ABSTRACT: Appendicitis
is an inflammation of an obstructed appendix that may become infected,
gangrenous, and perforated. Classically, it presents as abdominal pain
that is often accompanied by nausea and vomiting. Appendicitis is the
most common cause of abdominal surgery in children, with the highest
incidence during the second decade of life. A ruptured appendix,
however, is more common in younger children. There is no one diagnostic
measure that is specific for appendicitis, and in many cases, a wrong
diagnosis may be made, particularly in children. The condition can be
managed with antibiotics as well as an appendectomy, if necessary.
Appendicitis is the most common cause of abdominal pain requiring surgical intervention, and the most common reason for emergent abdominal surgery in children.1-3 An individual’s lifetime risk for developing appendicitis is about 7%, and out of all the children who present to the emergency department with abdominal pain, 1% to 8% have appendicitis.1,3 In the United States, the current incidence of appendicitis is 86 per 100,000 patients per year, with 80,000 hospitalizations and more than one-third of hospital days for abdominal pain annually in patients under the age of 18 years.2,4 While appendicitis can affect all ages, including infants, the incidence is higher in the second decade of life.1 Moreover, the condition is slightly more common in males than females.1
Appendicitis with rupture (perforated appendicitis) is associated with a more severe clinical illness, higher morbidity, and longer hospital stays than non-perforated appendicitis.2 A ruptured appendix is common in young children, with rates as high as 100% for children under 1 year, reducing to about 82% for children between the ages of 1 and 5 years.5 The mortality rates for appendicitis ranges between 1% and 5% for young and elderly patients.6 This is partly due to the fact the younger children are unable to express themselves, and it is often assumed that they are suffering from gastroenteritis.
PATHOPHYSIOLOGY
The appendix is a long, thin diverticulum located on the posteromedial surface of the cecum, approximately 3 cm below the ileocecal valve.1,3 Its length varies from 8 to 13 cm in adults, and in children it averages around 4.5 cm.1,3 There is no known function of the appendix to date.1
Appendicitis occurs when the lumen of the appendix between the cecal base and the tip is occluded by fecaliths, adhesions, lymph node hyperplasia, foreign bodies, parasites, or, in rare cases, tumors.7,8 This obstruction causes the intraluminal pressure within the appendix to increase as mucosal secretions accumulate, leading to appendiceal distension and inhibiting lymphatic and venous drainage.1 The visceral afferent nerves that enter the spinal cord at T8 and T10 level are stimulated as a result of the distension, leading to a dull epigastric or periumbilical pain.1 A progressive condition causes overgrowth and translocation of bowel flora and leads to an acute inflammatory infiltrate in the wall of the appendix.1,2 As inflammation within the appendiceal serosa continues, it irritates the local somatic fibers on the parietal peritoneum, which leads to more localized pain. Within 24 to 36 hours of symptom onset, the appendix becomes gangrenous and is at risk for perforation, abscess formation, and peritonitis.9-11
ETIOLOGY AND RISK FACTORS
The organisms most commonly isolated from perforated appendicitis include Escherichia coli, alpha-hemolytic streptococci, Bacteroides species (e.g., Bacteroides fragilis), Bilophila wadsworthia, and Peptostreptococcus species.2 Various factors may increase or decrease a patient’s risk of developing appendicitis or perforation, which are detailed in TABLE 1.1-3

PRESENTATION
Classical appendicitis is characterized by constant pain that is poorly localized to the periumbilical or epigastric region.1 The pain is often accompanied by anorexia, nausea, and vomiting.1 As the condition progresses, the pain migrates towards the right quadrant at the area of the McBurney point and may be accompanied by low-grade fever.1 The McBurney point is one-third of the distance between the right anterior superior iliac spine to the umbilicus.2 Initially, the patient’s body temperature is normal or slightly increased, but fever develops as disease progresses.1
The clinical presentation may vary depending upon the location of the appendix, the host response to infection, and the age of the patient. Patients complaining of pain in the right flank, pelvis, testicle, suprapubic region, or even left lower quadrant (if the appendix crosses the midline) should be investigated for appendicitis.1 Furthermore, symptoms such as dysuria, increased urinary frequency, diarrhea, and tenesmus may be the result of appendicitis in patients whose appendix is in a slightly different location.12
