US Pharm. 2020;45(9):HS-2-HS-6.
ABSTRACT: Adverse drug events (ADEs) significantly contribute to increased morbidity and mortality in hospitalized patients who experience them. Adverse drug reactions (ADRs) are a subset of ADEs that can be especially problematic in women, and therefore, when these adverse drug effects might be predictable, extra vigilance and caution are warranted. This is the case with certain medications used for sleep, dementia, arrhythmias, and diabetes. Unpredicted ADRs that are observed should be reported, investigated, and evaluated for clinical relevance with consideration for the potential future impact, when communicated, both to the patient and to the medical community.
An adverse drug event (ADE) is a broad term that describes harm associated with both appropriate and inappropriate medication use. The Agency for Healthcare Research and Quality states that ADEs are among the most common preventable adverse events in healthcare, regardless of the practice setting, and have been associated with the ubiquitous and increasing use of OTC and prescription medications.1 ADEs in hospitalized patients are associated with increased healthcare costs, hospital length of stay, morbidity, and mortality.1 Studies have examined the trends of hospitalizations that involve ADEs. According to Weiss and colleagues, the trend of patients experiencing an ADE that originated during hospitalization has remained relatively stable, while those that were detected upon admission increased to 70% in 2014 compared with 60% in 2010.2
Medication errors are categorized as ADEs, can lead to significant patient harm, and are always unintended.1 Adverse drug reactions (ADRs) are also categorized as ADEs and are considered unexpected, unintended, undesired, or excessive responses to a drug that occur as a common consequence of the use of medications, which includes appropriate prescribing, dispensing, and administration.3,4 While ADRs would be considered unintended, the adverse effect of some medications may be used opportunistically for intended, desirable effects, such as the sleep-inducing effects of antihistamines. Up to 5% of all hospital admissions are reported to be attributed to an ADR.5
This article will discuss the reporting of ADRs and some differences in the response in women to certain medications used in the hospital setting that ADR reporting has helped clarify.
ADR Reporting
During the course of a drug’s development through clinical trials and finally to market, the FDA reviews safety and efficacy data to ensure that the benefit of the drug far exceeds the risk of harm when using that agent. During this review, the FDA evaluates the full spectrum of ADRs from those that are rare but serious to those that are common but not considered serious. However, it is important to recognize that the population included in clinical studies is often predetermined to be young and healthy, and arguably does not reflect a real-world population of patients who will eventually use the drug. Once a drug goes to market and there is widespread exposure to a previously excluded cohort of patients, the FDA continues to monitor the safety of the drug through systematic pharmacovigilance that includes scientific collection of data on detection, assessment, and evaluation of reported adverse events.6 In 2014, the FDA communicated their goal of, and commitment to, enhancing their process of data collection during postmarketing surveillance to provide more robust demographic diversity to evaluate.7 The FDA has recently acknowledged that increased ADR reporting has led to the recognition of demographic factors, including sex, that may pose greater risk to hospitalized female patients.5
Despite the potential health benefits of increased ADR reporting, the difference in terminology and inconsistency of definitions used to describe these events can limit the magnitude of the positive impact (TABLE 1).8-11Additionally, the FDA contributes to this ongoing inconsistency with the interchangeable use of the different terms ADE and ADR in package labeling for events that could otherwise be considered side effects by those who avoid the use of the term ADR for anything other than a serious medication event.11,12
