US Pharm. 2011;36(9):HS-35-HS-37.
As pharmacists, we are seeing more and more technologies introduced to streamline patient care, and electronic prescribing, or e-prescribing, is an invaluable tool when it is utilized correctly. There is considerable confusion in the marketplace over the definition of e-prescribing, however. So let's set the stage to understand what e-prescribing is and how is it different from other modalities currently in use.
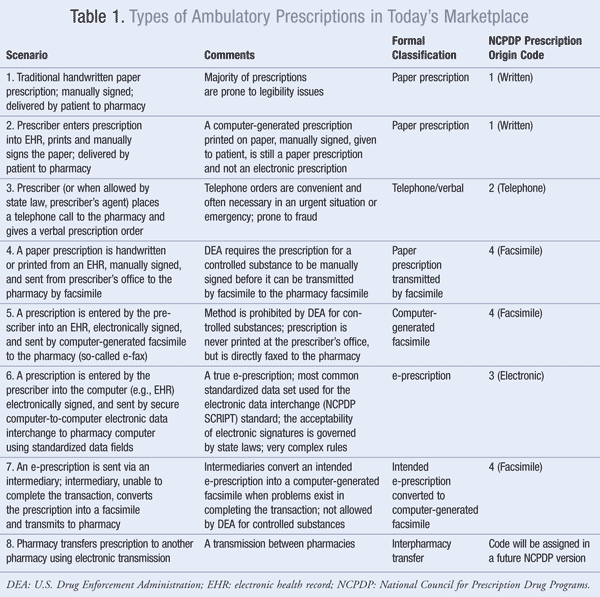
It is very important for pharmacists to understand the differences between true e-prescribing and other methods of generating or transmitting prescriptions, including computer-generated paper prescriptions, facsimile, and the so-called e-fax. Pharmacists should thoroughly understand how each type of prescription is generated and transmitted to the pharmacy. To assist with this discussion, refer to TABLE 1, which outlines various types of prescribing scenarios, types of prescriptions, and some of their properties. Also described is the Prescription Origin Code, which is required by many insurers, including Medicare, on the ambulatory pharmacy claim. Prescription origin codes are part of the National Council for Prescription Drug Programs (NCPDP) ambulatory code set. The pharmacist must understand each type of prescription in order to know how to properly apply the prescription origin code. And no wonder the confusion! When one reviews TABLE 1, there are just so many options in play in the marketplace, it becomes an abyss of information.
Parsing the Traditional Process
To understand the various scenarios, it is helpful to dissect the traditional written paper prescription process. The practitioner writes the prescription on a blank form and then manually signs the prescription. In some cases, an agent may actually prepare the prescription and then the legally authorized prescriber will manually sign off. The paper prescription is then given to the patient (or the patient's representative), who physically presents the original signed paper prescription to the pharmacy. The pharmacist would code this as a written paper prescription (Prescription Origin Code = 1). This simple example reveals several key processes. These are: 1) the process by which the prescription is produced; 2) the process by which the prescription is signed; and 3) the process by which the prescription is presented to the pharmacy.
In this scenario, the manual preparation of the prescription is the step where errors can be introduced. The prescriber must check for appropriateness of the dose, allergies and adverse reactions, drug interactions, formulary compliance, and clinical appropriateness regarding the patient's condition. Then the prescription must be written in such a manner as to be legible. For example, look-alike drug names can cause potential problems. Poor penmanship may lead to clinical errors.
The second scenario introduces a technical solution to address some of the issues raised in the first. In the second scenario, the preparation of the prescription is carried out on a computer, typically in an electronic health record (EHR), and then printed on paper. The obvious advantage here is that the printed-paper copy eliminates issues caused by poor penmanship. Another potential advantage is that the software package of the EHR can perform a number of edits to reduce the occurrence of clinical errors. The software can check the prescription against a list of known allergies and against a list of the patient's other medications. This functionality is known as computer decision support. Decision support can be very robust and might incorporate a number of edits, including drug-laboratory interactions, drug-age interactions, drug-allergy interactions, drug-drug interactions, drug-disease interactions, drug-formulary compliance, etc. Once the prescription is printed on paper and manually signed, it is still a written paper prescription. If the prescription were given to the patient to deliver to the pharmacy, then the pharmacy would still code the prescription as Prescription Origin Code 1. This is not an electronic prescription primarily because it is not transmitted to the pharmacy using a computer-to-computer electronic data interchange protocol with standard data fields. It is, however, related to an electronic prescription because it uses the same production process as an e-prescription.
Included in TABLE 1 are telephone orders to point out that the Prescription Origin Code for these is 2. Obviously, although the telephone is an electronic device, these prescriptions do not qualify as electronic prescriptions. Telephone prescriptions are becoming an increasingly problematic source of fraud, especially when the pharmacist is unable to positively validate the identity of the caller.