A positive Rovsing sign (pain in the right lower quadrant when the left lower quadrant is pressed) is often a sign of appendicitis.1 However, the sensitivity and specificity of the Rovsing sign, psoas sign (abdominal pain that results when the patient flexes the thigh at the hip), and obturator sign (pain upon flexion and internal rotation of the hip), which are useful for diagnosis in adults, are not well established in children.1,2 Furthermore, patients suffering from appendicitis often display rebound tenderness or guarding as a result of the inflamed parietal peritoneum.1
It is important to note that while the classic presentation is fairly well established in adults, the signs and symptoms of appendicitis in children deviate greatly from the classical description.5 Furthermore, the child’s clinical condition varies in severity and clinical presentation.5 Anatomical and developmental differences contribute to age-based differences in pediatric appendicitis.2 Children’s appendices are less likely to be attached to the cecum, the ascending colon, or the abdominal wall, and, therefore, the appendix tends to be more mobile.2
In children under the age of 2 years, vomiting, abdominal pain, fever, abdominal distension, diarrhea, irritability, right hip pain, and limp are the most common symptoms.3 Physical findings include diffuse tenderness and fever.3 In children between the age of 2 and 5 years, abdominal pain precedes vomiting and is usually associated with fever and anorexia as well as right lower quadrant tenderness, fever, and involuntary guarding.2
Infants and toddlers commonly experience tenesmus that may be misdiagnosed for diarrhea.13 School-aged children describe abdominal pain that is constant and worsens with movement or coughing as well as nausea, vomiting, anorexia, tenesmus, and dysuria.3 They tend to have localized right lower quadrant tenderness or diffuse guarding and rebound tenderness.
The incidence of appendicitis peaks in adolescence and the late teen years.3 In females of childbearing age, pelvic pathology is easily confused with appendicitis. Appendicitis in older children may present as the feeling of hunger.3
DIAGNOSIS
Acute abdominal pain can be the result of many disorders including appendicitis, so it is useful to rule out the following diagnoses: infectious gastroenteritis, cholecystitis, right hip septic arthritis, pneumonia, urinary tract infection (UTI), mesenteric adenitis, intussusception, inflammatory bowel disease, Meckel’s diverticulum, hernia, primary peritonitis, orchitis, testicular torsion, blunt abdominal trauma, and ovarian cysts (females).2
A delay in diagnosis and treatment, possibly due to a difficulty in establishing a diagnosis or lack of access to healthcare services, substantially increases morbidity, length of hospital stay, and cost.3,14 Children are at a particularly higher risk of delayed diagnosis of appendicitis and its complications.1 This population may initially be misdiagnosed with gastroenteritis, UTIs, otitis media, or respiratory tract infections, resulting in increased perforation rates and hence longer hospital stays.15-17 A high index of suspicion is necessary during evaluation, and appendicitis should be considered as a differential diagnosis of abdominal pain.1 It is important to note that there is no single test specific for appendicitis.
Laboratory Tests
It has been shown that either the white blood cell (WBC) or neutrophil percentage is elevated in 90% to 96% of children with appendicitis.3 However, WBC counts are a useful but poor predictor of appendicitis on their own since elevations are also noted in nearly half of all patients with gastroenteritis, mesenteric adenitis, pelvic inflammatory disease, and other infectious disease.3 Furthermore, WBC or neutrophil counts do not discriminate between perforated and nonperforated appendicitis.3
The C-reactive protein (CRP) count has a low (43%-92%) sensitivity and low specificity (33%-95%) in children with acute abdominal pain.1,3 The combination of the WBC and CRP counts is a fairly accurate test for appendicitis in adults, but not in children.1,2 A urinalysis may be conducted to test for pyuria and hematuria, UTI, or nephrolithiasis.2
Imaging Studies
While newer modalities are increasingly used over the traditional WBC and CRP tests, some healthcare professionals feel that these tests increase costs but not accuracy.3 Plain film or radiography has limited use in the detection of appendicitis and is misleading in about 82% of children.3 Radioisotope-labeled WBC scanning in children shows sensitivity of about 27% to 97% and specificity of 38% to 94%. This procedure is lengthy and prone to poor interpretation agreement. Moreover, many emergency centers are unfamiliar with this technique.3
An ultrasound is particularly useful in children in whom harmful ionizing radiation is to be avoided.1 It is sensitive and specific for appendicitis (90%-95%) but may give a false negative if the appendix is perforated.18 In some cases, the appendix may not be visible at all on an ultrasound even if the sonographer is highly experienced.1
