US Pharm. 2015;40(7):36-41.
ABSTRACT: Over the last several decades, multiple studies have identified improper asthma rescue inhaler technique as a clinically important correlate of unstable disease and increased use of healthcare services. It is believed that inconsistencies in what is described as “proper” technique by various sources may contribute to patient confusion and poor outcomes in clinical practice. It is important to review pressurized metered-dose inhaler (pMDI) protocols as defined by the literature and manufacturers, as well as to understand how the technique employed in each step affects the delivered dose. Presumably, knowledge of this information will clarify aspects of pMDI usage that are potentially confusing, as well as inform patient education and the development of effective asthma action plans.
Asthma is a chronic, debilitating, and potentially fatal pulmonary disorder characterized by persistent inflammation and reversible obstruction of the airways.1 According to 2013 data from the CDC’s National Health Interview Survey, there are currently 16.5 million adults (7%) and 6.1 million children (8.3%) living with asthma in the United States. Additionally, it is estimated that 37 million Americans (12%) will be affected by asthma in their lifetime.2 The National Heart, Lung, and Blood Institute’s 2007 Expert Panel Report 3 presents clinical practice guidelines for the diagnosis and management of asthma and identifies four components deemed essential for effective management of the disease. Inherent in these components is a recognition of the need for proper, thorough, and repeated education on the use of asthma inhalers.1
Despite such calls for patient education (and demonstrable efforts to impart such instruction), literature on the widespread improper use of pulmonary drug delivery devices abounds.3-5 Perhaps just as disconcerting are the accompanying reports that healthcare providers (HCPs) are commonly unable to properly operate these devices.5,6 A recent assessment of patient ability to properly self-administer asthma rescue medication found that only 7% of patients surveyed were able to operate a pressurized metered-dose inhaler (pMDI) per the approved protocol.7 Equally striking was the finding that 63% of those with flawed technique missed three or more steps. A similar study from 2005 concluded that “between 28% and 68% of patients do not use MDIs or powder inhalers well enough to benefit from the prescribed medication.”5 Indeed, pMDI misuse has been associated with unstable disease, as well as with increased emergency department visitation and hospitalization.8 Such failings present a clear challenge to asthma maintenance goals and an obvious risk to patient welfare. Even in cases in which asthma is acceptably controlled (despite poor technique), pMDI operator error contributes to an estimated $5 to $7 billion annual loss of misfired medication and to the increased frequency of adverse events resulting from the systemic exposure that accompanies undesired oropharyngeal deposition of medication.5,9,10
Patient Inhaler Competency
Patient education has been shown to be effective at decreasing both inhaler misuse and the need for emergency medical treatment.11,12 Moreover, it has been demonstrated that repeated education on proper inhaler technique is critical and should be performed during every HCP interaction.1,4 However, several factors can interfere with a patient’s ability to comprehend the proper use of a pMDI, and these must be acknowledged if an appropriate strategy is to be enacted.
