital skin prior to local anesthetic infiltration is 1 g per 10 cm2 applied to the surface of the skin for 15 minutes.11 A topical anesthetic should not be used as the sole agent, as it will not provide sufficient analgesia.10
The average risk of acquiring an infection from a vasectomy is approximately 3.4%, which includes wound, urinary, and epididymal infections.13 Consequently, use of preoperative prophylactic antibiotics when using the no-scalpel technique is not universally recommended.14 Men with specific risk factors may be candidates for prophylactic antibiotics. The American Urological Association defines these risks factors as diabetes, advanced age, urinary tract abnormalities, poor nutritional status, smoking, chronic corticosteroid use, immunodeficiency, distant coexistent infection, and prolonged hospitalization. If a patient has one or more of these risk factors, indication for antimicrobial prophylaxis is not automatically warranted and should be left up to the individual physician performing the surgery.8
Procedure
Generally, all vasectomy procedures can be safely performed with local anesthesia alone. As stated previously, vasectomies are seldom performed under general anesthesia, but, on rare occasions, IV sedation may be necessary when local anesthesia alone is not enough.8 Once the vas deferens is isolated and positioned superficially, the local anesthetic is delivered.15 Lidocaine 1% to 2% without epinephrine appears to be the most commonly used anesthetic. Depending on the technique used, 3 to 10 mL of lidocaine is injected to deliver the standard anesthetic vasal block.16
Delivery methods used to anesthetize both the superficial scrotal area and the vas deferens are continually evolving. The focus on differing methods is to decrease pain associated with the initial injection. The two most common techniques used are the traditional injection technique and the newer no-needle jet injection technique. The traditional method essentially involves injection of the anesthetic into the scrotal skin following advancement of the needle into the deeper tissues to deliver the vasal nerve block.17 Needle-gauge size used to deliver the anesthetic may contribute to the amount of pain associated with the procedure. Shih et al set out to determine if using a smaller 30-gauge needle versus the standard 27-gauge would improve pain scores when performing a no-scalpel vasectomy. They concluded that there were no significant differences in pain measure between the two-needle sizes.16
The no-needle technique uses a pneumatic injector that delivers a high-pressured anesthetic spray through the scrotal skin and into the tissue around the vas deferens.18,19 This technique has been developed to help alleviate fear and concern that is associated with injection site pain. A study by White and Maatman set out to determine the effectiveness of this newer technique. They concluded that pain scores for initial pain were significantly decreased, but pain scores were not different for pain during the procedure when compared to the traditional method.19 In a separate cohort study, the no-needle technique was not found to be the best technique in terms of reducing pain scores.20 The no-needle technique seems to be as effective as the traditional delivery method, but one drawback is the price of the device.18,19
Lastly, the addition of medications like epinephrine, buffers, or corticosteroids to the anesthetic to aid in reducing pain and inflammation has been given some thought. Currently, there is not enough evidence to support their use when preforming a vasectomy, and the guidelines recommend against using them.8
Postoperative
Postoperative instructions should be given to the patient after a vasectomy procedure is performed. The patient will usually require some form of oral analgesia. The oral pain regimen used will vary and is up to the individual physician. Therefore, regimens as simple as OTC analgesics and more aggressive treatment with opioid therapy may be encountered. Additionally, application of cold temperatures may help decrease pain and inflammation.8
A more serious side effect associated with vasectomy is chronic testicular pain. Chronic testicular pain is considered a serious and long-term complication. This type of pain may be secondary to congestion epididymitis or sperm granuloma.13 Treatment may vary depending on the etiology of the pain.21 Generally speaking, treating chronic scrotal pain with regular use of acetaminophen or nonsteroidal anti-inflammatory drugs (NSAIDs) seems to be the most appropriate first step.21-23 If the initial attempt to control the pain fails, more aggressive pain management or surgical interventions may be needed.21 Other less common complications associated with the procedure include hematoma, infection, trauma, sperm granuloma, failure, urolithiasis, and death.13
Reversal
A vasectomy reversal procedure is available for men to pursue. The reversal process requires bypass of all obstructions within the male ductal system, which occur at the vasectomy sites but may also occur within the epididymis after vasectomy.24,25 Two common procedures in the vasectomy reversal process are vasovasostomy and vasoepididymostomy. In cases of vasal obstruction at the vasectomy site, vasectomy reversal can be accomplished by vasovasostomy. This process consists of a reestablished connection between the remaining testicular and abdominal portions of the vas deferens. Vasoepididymostomy is used in cases of secondary epididymal obstruction. The remaining abdominal portion of the vas deferens is connected to the epididymal tubule on the testicular side of the secondary epididymal obstruction. Although the procedure may be performed with any anesthetic, general anesthesia is preferred.26 Vasectomy reversal success rates have been documented in over 50% of men.27 Patency success rates as high as 99.5% have been achieved.28 Success rates decline as the time between vasectomy and reversal increases.25
Conclusion
Vasectomy is a safe and effective form of birth control in men that requires no preparation before or during sexual intercourse. Though a reversal procedure is available, patients should consider vasectomy a permanent form of birth control. As the number of vasectomies performed increases, the procedural methods as well as the type of anesthesia used continue to focus on increasing patient comfort and decreasing adverse events.