CT scans are widely available and can detect other abdominal disorders.1 A CT scan has a sensitivity (92%-97%) and specificity (98.6%-99%) that are higher than ultrasound in both adults and children.2,3 However, a CT scan exposes patients to radiation and delays diagnosis.1 MRI techniques are also useful since they avoid the use of radiation and contrast.3
Clinical Scores
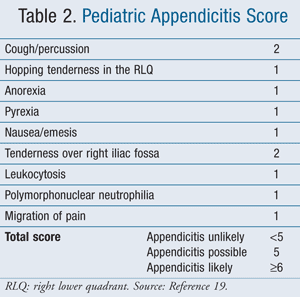
The Alvarado or MANTRELS score (Migration to the right iliac fossa, Anorexia, Nausea/vomiting, Tenderness in the right iliac fossa, Rebound pain, Elevated temperature, Leukocytosis, and Shift of leukocytes to the left) has been adapted for use in children and named the Pediatric Appendicitis Score (PAS). It is used to determine the likelihood that a child has appendicitis (TABLE 2).19
TREATMENT
The choice of treatment depends upon the severity of the condition as well as the patient’s general condition. Children with appendicitis are frequently dehydrated and may be febrile, acidotic, and septic.5 If appendicitis is suspected, the child should receive nothing by mouth and be started on IV volume replacement.3 Adequate hydration is important for patients with suspected appendicitis, and in many cases IV fluids may be required even after an appendectomy.20 Broad-spectrum IV antibiotics effective against enteric aerobes and anaerobes should be administered immediately to all children with obvious perforation.3 Appendectomy is performed in nonperforated appendicitis and in some cases on perforated appendicitis.21 Analgesics may be administered for symptomatic relief and antiemetics for vomiting.1
Appendectomy
An appendectomy is a definitive treatment for appendicitis that can be performed using either laparoscopy or open technique. It is indicated in all patients that have early, nonperforated appendicitis. However, in cases where the appendix is perforated, an appendectomy may provide no further benefit and may be avoided altogether.5 Percutaneous drainage may be required in patients who develop an intra-abdominal abcess.22
Antibiotic Therapy
Antibiotic therapy is an important aspect of the management of ruptured (perforated) appendicitis.20 IV antibiotics that are effective against enteric gram-negative organisms and anaerobes including E coli and Bacteroides species should be initiated as soon as the diagnosis of appendicitis is established.1,20 If an appendectomy is required, preoperative antibiotics therapy may be continued to prevent postoperative infections. These can be stopped postoperatively if the appendix is not found to be gangrenous or perforated during the operation.20 In cases where the appendix is gangrenous, postoperative antibiotic therapy should be continued for 24 to 72 hours and for a minimum of 7 to 10 days if the appendix is ruptured. Postoperative antibiotics can be administered orally if the patient is otherwise well enough to be discharged.20
A single antibiotic is sufficient for a nonperforated appendicitis. Second- or third-generation cephalosporins such as cefoxitin or cefotetan are used in uncomplicated cases.1 Broader-spectrum coverage is obtained with piperacillin-tazobactam, ampicillin-sulbactam, ticarcillin-clavulanate, or imipenem-cilastatin.23 For perforated appendicitis, the most common combination is ampicillin, clindamycin (or metronidazole), and gentamicin.20 Alternatives include ceftriaxone-metronidazole or ticarcillin-clavulanate plus gentamicin. Antibiotic dosing is provided in TABLE 3.24-32

Penicillins: Bactericidal antibiotics inhibit the biosynthesis of cell wall mucopeptide. They are used in combination with beta-lactamase inhibitors in the management of appendicitis for extended coverage. These agents include:
- Ampicillin-sulbactam (beta-lactamase inhibitor), which demonstrates activity against some gram-positive, gram-negative, and anaerobic bacteria.24 It is available as a combination in a ratio of 2:1 (ampicillin:sulbactam)
- Piperacillin-tazobactam, which is useful against gram-positive, gram-negative, and anaerobic bacteria25
- Ticarcillin-clavulanate, which is effective against most gram-positive and gram-negative bacteria and most anaerobes26
- Imipenem-cilastatin, which can be used for the treatment of multiple-organism infections in which other agents do not have wide-spectrum coverage or are contraindicated because of the potential for toxicity.27
The most common side effects seen with penicillins are hypersensitivity reactions, which can give rise to a number of clinical syndromes including anaphylaxis, angioedema, and urticarial and maculopapular rashes in the early stages and serum sickness–like reactions and hemolytic anemia in later stages.22
Large IV doses of penicillins may be associated with hemolytic anemia and neutropenia.22 Gastrointestinal (GI) effects are common with oral administration. Patients receiving high doses or long courses of beta-lactams are at risk of neutropenia. Healthcare professionals should watch out for fever, rash, and eosinophilia.22
Aminoglycosides: These antibiotics are employed primarily to treat gram-negative infections. They bind to the 30S and 50S subunits of bacterial ribosomes and achieve their bacteriostatic effects by interfering with bacterial protein synthesis.
