US Pharm. 2017;42(6)(Generic Drugs suppl):14-18.
ABSTRACT: Triptans, as combination therapy or monotherapy, are the first-line option for the treatment of migraine in adults aged 12 years and older. Currently, seven triptans are on the market that may be administered in oral, SC, and nasal formulations. Various trials have proven the efficacy of triptans for acute migraine attacks and compared tolerability between drugs within the class. Most triptans are available as generics; however, pricing may become a challenge for patients experiencing more than three migraines per month, given the limits set by insurance companies. Adverse events commonly resulting from triptan therapy include feelings of tingling, numbness, warmth, and pressure or tightness in the chest and neck. The selection of triptan therapy should be individualized to the patient’s preference, comorbidities, risk of adverse events, and insurance coverage.
Migraine is one of the most common neurologic disorders in the United States. It has been reported that approximately 12% of Americans suffer from these headaches. According to the American Migraine Prevalence and Prevention study, 90% of migraineurs report being unable to function normally on days that they have a migraine, and nearly 30% require bed rest.1 Limited data are available regarding the mechanisms and physiology underlying the disorder; however, migraines are commonly associated with pain, nausea, vomiting, photophobia, and phonophobia.
Migraines are sometimes preceded by an aura, which is a sensation perceived before or during the migraine. Examples include visualizing flashing lights, smelling a distinct odor, feeling a breeze, and experiencing numbness, weakness, or difficulty speaking. A migraine aura develops gradually over a period of 5 minutes or more and may last as long as 60 minutes, and the visual and sensory symptoms are fully reversible.2 The aura is thought to be the result of cortical spreading depression (CSD), a wavelike depolarization across the cerebral cortex. CSD enables the activation of sensory components throughout the brain that play a significant role in pain processing during a migraine. The widespread pain of migraines is caused by specific connections between sensory components and the release of vasoactive neuropeptides. This cascade of events leads to neurogenic inflammation that may prolong the duration and worsen the pain of a migraine.3
Triptans are selective 5-hydroxytryptamine (5-HT) receptor agonists with high affinity for 5-HT1B and 5-HT1D receptors. Triptans relieve migraine pain by reducing neurogenic inflammation, lessening vasoconstriction of meningeal vessels, and modulating second-order neurons.4 These receptors are located on smooth-muscle cells of blood vessels, perivascular trigeminal nerve terminals, and the dorsal horn. Stimulation of 5-HT1B results in vasoconstriction; 5-HT1D stimulation blocks the release of vasoactive peptides and the release of neurotransmitters in the dorsal horn that carry nociceptive information to the thalamus.5
This article will summarize the role of triptans in the treatment of migraine, review evidence supporting their use, and discuss the cost, strengths, and formulations of available generics.
Migraine Management Guidelines
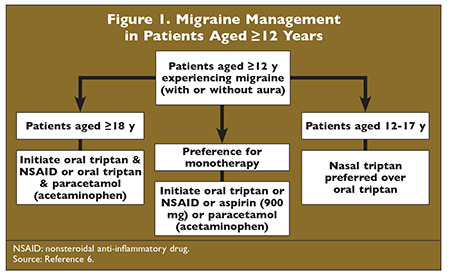
According to the guideline published in 2012 by the National Institute for Health and Care Excellence (NICE), first-line therapy for the acute treatment of migraine in persons aged 12 years and older is combination therapy with an oral triptan and a nonsteroidal anti-inflammatory drug (NSAID) or with an oral triptan and paracetamol (acetaminophen). Factors that should be considered in therapy selection include patient preference, comorbidities, and risk of adverse events (AEs). Based on clinical evidence, nasal triptans are preferred over oral triptans for any patient aged 12 to 17 years.6,7 In patients who prefer monotherapy to combination therapy for acute treatment, the recommended options include oral triptans, NSAIDs, aspirin, and paracetamol (acetaminophen). The initial treatment with a triptan should be determined based on patient preference, evidence, and affordability. If the first agent used is ineffective, one or more alternative triptans may be used.6
The NICE guideline recommends a step-down approach for acute treatment of migraine (FIGURE 1), a clinically effective and cost-effective method. This approach embraces initial treatment with the most effective combinations, followed by a reduction in the dose or number of treatments in order to determine the minimum dose and frequency of treatments needed to effectively treat the migraine. To prevent medication-overuse headache, it is important not to overuse triptans. Medication overuse (regular overuse of acute headache medication taken at least 10 days per month) can precipitate headaches. It is best to give the patient a higher-strength triptan on fewer occasions in order to prevent complications associated with overuse.8
Clinical Trials of Triptans