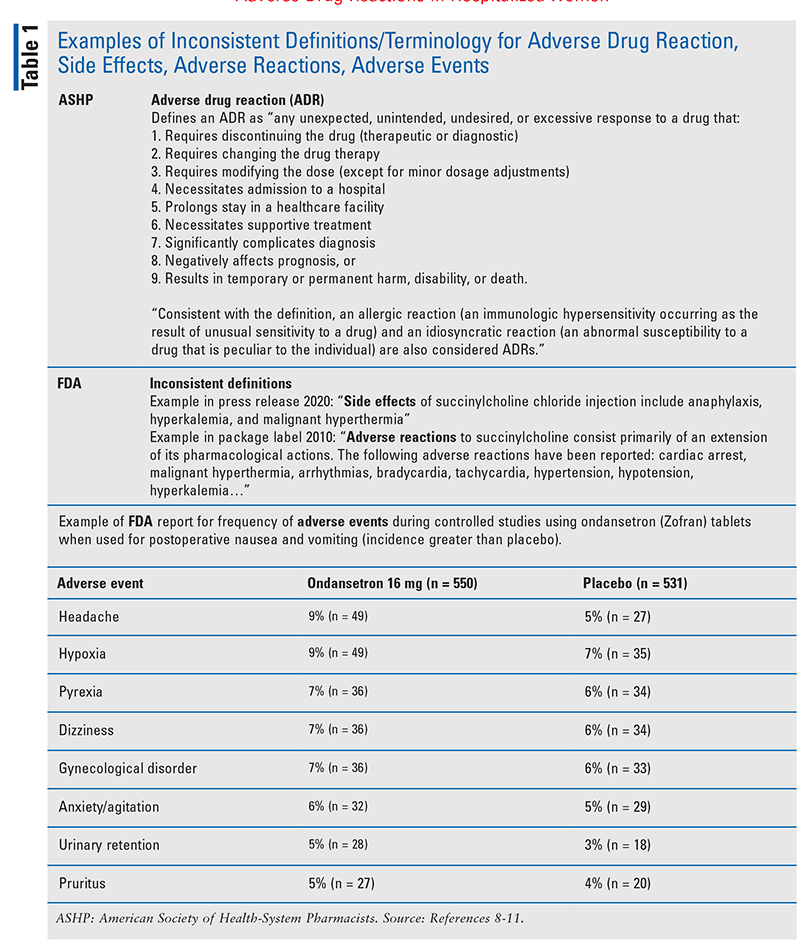
In addition to the issues surounding conflicting terminology, there is debate about the benefits and consequences of reporting. Chief among these debates is reluctance associated with prescriber perceptions that the recognition and subsequent reporting of ADRs poses increased risk for professional liability and administrative punitive outcomes.13 The medication-safety community, including such organizations as The Joint Commission, has encouraged identification and reporting of ADRs as a patient-safety measure.13
The continual refinement of the drug development and regulatory approval process has led to recognition that drug response and pharmacokinetic behavior is different in subgroup populations. Although the complete rationale is not known at this time, demographic differences and the emergence of demographic subgroups, including race, age, and sex, do translate into clinically meaningful differences that continue to be studied.14 The emergence of “at-risk groups” that have not been previously detected during initial clinical studies has recently prompted FDA action resulting in changes to dose and/or monitoring parameters of medications available in the United States. One such group is women.
Differences in Women
Women are reported to have up to a 1.7-fold greater chance of developing an ADR compared with males.5 However, some studies have observed higher death rates associated with ADRs in men.1 Some studies have reported that women are more likely to report ADRs and, thus, contribute to the overall higher rate of ADRs in women. Investigation of the differences in pharmacokinetic, hormonal, and other sex-based differences continue to influence research in this field.15
There is increasing evidence that idiosyncratic drug reactions are of immunologic origin. These medication reactions, also known as type B reactions, are known to occur rarely and unpredictably.5 It is hypothesized that sex-based differences in T-cell activation and growth compound women’s risk of developing diseases such as systemic lupus erythematosus, as well as increasing their risk for ADRs, due to this immune etiology.5
Pharmacokinetics
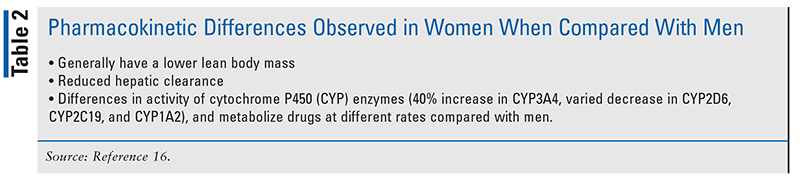
Pharmacokinetic outcomes have been reported to differ by sex.5 Conjugation, absorption, protein binding, and renal elimination all exhibit varying degrees of sex-based differences (TABLE 2).5,16 In addition to pharmacokinetic differences, reasons for the increased risks of ADRs in women have been reported to be based on differences in hormonal as well as immunologic factors and differences in medication use when women are compared with men.5
According to Parekh and colleagues, there is an increasing level of evidence, based on compelling cases that have resulted in adverse outcomes or observed differences in pharmacokinetics, for sex-based differences in medication response.14
Antiemetic: One example is the antiemetic drug ondansetron (Zofran). The FDA-approved package labeling includes data showing that women exhibit up to twofold higher drug concentrations and lower clearances, with no sex-specific dosing adjustments recommended for this variation. 11,14,17
Antipsychotic: In contrast, olanzapine (Zyprexa), a second-generation antipsychotic approved by the FDA for numerous psychiatric conditions, does recommend a lower dose based on patient-specific factors, including sex. Despite the black box warning to avoid use in older adults with dementia-related psychosis, the use of antipsychotics in older adults for the treatment of the behavioral and psychologic symptoms of dementia is increasing; thus, recognizing and planning for the appropriate use of these agents are essential.18
Olanzapine is extensively metabolized through CYP1A2 before reaching the systemic circulation, and in smokers, this metabolism is induced significantly more, resulting in a roughly 40% greater clearance. The labeling for olanzapine recommends a lower starting dose of 5 mg daily for patients with a combination of factors (e.g., nonsmoking female patients aged 65 years or older), because higher plasma concentrations and dose-dependent associated ADRs are expected to occur more frequently in these patients. Clinicians should be vigilant in predicting a higher olanzapine concentration in elderly nonsmoking females than would be seen, for example, in young males who smoke.14,19