REFERENCES
1. Lohiya NK, Manivannan B, Mishra PK, Pathak N. Vas deferens, a site of male contraception: an overview. Asian J Androl. 2001;3(2):87-95.
2. Cook LA, Pun A, Gallo MF, et al. Scalpel versus no-scalpel incision for vasectomy. Cochrane Database Syst Rev. 2014;(3):CD004112.
3. Aradhya KW, Best K, Sokal DC. Recent developments in vasectomy. BMJ. 2005;330(7486):296-299.
4. Eisenberg ML, Henderson JT, Amory JK, et al. Racial differences in vasectomy utilization in the United States: data from the national survey of family growth. Urology. 2009;74(5):1020-1024.
5. Trussell J, Lalla AM, Doan QV, et al. Cost effectiveness of contraceptives in the United States. Contraception. 2009;79(1):5-14.
6. Barone MA, Irsula B, Chen-Mok M, Sokal DC; Investigator study group. Effectiveness of vasectomy using cautery. BMC Urology. 2004;4:10.
7. Schwingl PJ, Guess HA. Safety and effectiveness of vasectomy. Fertil Steril. 2000;73(5):923-936.
8. Sharlip ID, Belker AM, Honig S, et al; American Urological Association. Vasectomy: AUA guideline. J Urol. 2012;188(6 suppl):2482-2491.
9. Cooper TP. Use of EMLA cream with vasectomy. Urology. 2002;60(1):135-137.
10. Thomas AA, Nguyen CT, Dhar NB, et al. Topical anesthesia with EMLA does not decrease pain during vasectomy. J Urol. 2008;180(1):271-273.
11. EMLA (lidocaine 2.5% and prilocaine 2.5%) package insert. Lake Forest, IL: Oak Pharmaceuticals, Inc. (Akorn, Inc.); November 2012.
12. Hahn IH, Hoffman RS, Nelson LS. EMLA-induced methemoglobinemia and systemic topical anesthetic toxicity. J Emerg Med. 2004;26(1):85-88.
13. Awsare NS, Krishnan J, Boustead GB, et al. Complications of vasectomy. Ann R Coll Surg Engl. 2005;87(6):406-410.
14. Seenu V, Hafiz A. Routine antibiotic prophylaxis is not necessary for no scalpel vasectomy. Int Urol Nephrol. 2005;37(4):763-765.
15. Ramasamy R, Schlegel PN. Vasectomy and vasectomy reversal: an update. Indian J Urol. 2011;27(1):92-97.
16. Shih G, Njoya M, Lessard M, Labrecque M. Minimizing pain during vasectomy: the mini-needle anesthetic technique. J Urol. 2010;183(5): 1959-1963.
17. Li PS, Li SQ, Schlegel PN, Goldstein M. External spermatic sheath injection for vasal nerve block. Urology. 1992;39(2):173-176.
18. Weiss RS, Li PS. No-needle jet anesthetic technique for no-scalpel vasectomy. J Urol. 2005;173(5):1677-1680.
19. White MA, Maatman TJ. Comparative analysis of effectiveness of two local anesthetic techniques in men undergoing no-scalpel vasectomy. Urology. 2007;70(6):1187-1189.20. Aggarwal H, Chiou RK, Siref LE, Sloan SE. Comparative analysis of pain during anesthesia and no-scalpel vasectomy procedure among three different local anesthetic techniques. Urology. 2009;74(1):77-81.
21. Heidelbaugh JJ, Llanes M, Weadock WJ. An algorithm for the treatment of chronic testicular pain. J Fam Pract. 2010;59(6):330-336.
22. Schmidt SS. Spermatic grauloma: an often painful lesion. Fertil Steril. 1979;31(2):178-811.23. Keoghane SR, Sullivan ME. Investigating and managing chronic scrotal pain. BMJ. 2010;341:c6716.
24. Silber SJ. Vasectomy and its microsurgical reversal. Urol Clin North Am. 1978;5:573-584.
25. Belker AM, Thomas AJ Jr, Fuchs EF, et al. Results of 1,469 microsurgical vasectomy reversals by the Vasovasostomy Study Group. J Urol. 1991;145: 505-511.
26. Bernie AM, Osterberg EC, Stahl PJ, et al. Vasectomy reversal in humans. Spermatogenesis. 2012;2:273-278.27. Sharlip ID. What is the best pregnancy rate that may be expected from vasectomy reversal? J Urol. 1993;149:1469-1471.
28. Goldstein M, Li PS, Matthews GJ. Microsurgical vasovasostomy: the microdot technique of precision suture placement. J Urol. 1998;159:188-190.