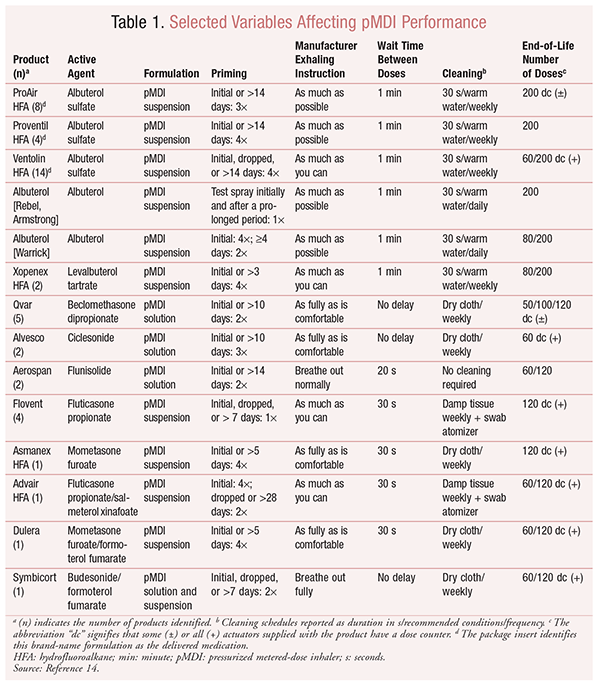
First, HCPs must recognize that competent use of one inhaler device does not necessarily translate to competency with others.13 TABLE 1 demonstrates that, even among devices delivering the same medication, differences exist in manufacturers’ package insert (PI) instructions on proper use.14 (The data in TABLE 1 are not exhaustive, but rather exemplify the variety of instructions available to patients. Some products [i.e., several of the ProAir products] are repackaged for direct distribution by physicians and are not intended for resale.) Further confounding attempts to effectively train patients is the 2013 finding that 17.5% of U.S. adults read at or below the most basic level of literacy.15 This is particularly alarming, since inadequate literacy strongly correlates with poor knowledge of asthma and improper pMDI use.16 There is cause for concern that such patients may not be able to satisfactorily follow printed directions. Fortunately, there is evidence that patients at more basic literacy levels can achieve inhaler competency with personalized instruction, a point that further supports the need for appropriate education.17
Effect of Improper Technique
The purpose of this article is to review patient-dependent aspects of inhaled pulmonary drug delivery and clearly describe how improper technique is expected to affect the respirable dose delivered by rescue pMDIs. While certain variables may be of greater or lesser importance in ensuring optimal drug delivery from solution or suspension pMDI formulations (e.g., shaking a suspension before actuation may be expected to be more critical than shaking a solution), this review will emphasize protocols that will work with both formulations.18 This should help reinforce intrapatient consistency of technique, even when multiple medications with differing instructions are indicated for maintenance and rescue.13 Likewise, much literature covering the performance of pMDIs addresses the original chlorofluorocarbon (CFC) formulations. While CFC-based devices are less efficient at delivering a respirable dose than hydrofluoroalkane (HFA) formulations and therefore can be expected to potentially respond differently to patient-controlled variables, concerns that are pertinent in CFC-based devices have been shown to affect HFA formulations as well.19
Short-acting beta-adrenergic agonists (SABAs) are currently the therapy of choice as rescue medications for acute asthma exacerbations. These medications are most often administered via pMDI and therefore are of particular concern in discussing appropriate inhaler technique.1 The preeminence of SABAs as inhaled rescue medications notwithstanding, inhaled corticosteroids (ICS) and combination medications will also be discussed, since differences in their use may contribute to patient confusion and improper technique.1,13
Critical Aspects of pMDI Operation
Storage: The inhaler should be stored at room temperature in the upright position, so that the tip of the canister valve is facing down. Temperature extremes are thought to change the vapor pressure of the propellant and can lead to changes in actuated particle size, flight, and deposition.20 Storing the canister valve-down helps keep the internal gasket wetted with propellant. If the canister is stored for extended periods in such a way that the gasket is not in contact with the liquid propellant, the gasket can become brittle and fail, potentially resulting in the ingress of moisture. The accumulation of moisture in the canister can affect aerosol formation and propellant evaporation upon actuation. Changes in these factors can affect particle size, flight, and deposition and ultimately decrease the respirable dose.21
Gasket failure may also result in the loss of propellant from leakage. Not only can this affect the pressure of the aerosol generated on actuation, it can also result in concentration of the medication and potentially lead to irreversible aggregation or precipitation in the canister.22
Shaking: Vigorous shaking of the pMDI before each actuation is of the utmost importance in ensuring reproducible delivery of the optimal respirable dose, with medications formulated as suspensions (all SABAs) being of particular concern. Flocculation—the aggregation of suspended drug particles into woolly clumps—occurs rapidly by design in pMDI canisters containing suspension formulations (FIGURE 1).23 These rapidly formed clumps readily disperse into a reasonably homogeneous suspension with vigorous shaking. While such flocculated systems are prone to form less easily dispersed creams over extended periods, formulations that allow for slower aggregation of drug particles are prone to caking. This yields a dense packing of drug that is much more difficult to resuspend.24 The salient point is that the suspension flocculates fairly quickly and must be redispersed before each actuation. PI instructions for solution-based pMDI formulations differ as to shaking requirements, with two products (Qvar and Alvesco) indicating no need for shaking. As the various SABA and ICS pMDI formulations are not expected to react adversely to shaking, advising patients to shake all pMDIs in the same manner may simplify patient education.