Prior to 2000, various trials established the effectiveness of sumatriptan in the treatment of acute migraine attacks. One crossover, double-blind, randomized, multicenter trial evaluated the efficacy and safety of oral sumatriptan 50 mg compared with placebo in 233 patients over the course of 12 migraine attacks. The crossover design resulted in a carry-over effect because the placebo was present only once for each randomized block of four attacks, meaning that each patient received nine active treatments and three placebo treatments for his or her 12 migraine attacks. Patients, who were aged 18 to 65 years, met International Headache Society criteria for migraine and had experienced migraine attacks of moderate-to-severe intensity for at least 1 year, with a frequency of one to six attacks per month. In the study, oral sumatriptan effectively relieved associated symptoms such as such as nausea, vomiting, photophobia, and phonophobia and reduced clinical disability caused by migraines. The efficacy rate was statistically significant for sumatriptan over placebo at 2 hours (sumatriptan 60% vs. placebo [PLO] 38%; P <.001) and 4 hours (sumatriptan 79% vs. PLO 47%; P <.001). The incidence of AEs did not differ between treatment groups. Results indicate that sumatriptan 50 mg oral tablets are more effective than placebo in the treatment of multiple migraine attacks.9
A randomized, parallel-group, double-blind, placebo-controlled trial investigated the use of oral naratriptan 2.5 mg in nonresponders to oral sumatriptan. Participants at 57 different sites in the U.S. had had an unsatisfactory response to sumatriptan, had a history of migraine for at least 1 year, and had experienced at least one migraine attack in each of the 2 months preceding the study. The study objective was to determine whether patients who responded poorly to sumatriptan would respond to naratriptan. The study found that naratriptan was superior to placebo for relief of migraine pain at 4 hours (P <.001), and that it was superior to placebo in patients with no pain at 4 hours (P = .022) and for headache relief at 2 hours (P = .005); however, it did not achieve statistical significance in patients with no pain at 2 hours. No significant AEs occurred. Overall, this trial established naratriptan as an effective treatment option for patients who are nonresponders to sumatriptan and demonstrated that patients who did not respond to one triptan may respond to other triptans.10
A different randomized, double-blind, placebo-controlled clinical trial assessed the efficacy and safety of oral almotriptan (12.5 mg and 25 mg) versus sumatriptan 100 mg and placebo in 668 migraine patients aged 18 to 65 years. Participants, whose migraine onset occurred before age 50 years, had at least a 1-year history of up to six migraine attacks per month, with at least 24 hours between attacks. Almotriptan 12.5 mg was tolerated to the same degree as placebo and was significantly better tolerated than sumatriptan (P <.001) when the incidence of AEs was considered. There was no statistically significant difference in the incidence of AEs between almotriptan 25 mg and sumatriptan 100 mg. Results indicate that almotriptan is an effective and well-tolerated treatment for migraine pain.11
Three similar randomized, placebo-controlled, head-to-head trials assessing the efficacy of eletriptan 40 mg compared with sumatriptan 100 mg were conducted. Participants were 2,906 adults who experienced up to six migraine attacks, with or without aura, per month. Eletriptan use resulted in significantly higher headache-response rates versus sumatriptan at both 1 hour and 2 hours (P <.0001), as well as higher pain-free response rates at 2 hours versus sumatriptan and placebo (P <.0001). Eletriptan showed superior efficacy compared with sumatriptan across all clinically relevant outcomes. Both eletriptan and sumatriptan were well tolerated, and reported AEs were mild and transient. Regarding AEs, eletriptan demonstrated tolerability comparable to that of placebo, with the exception of asthenia. In summary, the use of eletriptan to treat an acute attack was associated with a significantly higher probability of response compared with sumatriptan.12
AEs and Drug-Drug Interactions of Triptans
The most common AEs in randomized, controlled trials of triptans included feelings of tingling, numbness, warmth, and pressure or tightness in the chest and neck. These effects, frequently referred to as triptan symptoms or triptan sensations, occur more often in women and younger people. The most alarming symptoms are the tightness and heaviness of the chest, neck, or throat, which mimic ischemic myocardial dysfunction. However, patients with these symptoms show no evidence of decreased myocardial perfusion on ECG; therefore, chest and throat symptoms are not thought to be of cardiac origin. Although cardiac ischemia in association with triptan use is rare, triptans are contraindicated in patients with coronary artery disease (CAD), cerebrovascular disease, peripheral vascular disease, and uncontrolled hypertension. The coronary artery vessels have an abundance of 5-HT1B/1D receptors, which is significant because about 20% to 30% of the constrictor response is mediated through 5-HT1, whereas the 5HT2 receptors are the main receptors responsible for vasoconstriction. Newer second-generation triptans are more selective than sumatriptan in their action on the cerebral vessel, thereby decreasing cardiac risks, but all drugs in this class are contraindicated in patients with coronary disease.13