Role of the Facsimile
The next scenario introduces the facsimile (fax) as a method of transmitting an image of the paper prescription to the pharmacy. In this scenario, the facsimile replaces the patient, who is the courier in the first two scenarios. Again, although a facsimile is an electronic method of transmission, it is not an e-prescription. This is because the facsimile does not conform to a national standard for data fields. What exactly does this mean? A standardized data field has prespecified characteristics that define the allowed content of the field. For example, the data field for the patient's date of birth might be formatted as mm/dd/yyyy (month/day/year). Furthermore, such a structured data field can be read directly by the pharmacy computer. The computer software will be preprogrammed with the national standard, so that the software will read the first two characters as the month. The importance of adopting a national standard can be appreciated by the following logic. Another possible format for the date of birth could be dd/mm/yyyy. Hence, if the pharmacy computer were presented with the date 02/05/1956, in the absence of a standard the computer would not be able to determine if the correct date was May 2, 1956, or February 5, 1956. Thus, it is absolutely critical that both the sending computer and the receiving computer be using the identical standards. This is known as interoperable computer-to-computer electronic data interchange. Industrywide adoption of a single standard is critical to achieve interoperability.
Information coming in over a fax machine may or may not directly enter into the pharmacy computer. If it does, then the data are not structured as discrete computer-readable standard-based fields. Hence, the facsimile is a method of electronic transmission, but it does not meet the definition of e-prescribing. In most cases, the information transmitted by facsimile must be manually entered into the pharmacy computer. This transcription requirement with facsimiles is another human touch point where additional errors can creep into the prescribing process. The U.S. Drug Enforcement Administration (DEA) requires all paper prescriptions for controlled substances to be manually signed before they can be faxed to a pharmacy. State requirements would govern facsimiles in each state for noncontrolled prescriptions.
In the fifth scenario shown in TABLE 1, a prescriber enters the prescription into the computer (e.g., to an EHR), and then electronically signs the prescription. An image of the prescription is then sent directly by computer-generated facsimile to the pharmacy. In this case, the prescription is never printed on paper and is never manually signed by the prescriber. As in the case of the traditional facsimile, this so-called e-fax is not an e-prescription for the same reasons as stated above. The e-fax is an electronically created image of a hypothetical paper prescription, but it does not contain defined computer-readable structured data fields as specified by a national standard. This method of transmitting a prescription is prohibited by the DEA for controlled substances. The legality of an e-fax and the electronic signature for noncontrolled substances is subject to individual state laws.
The True e-Prescription
The sixth scenario on the table is a true e-prescription. In this case, the prescriber enters the prescription on the computer (e.g., into an EHR). The computer decision-support software then performs a series of edits as previously. The prescriber signs the prescription electronically. Then, the prescription is parsed by the software into a series of structured data fields following a national standard. The most commonly employed industry standard is the NCPDP Prescriber/Pharmacist SCRIPT standard. Updates to this standard are released periodically by the NCPDP. The structured data fields are then transmitted electronically by a secure protocol (encrypted) to the pharmacy computer. The transmission often goes through an intermediary, or e-prescribing, network that acts like a switch to route the prescription to the correct pharmacy. The pharmacy computer follows the same script standard and can directly read each structured data field and incorporate the data directly into its database. It should be noted that some older versions of pharmacy software printed the data but did not incorporate the information into the pharmacy database; the data needed to be transcribed back into the system by a human. This clearly defeats one of the key features of e-prescribing. It is highly recommended that pharmacies upgrade to modern software so as to avoid this problem. Both the DEA and many states have complex rules regarding the e-prescribing of controlled substances. These rules will be the focus of a future column. For noncontrolled prescriptions, state laws govern the use of electronic signatures.
The seventh scenario presents a twist on e-prescribing. In this case, the intention of the prescriber is to send a true e-prescription. However, the intermediary is unable to complete the transaction with electronic data interchange and converts the intended e-prescription into a facsimile. The pharmacy in this case will receive a computer-generated facsimile. The prescriber will usually not be aware that the intended e-prescription was converted into a facsimile. Why would the intermediary do this? Some common reasons are that an electronic network is down due to a power failure, a pharmacy does not accept e-prescriptions, or the pharmacy is not part of the intermediary's network. It should be noted, however, that the DEA prohibits intermediaries from converting an e-prescription for a controlled substance into a facsimile. In the event that such an e-prescription cannot be electronically delivered to the pharmacy computer, the intermediary is required to notify the prescriber. Then, the prescriber can print and manually sign the prescription and transmit the signed prescription to the pharmacy by facsimile.
The eighth scenario involves the electronic transfer of a prescription from one pharmacy to another. The currently available Prescription Origin Codes do not describe this scenario. The NCPDP has already planned a new code to describe this scenario, and the new code will be implemented in a future version of SCRIPT.
Conclusion
In summary, it is clear that marketplace technological innovations have resulted in a variety of processes to generate a prescription, to sign a prescription, and to transmit it to a pharmacy. Only true computer-to-computer electronic data interchange using a nationally accepted data standard is defined as genuine electronic prescribing. Various methods that employ a computer to print a prescription, or that employ facsimile, are not classified as e-prescribing.
To comment on this article, contact rdavidson@uspharmacist.com.