It is important to recognize that the contents of the pMDI metering chamber do not change substantially during the shaking process.25 Instead, shaking determines the contents of the next dose, which fills the metering chamber as the canister recoils after actuation. If the pMDI is primed before use (see below), it should be shaken before each priming actuation to ensure that the metering chamber is filled with a homogeneous suspension of medication. If the device is not thoroughly shaken before actuation, drug-free propellant can fill the metering chamber, resulting in an exceedingly low subsequent dose.26
Priming: Despite the recommendation to store the pMDI canister valve-down, doing so for relatively short periods (<24 hours) has been shown to result in leakage of suspended drug from the metering chamber back into the canister. Thorough shaking before actuation will not rectify this loss, so the first actuation after prolonged storage can be expected to deliver a significantly diminished dose.26 To counter the loss of medication to leakage, it is advised that multiple primer actuations be wasted to air before the first dose is administered for inhalation.
As mentioned above, it is necessary to thoroughly shake the device before each actuation, as this will ensure that the metering chamber is consistently filled with an optimal dose of medication. Not only does the duration of disuse requiring priming differ between inhalers, but a given inhaler may need a different number of priming actuations in different situations (TABLE 1). Also, dropping the inhaler is a specific indication for the priming of some (although not most) products. For some patients, it is anticipated that using a single consistent protocol will lead to better outcomes and that it may be advisable to prime all pMDIs according to the most frequent schedule. Although such a strategy may normalize usage, it will likely lead to unnecessarily wasted medication. Therefore, clear and thorough training on the proper use of each prescribed pMDI is optimal and is the most economical approach for patients.
Exhalation Prior to Actuation: A 2015 study found that “exhaling to functional residual capacity or residual volume” (i.e., normal, comfortable expiration) before actuating the canister was the most commonly missed step in the 93% of patients who failed to properly operate their rescue inhaler.7 Similarly, a 2002 study rated forced expiration as the most common error among patients misusing their ICS pMDI.4 Confusion over this step is understandable, considering the variety of instructions patients face pertaining to predose expiration (TABLE 1). Given that all albuterol rescue inhaler PIs instruct the patient to “breathe out as much as possible” or “as much as you can,” it should perhaps come as no surprise that the most common mistake in such studies is the failure to simply exhale normally.
It is unclear why forced expiration was considered an error in the 2002 study, other than that it did not conform to what the authors defined as proper technique. Forced expiration may result in poor control over the initial rate of the dosed inhalation, although the effects on airway deposition are unclear. Perhaps of greater concern is the lack of conscious expiration before dosing, which will prevent the deep inhalation necessary for optimal delivery of medication.27
In any event, full exhalation (presumably best defined by the relevant PI) immediately prior to actuation of the device is necessary for optimal drug delivery.4,7 Patients should be counseled to completely exhale before bringing the mouthpiece to their lips. A mistimed actuation coupled with exhalation into the mouthpiece will result in deposition of the dose in the actuator or spacer chamber. In addition, the high moisture content of exhaled air can affect the evaporation of the aerosolized propellant, thereby decreasing the extent of drug delivery by increasing both particle size (aggregation) and drug deposition in the actuator/spacer/mouth, even if the patient has fully exhaled before actuating the device.28
Actuation: Actuation should occur immediately after the inhaler is shaken. The device must be held in the proper upright orientation with the mouthpiece down, since tilting will lead to deposition of medication on the tongue or roof of the mouth. Multiple doses should not be cumulatively actuated for a single inhalation, but rather inhaled individually in succession according to the manufacturer’s instructions, including the recommended wait time between actuations (TABLE 1). Aggregation resulting in a decreased respirable dose has been shown to occur when multiple doses are actuated into an attached spacer; therefore, use of a spacer does not influence the need for inspiration following each actuation.29 It has been suggested that rapid successive actuations will lead to excessive cooling of the metering chamber owing to flash boiling of the propellant.25 This cooling could potentially result in contraction of the metering-chamber volume, thereby restricting the volume of the next loaded dose. In addition, excessive cooling of the propellant in the metering chamber can change the pressure gradient responsible for creating the aerosol plume, thereby decreasing the delivered dose.
