US Pharm. 2013;38(12):24-28.
ABSTRACT: Nausea
and vomiting (N/V) is a common complaint among patients across all ages
and levels of care. Symptoms of N/V may be self-treated or require
referral to another provider for further work-up of possible causes.
Selection of appropriate prevention and treatment is reliant upon
understanding the precipitating physiological mechanisms from which N/V
results. N/V may be caused by the vestibular system, stimulation of
receptors at the chemoreceptor trigger zone, or infectious origins.
Disease states may exacerbate symptoms or cause secondary N/V. The
pharmacist plays an important role in the management of N/V including
proper referral, medication selection, review of drug interactions or
causes, and patient counseling.
Nausea and vomiting (N/V) is a common complaint among patients across all ages and levels of care. Nausea is defined as the feeling of a need to vomit and vomiting as the expulsion of gastric contents via abdominal and chest wall contractions.1 The clinical presentation of N/V may involve the symptoms described above as well as signs of dehydration and electrolyte imbalance such as hypokalemia or metabolic acidosis. A thorough patient history for onset, timing of symptoms, nature of vomitus, and other signs that referral is appropriate is necessary to accurately assess the correct treatment option.1,2 The pharmacist should be able to assess medication efficacy for preventing and treating N/V to help establish a firm recommendation to the patient or provider.
Pathophysiology
Various receptors are involved in the process of N/V, which stimulate the central vomiting center (CVC) in the medulla to initiate the physiological process of N/V. The chemoreceptor trigger zone (CTZ) responds to noxious stimuli, gastrointestinal (GI) irritants, or chemotherapy. Located outside the blood-brain barrier, the CTZ is sensitive to systemic toxins, acidosis, and uremia. Dopamine-2 (D2), serotonin (5HT3), and neruokinin-1 (NK1) receptors are located in the CTZ and are mediated by GI distention, infection, and mucosal irritation. The vestibular system, as it pertains to N/V, is stimulated by the action of histamine1 (H1) and muscarinic (M1) receptors. Sensory input to the cerebral cortex is responsible for activation of the CVC and subsequent N/V. N/V is best differentiated based on the type of receptor influencing the reaction, and selection of the best treatment is dependent upon the mediating receptor and stimuli.1-4
Causes of N/V
Medications: N/V is a common side effect of medications to varying degrees of clinical relevance. Most instances of drug-induced N/V are from activation of D2, 5HT3, H1, and M1 receptors and are dependent on the specific medication utilized. Some drugs, such as metformin, may cause N/V when administered without food; simply administering with food would save a beneficial drug from being discontinued due to unpleasant side effects. Classes of medications commonly associated with N/V include opiates, digoxin, dopamine agonists, hormones, and nicotine.1
Chemotherapy: Cytotoxic medications used for cancer chemotherapy are especially tied to N/V, which is classified as postchemotherapy nausea and vomiting (PCNV) or chemotherapy-induced N/V (CINV). PCNV is further subdivided into the acute phase, which is N/V within 24 hours of chemotherapy; the delayed phase, which occurs after 24 hours postadministration of chemotherapy; and the anticipatory phase, brought about by the anticipation of chemotherapy administration. The treatment of PCNV varies on the emetogenicity of the chemotherapy agent used and usually necessitates more than one class of antiemetic.1
Disease States: Diseases that may induce N/V include gastroparesis, cyclic vomiting, migraine, gastric outlet obstruction, and peptic ulcer disease. Gastroparesis may be medication-induced or caused by physiological changes such as those found in patients with long-standing diabetes mellitus who rely on insulin for glucose control.5 Drugs that are found to induce gastroparesis include narcotics, clonidine, dopamine agonists, tricyclic antidepressants, calcium channel blockers, lithium, and progesterone.1