Nonbenzodiazepine: Another medication class that has emerged recently with sex-specific dosing is the nonbenzodiazepine, benzodiazepine receptor agonist drugs (also known as the “Z” drugs”). Based on reports of excessive next-day serum concentrations, the FDA required an official reduction in dose.20 Additionally, those drugs recently received a class-specific black box warning that advises of complex sleep behaviors such as sleepwalking and sleep driving; however, to date, the FDA has not reported these behaviors to be more common in women.21
QTc Prolongation
One commonly encountered consequence of both pharmacokinetic and pharmacodynamic sex-based differences is QTc prolongation. Because this prolongation can result from a drug interaction that increases the serum concentration of the QTc-altering drug or a combination of drugs that share the same or greater QTc-prolonging risk, this medication-management challenge requires exceptional diligence on the part of the pharmacist to reduce the risk as much as possible.
Women are known to be at higher risk of QTc prolongation when compared with men, however; they are at even greater risk of QTc prolongation when given certain antiarrhythmic drugs compared with men even at equivalent serum concentrations. The mechanisms are unknown; however, there are studies examining the protective effect of testosterone, a physiologic sex divergence observed at puberty. Despite both sexes having similar prepubertal baseline QTc, men experience a shortening of baseline QTc measurements.10,11,13 Women have been associated with a longer baseline QTc interval and have an increased propensity for experiencing drug-induced torsades de pointes (TdP).22,23
Although some of these sex-based differences are illustrated in examples found in older studies, the accumulating evidence has now caught the attention of those involved in current drug development and regulatory review and approval processes. While these earlier examples reflected primarily pharmacokinetic differences, certain specific cases suggest that pharmacokinetics are not the entire story and involve critical care medications with a low threshold for causing serious harm.24-26
TdP Risk
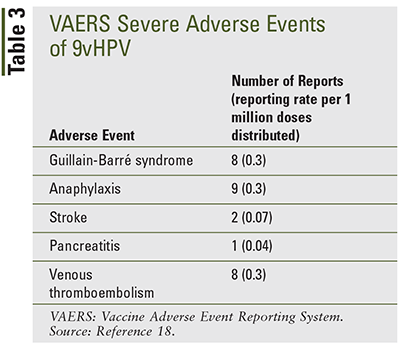
Antiarrythmic: Regarding TdP risk, one example is the antiarrhythmic dofetilide (Tikosyn), which is significantly affected by reduced renal clearance that impedes its primary elimination pathway.24 Higher than desired exposures to dofetilide can result in serious arrhythmias and TdP. According to Roukoz and Saliba, dofetilide serum concentrations were reported to be up to 22% higher in women after correcting for sex-related differences in creatinine clearance and body weight, resulting in nearly a threefold higher risk of TdP when compared with men.24 This greater exposure, coupled with what is now recognized to be a sex-associated heightened sensitivity to this adverse effect, requires concomitant consideration. In the case of dofetilide, the FDA has approved labeling that recommends a person-centered approach to dosing and monitoring of both creatinine clearance and QTc intervals at baseline and throughout the course of treatment for both men and women. Because estimated creatinine clearance factors in age, body weight, and a correction factor for women, calculated doses are generally less for women.25 This higher sensitivity and subsequent risk of QTc prolongation has also been reported in women for sotalol, and for quinidine (TABLE 3) at doses equal to those administered to men.22,25
Anticoagulants: An additional medication class that poses a potential increased risk of ADRs in women is the anticoagulants. Higher heparin serum concentrations have been observed for any given dose, and 17% less heparin was required to achieve therapeutic activated partial thromboplastin time when compared with men, even when there was a correction for body weight. This sex-based difference results in a higher bleeding risk, despite the use of dosing strategies that minimize bleeding events while maintaining therapeutic treatment goals.26