Inhalation: Poor coordination of pMDI actuation and inhalation has been repeatedly identified as a considerable problem for asthma control. Patients should be advised, following full expiration, to place the mouthpiece between their lips and begin a slow, deep breath. Inspiration should begin immediately before actuation of the device, and the breath should continue until inspiratory capacity is reached.4 The majority of patients inhale too rapidly, which results in inertial impaction of drug to the back of the throat.27 A slow, deep inspiration is required to ensure optimal delivery of medication to the bronchial tree.
Spacers are effective at decreasing oropharyngeal deposition of inhaled medication, yet do not appreciably decrease the delivery of small respirable particles.30 When a spacer is used, the coordination of actuation and inhalation is less critical, and a brief delay (~1 second) has been shown to allow for inspiration of a full dose.31,32 If a spacer is used, it should be made of static-free material or regularly washed (see below) to minimize the buildup of static charge.33
Not every PI instructs the patient to hold his or her breath for a specific amount of time. Most state that the breath should be held for 10 seconds, although some simply say “as long as possible.” The majority of PIs direct the patient to remove the device from the mouth immediately after actuation, while others are unclear in this regard. Since exhalation into the mouthpiece can humidify the device and potentially interfere with delivery of a second dose, patients should be clearly instructed to remove the device from the mouth before exhaling.
Counting Remaining Doses: A recent study examining pMDI hardware concluded that patients commonly are unable to determine the end of life of their canister.34 While underestimation leads to wasted drug, overestimation renders unreliable any dose actuated beyond the nominal canister capacity. Techniques such as floating the inhaler in water, shaking the inhaler, or checking to see whether the inhaler still puffs are inaccurate and should not be recommended. Several modern actuators are equipped with built-in dose counters, although not all rescue inhaler PIs specifically state this. Of eight products marketed in the U.S. that were identified as containing repackaged ProAir HFA, only four mention the built-in dose counter in the PI.14 Since not all inhalers feature a dose counter, the patient’s asthma action plan must be carefully evaluated when switching products so as to ensure that the patient can maintain an accurate count of the doses available in the device.
Cleaning: For most pMDIs, regular cleaning is important for proper function. Residual medication can accumulate on the spray orifice after repeated actuations, resulting in direct blockage of the aerosol. Additionally, the processes of aerosol formation and propellant evaporation introduce a static charge to actuated drug particles.35 Actuators and spacers also develop a static charge in the course of normal use, which significantly decreases the respirable dose because of the pursuant electrostatic attraction between drug and device.32 Although static-free spacers have been developed, regular cleaning of plastic spacers with household detergent effectively removes static charge and improves drug delivery.33,36 TABLE 1 lists the recommended cleaning procedure for available pMDIs. The canister must not get wet and should be removed during the washing process to prevent ingress of water. In cleaning an actuator or spacer with water, the device should be allowed to dry completely before reassembly. Shaking the actuator after cleaning removes excess water and decreases drying time. If the inhaler is needed before it has dried, it should be actuated twice before use, and the cleaning procedure should be repeated afterward.