Infections: Infectious causes of N/V are typically acute in onset and self-limiting. Viral gastroenteritis and bacterial sources may be responsible. For example, food poisoning from staphylococcal enterotoxin will typically present within 6 hours of ingestion, with symptoms lasting up to 48 hours.6
PONV: Postoperative nausea and vomiting (PONV) may be experienced from inpatient or outpatient procedures. The overall incidence of N/V from outpatient procedures postdischarge has been estimated at 17% for nausea and 8% for vomiting.7 Symptoms may present and last up to 3 days after the procedure, making it difficult for providers to evaluate the patient for appropriate treatment in out-patient procedures. Pain, hypotension, dehydration, and anesthesia have been identified as risk factors for PONV, with treatment depending on the individual patient and on maximizing antiemetic therapy with multiple medications.7
Motion Sickness: N/V related to motion sickness and vertigo is due to stimulation of the vomiting center located in the vestibular system. Sensory nerves located here contain H1 and M1 receptors.3
Pregnancy: Pregnancy is frequently associated with N/V and is commonly referred to as morning sickness. The incidence of pregnancy-induced N/V is high, with up to 70% of women reporting N/V in the first trimester.1 One mechanism behind N/V in pregnancy is human chorionic gonadotropin (hCG) stimulation of the CTZ.4 Women often experience most N/V in the first trimester when hCG levels are highest, though it can occur at any point throughout the pregnancy.4,8 Other factors contributing to N/V in pregnancy include elevated estrogen levels, gastric reflux, and hyperthyroidism.4
Nonpharmacologic Therapy
Acupressure Wristbands: Devices such as the Sea-Band function by stimulating the pericardium 6 (P6) point on the forearm. They can be useful in preventing N/V in a variety of conditions, such as pregnancy or motion sickness, where traditional drug therapy may not be warranted or desired.2
Vitamins/Herbals: A variety of botanical products have been advocated for use in N/V. Ginger (Zingiber officinale) has some limited data to support use in pregnancy, motion sickness, and surgery.2 Ginger does not produce central nervous system (CNS) depression, and may be an alternative to antihistamines. Peppermint (Mentha piperita), chamomile (Matricaria recutita), and lemon balm (Melissa officinalis) are thought to have antispasmodic properties that may be helpful in treating N/V.2,6 Vitamin B6 (pyridoxine) has also been advocated for use in pregnant women experiencing N/V and is included in the recently approved product Diclegis (doxylamine-pyridoxine).9,10
Pharmacologic Therapy
Often pharmacologic means are necessary to prevent or treat N/V. Effective regimens are varied and carry unique considerations as to use in certain disease states, drug interactions, and adverse effects. Antiemetics are listed in TABLES 1 and 2.1,10-16

Antihistamines: These agents exhibit their antiemetic effect as a result of their antimuscarinic properties. Most of their efficacy is seen with the prevention of N/V, especially from vestibular causes such as motion sickness.1 Ideally, patients should be instructed to take the antihistamine 30 to 60 minutes before they expect to experience the stimulating event.2 The main side effects of antihistamines are anticholinergic, notably varying degrees of drowsiness.1 Worsening urinary retention should be considered in those predisposed to this effect because of conditions such as benign prostatic hyperplasia.1,2 The combination product Diclegis (doxylamine-pyridoxine) contains an antihistamine along with vitamin B6. It is Pregnancy Category A and is useful in N/V of pregnancy.10
Phenothiazines: These drugs work centrally at dopamine receptors in the CTZ and are available in a variety of dosage forms. They are useful for treating moderate-to-severe episodes of N/V related to motion sickness, PONV, and migraine. Adverse effects of this drug class include sedation, orthostasis, and extrapyramidal effects. Rare cases of neuroleptic malignant syndrome have been reported.1
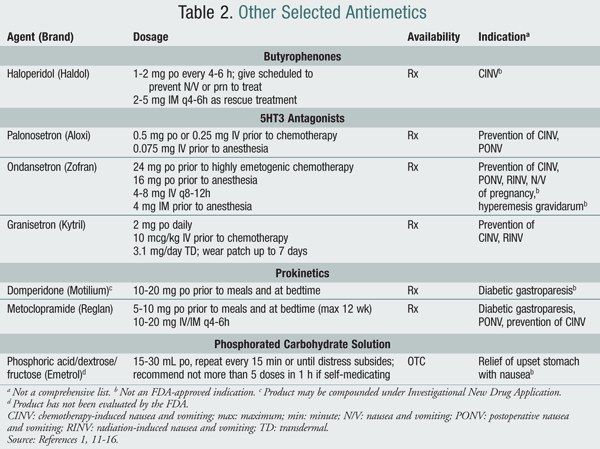
Butyrophenones: These agents are useful for PONV and anticipatory N/V caused by chemotherapy. Butyrophenones, such as haloperidol, have mechanisms and adverse-effect profiles similar to phenothiazines.1