Gentamicin is effective against gram-negative bacteria including pseudomonads, and works synergistically with beta-lactams against enterococci.28 The dose of gentamicin needs to be adjusted based on the creatinine clearance and changes in volume of distribution. This drug may be administered intravenously or intramuscularly.28
Aminoglycosides can produce irreversible, cumulative ototoxicity that can affect both the cochlea and the vestibular system, resulting in hearing loss and dizziness, respectively.22 Other side effects include reversible nephrotoxicity, acute renal failure, respiratory depression, muscular paralysis, and hypersensitivity reactions. Concurrent use of nephrotoxic drugs as well as those that can cause neuromuscular blockage should be avoided.22
Metronidazole: This synthetic nitroimidazole derivative antibacterial and antiprotozoal agent is often used in combination with aminoglycosides and provides broad gram-negative and anaerobic coverage.29 Metronidazole in combination with aminoglycosides may be given orally or intravenously.
Metronidazole is associated with dose-related GI disturbances, including nausea, unpleasant taste in the mouth, vomiting, and diarrhea.22 Weakness, dizziness, ataxia, headache, drowsiness, insomnia, and changes in mood or mental state may occur. Some patients experience peripheral neuropathy and complain of tingling or numbness in the extremities.22
Clindamycin: This lincosamide is effective against gram-positive aerobic and anaerobic bacteria, except enterococci.30 The drug inhibits bacterial growth by binding to the 50S subunit of the bacterial ribosome and halting protein synthesis.
Clindamycin can cause GI side effects including diarrhea, pseudomembranous colitis, nausea, vomiting, abdominal pain, cramps, and an unpleasant or metalling taste in the mouth following the IV administration of high doses. Hypersensitivity reactions, leukopenia, and agranulocytosis have all been reported in patients taking clindamycin.30
Cephalosporins: Cefoxitin is a second-generation cephalosporin with activity against some gram-positive, gram-negative (non-Pseudomonas species), and anaerobic bacteria.31 Cefotetan is also a second-generation cephalosporin effective against gram-positive and gram-negative cocci.32 The side effects associated with cephalosporins are very similar to those with penicillins. Furthermore, patients who are hypersensitive to one group of antibacterials are highly likely to react to a drug from the other group, particularly penicillins and cephalosporins.
CONCLUSION
Despite the availability of effective therapies for the management of appendicitis, pediatric mortality rates remain high. This is mainly due to poorly developed diagnostic tools, particularly for young children. There is no one test that is specific for the diagnosis of appendicitis. Further investigation of diagnostic techniques as well as the development of guidelines for the management of this condition may assist in reducing complications in children.
REFERENCES
1. Horn AE, Ufberg JW. Appendicitis, diverticulitis and colitis. Emerg Med Clin N Am. 2011;29:347-368.
2. Long SS, Pickering LK, Prober CG. Principles and Practice of Pediatric Infectious Diseases. 4th ed. New York, NY: Elsevier Saunders; 2012.
3. Rothrock SG, Pagane J. Acute appendicitis in children: emergency department diagnosis and management. Ann Emerg Med. 2000;36:39-51.
4. Korner H, Sondenaa K, Soreide JA, et al. Incidence of
acute nonperforated and perforated appendicitis: age-specific and
sex-specific analysis. World J Surg. 1997;21:313-317.