Conclusion
The general considerations for the appropriate use of pMDIs provided in this review are not inclusive for all of the counseling points unique to each device. A detailed asthma action plan should be developed for each patient in accordance with his or her ability to understand and perform the described tasks. While the manufacturer’s instructions for a given inhaler should be followed, the information provided herein clarifies the rationale behind several critical factors affecting the respirable dose a pMDI delivers. It is hoped that this information will help identify points of potential confusion and optimize care for patients with asthma.
REFERENCES
1. National Asthma Education and Prevention Program. Expert Panel Report 3 (EPR-3): Guidelines for the Diagnosis and Management of Asthma—Summary Report 2007. J Allergy Clin Immunol. 2007;120(suppl 5):S94-S138.
2. Centers for Disease Control and Prevention. 2013 National Health Interview Survey (NHIS) data. www.cdc.gov/asthma/nhis/2013/data.htm. Accessed March 31, 2015.
3. van Beerendonk I, Mesters I, Mudde AN, Tan TD. Assessment of the inhalation technique in outpatients with asthma or chronic obstructive pulmonary disease using a metered-dose inhaler or dry powder device. J Asthma. 1998;35:273-279.
4. Giraud V, Roche N. Misuse of corticosteroid metered-dose inhaler is associated with decreased asthma stability. Eur Respir J. 2002;19:246-251.
5. Fink JB, Rubin BK. Problems with inhaler use: a call for improved clinician and patient education. Respir Care. 2005;50:1360-1374; discussion 1374-1375.
6. De Tratto K, Gomez C, Ryan CJ, et al. Nurses’ knowledge of inhaler technique in the inpatient hospital setting. Clin Nurse Spec. 2014;28:156-160.
7. Bonds RS, Asawa A, Ghazi AI. Misuse of medical devices: a persistent problem in self-management of asthma and allergic disease. Ann Allergy Asthma Immunol. 2015;114:74-76.e2.
8. Melani AS, Bonavia M, Cilenti V, et al. Inhaler mishandling remains common in real life and is associated with reduced disease control. Respir Med. 2011;105:930-938.
9. Cochrane MG, Bala MV, Downs KE, et al. Inhaled corticosteroids for asthma therapy: patient compliance, devices, and inhalation technique. Chest. 2000;117:542-550.
10. Au DH, Curtis JR, Every NR, et al. Association between inhaled beta-agonists and the risk of unstable angina and myocardial infarction. Chest. 2002;121:846-851.
11. Osman LM, Calder C, Godden DJ, et al. A randomised trial of self-management planning for adult patients admitted to hospital with acute asthma. Thorax. 2002;57:869-874.
12. Castro M, Zimmermann NA, Crocker S, et al. Asthma intervention program prevents readmissions in high healthcare users. Am J Respir Crit Care Med. 2003;168:1095-1099.
13. van der Palen J, Klein JJ, van Herwaarden C, et al. Multiple inhalers confuse asthma patients. Eur Respir J. 1999;14:1034-1037.
14. U.S. National Library of Medicine. Repository of current package inserts [database]. http://dailymed.nlm.nih.gov/dailymed/archives/index.cfm?query=aerosol%2C+metered&date=04%2F21%2F2015. Accessed April 20, 2015.
15. Organisation for Economic Cooperation and Development. OECD Skills Outlook 2013: First Results from the Survey of Adult Skills. OECD Publishing; 2013. http://dx.doi.org/10.1787/9789264204256-en. Accessed March 30, 2015.
16. Williams MV, Baker DW, Honig EG, et al. Inadequate literacy is a barrier to asthma knowledge and self-care. Chest. 1998;114:1008-1015.
17. Paasche-Orlow MK, Riekert KA, Bilderback A, et al. Tailored education may reduce health literacy disparities in asthma self-management. Am J Respir Crit Care Med. 2005;172:980-986.