5HT3 Antagonists: These drugs antagonize serotonin receptors both peripherally and centrally in the CTZ. In the periphery, 5HT3 receptors trigger vagal stimulation of the medulla, causing N/V.1 The selective blockade of both peripheral and central inhibition of 5HT3 receptors makes these agents a useful antiemetic for many forms of N/V.7 5HT3 antagonists are generally well tolerated and have become a staple for treatment of PCNV and CINV.1
Prokinetics: Antiemetics such as the benzamides antagonize dopamine receptors centrally, with effects both centrally and peripherally. Their prokinetic effects are useful for a variety of clinical N/V scenarios, especially those needing an increase in gastric motility, such as gastroparesis.12,15 Metoclopramide is primarily used in the United States because domperidone is only available as a compounded agent under an Investigational New Drug Application.11 Domperidone poorly penetrates the blood-brain barrier, and thus results in fewer central side effects such as tardive dyskinesias and extrapyramidal effects. Metoclopramide, due to its centrally mediated dopamine antagonism of receptors, has a significant adverse-effect profile that includes dystonia, dyskinesia, akathisia, and hyperprolactinemia, a potential cause of gynecomastia and lactation. The overall incidence of adverse effects is estimated at 10% to 20% with metoclopramide versus 5% to 10% with domperidone.1,12,15
Phosphorated Carbohydrate Solution: This mixture of fructose (1.87 g/5 mL), glucose (1.87 g/5 mL), and phosphoric acid (21.5 g/5 mL) is available OTC for N/V (Emetrol). Its mechanism may be related to delay of gastric emptying time and decreased smooth-muscle contraction. Because of its high carbohydrate content, this product is not recommended for patients with diabetes mellitus.2,16
When to Refer a Patient
Patients should be referred when presenting with severe or alarm symptoms such as signs of dehydration, fever, bloody vomitus, jaundice, changes in vision, severe headache, or neck pain. If a patient’s medical history includes asthma, chronic bronchitis, or emphysema, the patient may react adversely to OTC treatments and should be referred. If a pharmacist suspects that a patient’s N/V is related to anorexia or bulimia, he or she should ensure that the patient receives appropriate counseling by a trained professional. If poisoning is suspected, the pharmacist should call the Poison Control Help Line at 1-800-222-1222.2
Another patient presentation that requires referral is N/V due to a chronic condition, such as gastroparesis and refractory or prolonged N/V. Symptoms persisting longer than 1 week after an appropriate intervention is made could indicate a more serious etiology and should be referred.2
QT Prolongation Concerns
Prolongation of the QT may be a result of a medication’s mechanism of action, drug interactions, alteration of metabolism or elimination, or augmentation of concomitant QT prolongation effects from other agents. Many commonly prescribed antiemetics carry specific warnings regarding QT prolongation. For example, the FDA recently placed a warning for QT prolongation with ondansetron. Ondansetron’s QT prolongation is exhibited in a dose-dependent manner; hence, the FDA has advised to cease using the 32-mg single IV dosage.17 Domperidone may also significantly prolong the QT interval. For this reason, a baseline ECG is recommended with a corrected QT interval assessed prior to starting therapy.15,18,19
The manner in which pharmacists handle general QT prolongation warnings is dependent on the tools available to them. Most outpatient pharmacies will not have a patient laboratory or ECG capability, but should have the patient’s medication record. Watching for high-risk drug-drug and drug-disease state interactions, as well as educating patients on the risks of torsade de pointes, is necessary. Targeting patients with heart disease and individuals on medications that can increase the risk of potassium and magnesium abnormalities should help the pharmacist identify the appropriate counseling.18
Conclusion
N/V is a complex condition that presents with a great variety of disease states, medications, and other causes. Proper outpatient therapy is reliant on identifying the causative factors behind a patient’s N/V, knowing when referral is warranted, and selecting the appropriate treatment.