5. Morrow SE, Newman KD. Current management of appendicitis. Semin Pediatr Surg. 2007;16:34-40.
6. Hardin DM Jr. Acute appendicitis: review and update. Am Fam Physician. 1999;60:2027-2034.
7. Collins DC. 71,000 human appendix specimens. A final report summarizing forty years of study. Am J Proctol. 1963;14:365-381.
8. Hadl H, Quah H, Maw A. A missing tongue stud: an unusual appendicular foreign body. Int Surg. 2006;91:87-89.
9. Bickell NA, Aufses AH, Rojas M, et al. How time affects the risk of rupture in appendicitis. J Am Coll Surg. 2006;202:401-406.
10. Nance ML, Adamson WT, Hedrick HL. Appendicitis in the young child: a continuing diagnostic challenge. Pediatr Emerg Care. 2000;16:160-162.
11. Foulds KA, Beasley SW, Maoate K. Factors that influence length of stay after appendectomy in children. Aust N Z J Surg. 2000;70:43-46.
12. Guidry SP, Poole GV. The anatomy of appendicitis. Am Surg. 1994;60:68-71.
13. Horwitz JR, Gursoy M, Jaksic T, et al. Importance of diarrhea as a presenting symptom of appendicitis in very young children. Am J Surg. 1997;173:80-82.
14. Neilson IR, Laberge JM, Nguyen LT, et al. Appendicitis in children: current therapeutic recommendations. J Pediatr Surg. 1990;25:1113-1116.
15. Rappaport WD, Peterson M, Stanton C. Factors
responsible for the high perforation rate seen in early childhood
appendicitis. Am Surg. 1989;55:602-605.
16. Golladay ES, Sarrett JR. Delayed diagnosis in pediatric appendicitis. South Med J. 1988;81:38-42.
17. Cappendijk VC, Hazebroek FW. The impact of diagnostic delay on the course of acute appendicitis. Arch Dis Child. 2000;83:64-66.
18. Minkes RK. Pediatric appendicitis workup. Medscape. http://emedicine.medscape.com/article/926795-workup#aw2aab6b5b5. Accessed February 3, 2013.
19. Samuel M. Pediatric appendicitis score. J Pediatr Surg. 2002;37:877-881.
20. Minkes RK. Pediatric appendicitis. Medscape. http://emedicine.medscape.com/article/926795-overview. Accessed February 3, 2013.
21. Malik AA, Bari SU. Conservative management of acute appendicitis. J Gastrointest Surg. 2009;13:966-970.
22. Sweetman SC, ed. Martindale: The Complete Drug Reference. 34th ed. London, UK: Pharmaceutical Press; 2005.
23. Dominguez EP, Sweeney JF, Choi YU. Diagnosis and management of diverticulitis and appendicitis. Gastroenterol Clin North Am. 2006;35:367-391.
24. Ampicillin/sulbactam. MD Consult. www.mdconsult.com/das/pharm/body/402391908-8/1406642793/full/35. Accessed February 7, 2013.
25. Piperacillin/tazobactam. MD Consult. www.mdconsult.com/das/pharm/body/402391908-9/1406643143/full/491. Accessed February 7, 2013.
26. Ticarcillin/clavulanate. MD Consult. www.mdconsult.com/das/pharm/body/402391908-10/1406643453/full/612. Accessed February 7, 2013.
27. Imipenem/cilastatin. MD Consult. www.mdconsult.com/das/pharm/body/402391908-13/1406645113/full/308. Accessed February 7, 2013.
28. Gentamicin. MD Consult. www.mdconsult.com/das/pharm/body/402391908-14/1406645914/full/275. Accessed February 7, 2013.
29. Metronidazole. MD Consult. www.mdconsult.com/das/pharm/body/402391908-4/1406636161/full/398. Accessed February 7, 2013.
30. Clindamycin. MD Consult. www.mdconsult.com/das/pharm/body/402391908-5/1406637084/full/133. Accessed February 7, 2013.
31. Cefoxitin. MD Consult. www.mdconsult.com/das/pharm/body/402391908-6/1406640305/full/104. Accessed February 7, 2013.
32. Cefotetan. MD Consult. www.mdconsult.com/das/pharm/body/402391908-7/1406641311/full/103. Accessed February 7, 2013.
To comment on this article, contact rdavidson@uspharmacist.com.