Fracture Risk
Antidiabetic agents: Antidiabetic drugs have been implicated in a sex-based fracture risk that has been reported with the use of the thiazolidinedione class of antidiabetic drugs.27 The authors of the Diabetes Outcome Progression Trial concluded that long-term use of rosiglitazone in women with type 2 diabetes was associated with roughly double the risk of bone fracture when compared with metformin and glyburide, with no increased risk of fractures observed in men.27 This difference was further analyzed to confirm that the risk was increased both in pre- and postmenopausal women; the authors noted that this ADR was an unexpected finding and not part of the prespecified analysis plan.27 Both anticoagulants and antidiabetic drugs are considered high-alert medications for both men and women according to the Institute for Safe Medication Practices; however, only the sulfonylurea antidiabetic agents and insulin are currently listed on this precautionary list.28
Role of the Pharmacist
Pharmacists play a major role in the detection, investigation, resolution, and prevention of ADRs. Pharmacists can also educate and reassure prescribers that ADRs are most often not a result of poor prescribing and that the reporting of ADRs is not only an essential element of pharmacovigilance but also a professional and ethical obligation to ensure optimal patient-safety outcomes. In terms of women’s health within the hospital setting, pharmacists can share findings of what may appear to be potential subgroup emergence of ADRs and can be proactive in making recommendations to mitigate future events. Pharmacists are also vital in motivating patients to report any adverse effects of medication they experience, and in working collaboratively with their care team to resolve these drug-related problems that often lead to suboptimal therapeutic efficacy or treatment failure due to nonadherence.
Conclusion
Emerging and evolving regulatory practices that focus specifically on women’s health have been developed, and according to the FDA, the approach now used to account for sex differences ensures more appropriate drug therapy. The FDA has recently acknowledged that increased ADR reporting has led to the recognition of sex as a demographic predictor of ADRs that may pose greater risk to hospitalized female patients. The FDA continues to encourage collaboration with healthcare providers, including pharmacists in all practice settings, in reporting severe and/or unusual ADRs and conducting further analysis of subgroup populations as a means of postmarketing surveillance. The continued debate over the definition of ADRs challenges the healthcare community to find a common understanding and a collaborative, consistent process of reporting these events as well as a culture that encourages sharing data to ensure optimal patient safety.
The content contained in this article is for informational purposes only. The content is not intended to be a substitute for professional advice. Reliance on any information provided in this article is solely at your own risk.
REFERENCES
1. Agency for Healthcare Research and Quality. Medication errors and adverse drug events. https://psnet.ahrq.gov/primer/medication-errors-and-adverse-drug-events. Accessed June 23, 2020.
2. Weiss AJ, Freeman WJ, Heslin KC, Barrett ML. Adverse drug events in US hospitals, 2010 versus 2014. HCUP Statistical Brief. 2018;234.
3. Shepherd G, Mohorn P, Yacoub K, May DW. Adverse drug reaction deaths reported in United States vital statistics, 1999-2006. Ann Pharmacother. 2012;46(2):169-175.
4. American Society of Health-System Pharmacists. ASHP guidelines on adverse drug reaction monitoring and reporting. Am J Health-Syst Pharm. 1995;52(4):417-419.
5. Rademaker M. Do women have more adverse drug reactions? Am J Clin Dermatol. 2001;2(6):349-351.
6. FDA. Questions and answers on FDA’s Adverse Event Reporting System (FAERS) www.fda.gov/drugs/surveillance/questions-and-answers-fdas-adverse-event-reporting-system-faers. Accessed June 23, 2020.
7. FDA. action plan to enhance the collection and availability of demographic subgroup data www.fda.gov/media/89307/download. Accessed June 23, 2020.