Triptan symptoms may be reduced by switching to a different triptan or route of administration, but it should be noted that patients receiving SC sumatriptan are more likely to experience AEs versus those taking oral formulations.13 In most cases, AEs were brief and mild-to-moderate, resulting in no change of therapy. In long-term studies, adverse drug effects resulted in withdrawal of therapy for SC and oral sumatriptan and for oral zolmitriptan. The incidence of AEs with naratriptan and almotriptan was comparable to that for placebo. The mechanisms causing common triptan symptoms are poorly understood, despite extensive research.13,14 Other mild AEs associated with triptans include somnolence and asthenia. The central-nervous-system origin of these events may be a result of the migraine attack, not the triptan, as these effects are observed only after successful treatment of the headache.15 The triptans are classified as Pregnancy Category C, with limited studies regarding their use in pregnancy. Sumatriptan has the most evidence to suggest this agent is a safe option for the treatment of acute migraine attacks during pregnancy and breastfeeding, as minimal amounts are excreted into breast milk.16
Potential drug-drug interactions occurring with triptans are most commonly seen in drugs that also interact with monoamine oxidase A (MAO-A), CYP450 enzymes, and serotonin receptors. Each triptan is metabolized in the liver, but differs in the extent of metabolism via MAO-A and CYP enzymes; therefore, when determining the best treatment option, each agent should be individualized in terms of concurrent medications and potential interactions. Sumatriptan and rizatriptan are metabolized only by MAO-A, whereas eletriptan, naratriptan, and frovatriptan are metabolized only by CYP enzymes. Zolmitriptan is metabolized by both MAO-A and CYP enzymes, similar to almotriptan, which is metabolized by MAO-A and CYP enzymes and flavin monooxygenase 3.17
Metabolism via CYP enzymes should also be considered to prevent drug interactions, especially with eletriptan, which is metabolized only by CYP3A4 and can interact with other drugs that are substrates for this enzyme. Dose adjustments are recommended when eletriptan is coadministered with CYP3A4 inhibitors or inducers.18 Caution is advised with the concomitant use of triptans and selective serotonin reuptake inhibitors (SSRIs) or selective serotonin-norepinephrine reuptake inhibitors (SNRIs) because of the increased risk of serotonin syndrome, although evidence is insufficient to prove this correlation.19 All prescribers and pharmacists should use caution in the coadministration of triptans and MAO-A inhibitors, relevant CYP enzyme inducers or inhibitors, and, most important, commonly prescribed SSRIs and SNRIs that are known to affect the serotonin receptors.
Cost Comparison of Triptans
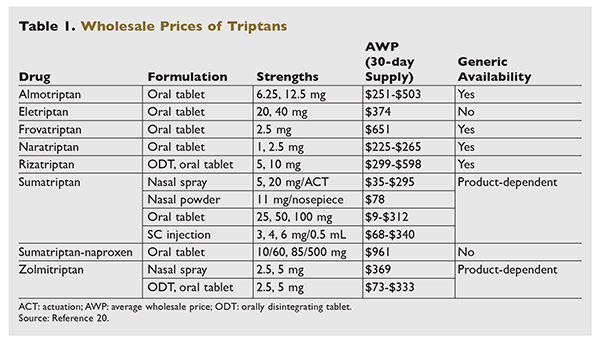
TABLE 1 summarizes the actual wholesale price (AWP) of each of the triptans currently on the market. AWPs for a 30-day supply range from $9 to >$600, and to >$900 for the combination product.20 Sumatriptan is not only the oldest triptan, but also generally the least expensive. Oral tablets are less expensive than the nasal spray and SC injection solutions. The brand-name combination therapy of sumatriptan and naproxen is significantly more expensive than these products purchased individually. All triptans that are available as generics are less expensive than the brand-name drugs. If migraine relief requires higher-strength tablets or nasal sprays, it is more advantageous, safer, and less expensive to use the higher-strength tablet or nasal spray rather than multiple doses of the lower-dose drugs.20,21 Frequent use of triptans is associated with medication-overuse headaches, which is why the prescriber should be contacted for the higher-strength formulation if the patient requires multiple doses of the lower-strength drug.8 Patients should also verify which triptans their insurance company covers, the number of doses covered per month, and whether there is a preferred triptan with lower copayments. Many insurance companies cover a limited number of triptan dosage forms per month, causing patients with three or more migraine attacks per month to pay high out-of-pocket costs for the additional necessary doses.21
Conclusion
Triptans are recommended as first-line therapy for the treatment of acute migraine attacks in combination with an NSAID or paracetamol (acetaminophen), or as monotherapy, if preferred. Triptans provide the best relief when taken as soon as migraine symptoms occur. The AEs and metabolism resulting in drug-drug interaction should be considered when therapy is being individualized. Triptans have shown favorable tolerability, and most formulations are available as generics, which has reduced costs and provided a greater selection when therapies are initiated or switched. Insurance companies often limit the number of doses a patient may receive each month, which becomes problematic for people who have frequent migraine attacks. Triptan therapy should be individualized to each patient’s needs and preferences through consideration of potential AEs, drug-drug interactions, and affordable prices.