18. Robinson CA, Tsourounis C. Inhaled corticosteroid metered-dose inhalers: how do variations in technique for solutions versus suspensions affect drug distribution? Ann Pharmacother. 2013;47:416-420.
19. Smyth HD. The influence of formulation variables on the performance of alternative propellant-driven metered dose inhalers. Adv Drug Deliv Rev. 2003;55:807-828.
20. Wilson AF, Mukai DS, Ahdout JJ. Effect of canister temperature on performance of metered-dose inhalers. Am Rev Respir Dis. 1991;143(5 pt 1):1034-1037.
21. Murata S, Izumi T, Ito H. Effect of the moisture content in aerosol on the spray performance of Stmerin® D hydrofluoroalkane preparations (2). Chem Pharm Bull (Tokyo). 2012;60:593-597.
22. Vervaet C, Byron PR. Drug-surfactant-propellant interactions in HFA-formulations. Int J Pharm. 1999;186:13-30.
23. Jinks P, Hunt K. Improving suspension MDI dose consistency in patient use by incorporation of a novel semi-permeable system component. www.aerosol-soc.org.uk/files2/DDL17-2006/27.Jinks.pdf. Accessed June 19, 2015.
24. Wu L, Zhang J, Watanabe W. Physical and chemical stability of drug nanoparticles. Adv Drug Deliv Rev. 2011;63:456-469.
25. Everard ML, Devadason SG, Summers QA, Souëf PN. Factors affecting total and “respirable” dose delivered by a salbutamol metered dose inhaler. Thorax. 1995;50:746-749.
26. Byron PR. Dosing reproducibility from experimental albuterol suspension metered-dose inhalers. Pharm Res. 1994;11:580-584.
27. Ammari WG, Chrystyn H. Optimizing the inhalation flow and technique through metered dose inhalers of asthmatic adults and children attending a community pharmacy. J Asthma. 2013;50(5):505-513.
28. Martin AR, Finlay WH. The effect of humidity on the size of particles delivered from metered-dose inhalers. Aerosol Sci Technol. 2005;39:283-289. doi:10.1080/027868290929314.
29. Barry PW, O’Callaghan C. Multiple actuations of salbutamol MDI into a spacer device reduce the amount of drug recovered in the respirable range. Eur Respir J. 1994;7:1707-1709.
30. Ahrens R, Lux C, Bahl T, Han SH. Choosing the metered-dose inhaler spacer or holding chamber that matches the patient’s need: evidence that the specific drug being delivered is an important consideration. J Allergy Clin Immunol. 1995;96:288-294.
31. Barry W, O’Callaghan C. The effect of delay, multiple actuations and spacer static charge on the in vitro delivery of budesonide from the Nebuhaler. Br J Clin Pharmacol. 1995;40:76-78.
32. Wildhaber JH, Devadason SG, Eber E, et al. Effect of electrostatic charge, flow, delay and multiple actuations on the in vitro delivery of salbutamol from different small volume spacers for infants. Thorax. 1996;51:985-988.
33. Clark DJ, Lipworth BJ. Effect of multiple actuations, delayed inhalation and antistatic treatment on the lung bioavailability of salbutamol via a spacer device. Thorax. 1996;51:981-984.
34. Stein SW, Sheth P, Hodson PD, Myrdal PB. Advances in metered dose inhaler technology: hardware development. AAPS PharmSciTech. 2014;15:326-338.
35. Chi Lip Kwok PC, Noakes T, Chan HK. Effect of moisture on the electrostatic charge properties of metered dose inhaler aerosols. J Aerosol Sci. 2008;39:211-226. doi:10.1016/j.jaerosci.2007.11.004.
36. Piérart F, Wildhaber JH, Vrancken I, et al. Washing plastic spacers in household detergent reduces electrostatic charge and greatly improves delivery. Eur Respir J. 1999;13:673-678.
To comment on this article, contact rdavidson@uspharmacist.com.