8. American Society of Health-System Pharmacists. ASHP guidelines on adverse drug reaction monitoring and reporting. Am J Health-Syst Pharm. 1995;52(4):417-419.
9. FDA. Coronavirus (COVID-19) Update: Daily Roundup June 17, 2020 www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-daily-roundup-june-17-2020. Accessed July 6, 2020.
10. Anectine (succinylcholine) package insert. Princeton, NJ: Sandoz Inc; 2010.
11. Zofran (ondansetron) package insert. Research Triangle Park, NC: GlaxoSmithKline; 2011.
12. Clozapine package insert. Rosemont, PA: HLS Therapeutics (USA), Inc: 2017.
13. The Joint Commission. Patient Safety Systems (PS). www.jointcommission.org/-/media/tjc/idev-imports/blogs/ps_chapter_omepdf.pdf. Accessed June 6, 2020.
14. Parekh A, Fadiran EO, Uhl K, Throckmorton DC. Adverse effects in women: implications for drug development and regulatory policies. Expert Rev Clin Pharmacol. 2011;4(4):453-466.
15. de Vries ST, Denig P, Ekhart C, et al. Sex differences in adverse drug reactions reported to the National Pharmacovigilance Centre in the Netherlands: an explorative observational study. Br J Clin Pharmacol. 2019;85(7):1507-1515.
16. Drici MD, Clement N. Is gender a risk factor for adverse drug reactions? Drug Saf. 2001;24(8):575-585.
17. Jann MW, ZumBrunnen TL, Tenjarla SN, et al. Relative bioavailability of ondansetron 8-mg oral tablets versus two extemporaneous 16-mg suppositories: formulation and gender differences. Pharmacotherapy. 1998;18(2):288-294.
18. Demler TL. The inpatient with dementia. US Pharm. 2014;39(11):HS18-HS25.
19. Zyprexa (olanzapine) U.S. FDA drug product labeling. http://dailymed.nlm.nih.gov.gate.lib.buffalo.edu/dailymed/drugInfo.cfm?id=31419. Accessed June 29, 2020.
20. FDA. FDA drug safety communication: Risk of next-morning impairment after use of insomnia drugs; FDA requires lower recommended doses for certain drugs containing zolpidem (Ambien, Ambien CR, Edluar, and Zolpimist). http://wayback.archive-it.org/7993/20170111080036/http:/www.fda.gov/Drugs/DrugSafety/ucm334033.htm. Accessed June 25, 2020
21. FDA. FDA adds Boxed Warning for risk of serious injuries caused by sleepwalking with certain prescription insomnia medicines. www.fda.gov/drugs/drug-safety-and-availability/fda-adds-boxed-warning-risk-serious-injuries-caused-sleepwalking-certain-prescription-insomnia. Accessed June 25, 2020.
22. Benton R, Sale M, Flockhart D. Greater quinidine-induced QTc interval prolongation in women. Clin Pharmacol Ther. 2000;67(4):413-418.
23. Cho L, Topol EI, Balog C, et al. Clinical benefit of glycoprotein Iib/IIIa blockade with abciximab is independent of gender. Pooled analysis from EPIC, EPILOG and EPISTENT Trials. J Am Coll Cardiol. 2000;36(2):381-386.
24. Roukoz H, Saliba W. Dofetilide: a new class III antiarrhythmic agent. Expert Rev Cardiovasc Ther. 2007;5(1):9-19.
25. Salazar DE, Much DR, Nichola PS, et al. A pharmacokinetic-pharmacodynamic model of d-sotalol Q-Tc prolongation during intravenous administration to healthy subjects. J Clin Pharmacol. 1997;37:799-809.
26. Campbell NR, Hull RD, Brant R, et al. Different effects of heparin in males and females. Clin Invest. Med. 1998;21(2):71-78.
27. Kahn SE, Haffner SM, Heise MA, et al. Glycemic durability of rosiglitazone, metformin, or glyburide monotherapy. N Engl J Med. 2006;355(23):2427-2443. 28. Institute for Safe Medication Practices. ISMP list of high-alert medications in acute care settings. www.ismp.org/sites/default/files/attachments/2018-08/highAlert2018-Acute-Final.pdf. Accessed August 11, 2020.
To comment on this article, contact rdavidson@uspharmacist.com.