REFERENCES
1. National Headache Foundation. Statistics from the American Migraine Prevalence and Prevention Study. www.headaches.org/2011/04/08/statistics-from-the-american-migraine-prevalence-and-prevention-study/. Accessed February 19, 2017.
2. Al-Quliti KW, Assaedi ES. New advances in prevention of migraine. Review of current practice and recent advances. Neurosciences (Riyadh). 2016;21:207-214.
3. Dux M, Sántha P, Jancsó G. The role of chemosensitive afferent nerves and TRP ion channels in the pathomechanism of headaches. Pflugers Arch Eur J Physiol. 2012;464:239-248.
4. Rapoport AM, Tepper, SJ, Sheftell FD, et al. Which triptan for which patient? Neurol Sci. 2006;27(suppl 2):S123-S129.
5. Pringsheim T, Becker WJ. Triptans for symptomatic treatment of migraine headache. BMJ. 2014;348:g2285.
6. National Institute for Health and Care Excellence. Clinical guideline (CG150). Headaches in over 12s: diagnosis and management. www.nice.org.uk/guidance/cg150/resources/headaches-in-over-12s-diagnosis-and-management-pdf-35109624582853. Accessed April 21, 2017.
7. Eiland LS, Hunt MO. The use of triptans for pediatric migraines. Paediatr Drugs. 2010;12:379-389.
8. Miller S, Matharu MS. Migraine is underdiagnosed and undertreated. Practitioner. 2014;258:19-24.
9. Bussone G, Manzoni GC, Cortelli P, et al. Efficacy and tolerability of sumatriptan in the treatment of multiple migraine attacks. Neurol Sci. 2000;21:272-278.
10. Stark, S, Spierings ELH, McNeal S, et al. Naratriptan efficacy in migraineurs who respond poorly to oral sumatriptan. Headache. 2000;40:513-520.
11. Dowson AJ, Massiou H, Laínez JM, Cabarrocas X. Almotriptan is an effective and well-tolerated treatment for migraine pain: results of a randomized, double blind, placebo-controlled clinical trial. Cephalalgia. 2002;22:453-461.
12. Diener HC, Ryan R, Sun W, Hettiarachchi J. The 40-mg dose of eletriptan: comparative efficacy and tolerability versus sumatriptan 100 mg. Eur J Neurol. 2004;11:125-134.
13. Nappi G, Sandrini G, Sances G. Tolerability of the triptans: clinical implications. Drug Saf. 2003;26:93-107.
14. Loder E. Triptan therapy in migraine. N Engl J Med. 2010;363:63-70.
15. Goadsby PJ, Dodick DW, Almas M, et al. Treatment-emergent CNS symptoms following triptan therapy are part of the attack. Cephalalgia. 2007;27:254-262.
16. Duong S, Bozzo P, Nordeng H, Einarson A. Safety of triptans for migraine headaches during pregnancy and breastfeeding. Can Fam Physician. 2010;56:537-539.
17. Fleishaker JC, Ryan KK, Jansat JM, et al. Effect of MAO-A inhibition on the pharmacokinetics of almotriptan, an antimigraine agent in humans. Br J Clin Pharmacol. 2001;51:437-441.
18. Buzzi MG. Pathways to the best fit of triptans for migraine patients. Cephalalgia. 2008;28(suppl 2):21-27.
19. Shapiro RE, Tepper SJ. The serotonin syndrome, triptans, and the potential for drug-drug interactions. Headache. 2007;47:266-269.
20. Red Book Online [Micromedex Solutions; subscription database]. Greenwood Village, CO: Truven Health Analytics, Inc. www.micromedexsolutions.com. Accessed February 19, 2017.
21. Using the triptans to treat migraine headaches: comparing effectiveness, safety, and price. Consumer Reports Best Buy Drugs. March 2013;1-21.
To comment on this article, contact rdavidson@uspharmacist.com.