REFERENCES
1. Quigley EM, Hasler WL, Parkman HP. AGA technical review on nausea and vomiting. Gastroenterology. 2001;120:263-286.
2. Berardi RR, Ferrari SP, Hume AL, et al. Handbook of Nonprescription Drugs: An Interactive Approach to Self-Care. 16th ed. Washington, DC: American Pharmacists Association; 2009.
3. Hoffman R, Benz EJ Jr, Silberstein LE, et al. Hematology: Basic Principles and Practice. 6th ed. Philadelphia, PA: Saunders; 2012.
4. Casey G. Treating nausea and vomiting. Nurs N Z. 2012;18:20-24.
5. Choung RS, Locke GR III, Schleck CD, et al. Risk of
gastroparesis in subjects with type 1 and 2 diabetes in the general
population. Am J Gastroenterol. 2012;107:82-88.
6. Scorza K, Williams A, Phillips JD, Shaw J. Evaluation of nausea and vomiting. Am Fam Physician. 2007;76:76-84.
7. Cruthirds D, Sims PJ, Louis PJ. Review and
recommendations for the prevention, management, and treatment of
postoperative and postdischarge nausea and vomiting. Oral Surg Oral Med Oral Pathol Oral Radiol. 2013;115:601-611.
8. Niebyl JR. Clinical practice. Nausea and vomiting in pregnancy. N Engl J Med. 2010;363:1544-1550.
9. Ashkenazi-Hoffnung L, Merlob P, Stahl B, Klinger G.
Evaluation of the efficacy and safety of bi-daily combination therapy
with pyridoxine and doxylamine for nausea and vomiting of pregnancy. Isr Med Assoc J. 2013;15:23-26.
10. Diclegis (doxylamine succinate and pyridoxine hydrochloride) package insert. Bryn Mawr, PA: Duchesnay USA, Inc; 2013.
11. Micromedex Healthcare Series [Internet database]. www.micromedex.com. Greenwood Village, CO: Truven Health Analytics. Accessed August 28, 2013.
12. Facts & Comparisons 4.0. St. Louis, MO: Wolters Kluwer Health Inc. www.factsandcomparisons.com. Accessed August 28, 2013.
13. Zofran (ondansetron) package insert. Research Triangle Park, NC: GlaxoSmithKline; 2004.
14. Aloxi (palonosetron) package insert. Woodcliff Lake, NJ: OSO Biopharmaceuticals, LLC; 2013.
15. Camilleri M, Parkman HP, Shafi MA, et al; American
College of Gastroenterology. Clinical guideline: management of
gastroparesis. Am J Gastroenterol. 2013;108:18-37.
16. Emetrol Cherry (phosphorated carbohydrate solution) package insert. Sarasota, FL: Preferred Pharmaceuticals, Inc; 2012.
17. Ondansetron (Zofran) 32 mg, single IV dose: updated
safety communication—product removal due to potential for serious
cardiac risks. FDA. December 4, 2012.
www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm330772.htm.
Accessed August 29, 2013.
18. Roden DM. Drug-induced prolongation of the QT interval. N Engl J Med. 2004;350:1013-1022.
19. Kallergis EM, Goudis CA, Simantirakis EN, et al.
Mechanisms, risk factors, and management of acquired long QT syndrome: a
comprehensive review. ScientificWorldJournal. 2012;2012:212178.
To comment on this article, contact rdavidson@uspharmacist.com.





