US Pharm. 2010;35(11):41-48.
Atypical or second-generation antipsychotics (SGAs) have become the mainstay of therapy for treating schizophrenia since their emergence in the 1990s. These medications have largely replaced the use of typical or first-generation antipsychotics (FGAs), as they have demonstrated similar efficacy with minimal to no extrapyramidal symptoms (EPS) at therapeutic doses. However, SGAs have gained much notoriety for causing metabolic derangements including weight gain, dyslipidemia, and hyperglycemia including new-onset type 2 diabetes mellitus (T2DM). These adverse effects are all risk factors for cardiovascular disease, and along with lifestyle and genetic components, contribute to the decreased lifespan of patients with schizophrenia.1 Therefore, it is important to fully assess, and inform patients of, the risks versus the therapeutic benefit of these agents.
Second-Generation Antipsychotics
Clozapine (Clozaril), the first atypical antipsychotic, was approved for use by the FDA in 1989. Currently, there are eight other SGAs indicated for the treatment of schizophrenia: aripiprazole (Abilify), asenapine (Saphris), iloperidone (Fanapt), olanzapine (Zyprexa), paliperidone (Invega), quetiapine (Seroquel), risperidone (Risperdal), and ziprasidone (Geodon). Although widely used in schizophrenia, SGAs are also used for other psychiatric disorders including bipolar disorder, major depressive disorder, and autism.2
SGAs demonstrate their efficacy by antagonizing the D2 receptors in the mesolimbic and mesocortical dopamine tracts in the brain. It is thought that dopaminergic dysfunction of these tracts contributes to the positive symptoms of schizophrenia. In addition, all SGAs except aripiprazole antagonize and express a greater affinity for the 5-HT2A than for the D2 receptor. Aripiprazole is unique in that it exhibits partial agonism of the D2 receptor and antagonism of the 5-HT2A receptor.3 While these are the main receptor systems involved, histaminergic, muscarinic, and adrenergic systems have also been characterized. SGAs differ from each other by the type of the receptors targeted, relative affinity, and agonism/antagonism properties. This is the probable explanation for the variability of side effects among these agents.4
One question that is commonly raised by clinicians is whether there is a dose-dependent relationship between SGA use and the metabolic complications. While the data are somewhat limited, there appears to be a relationship between the dose of clozapine and olanzapine, while aripiprazole, quetiapine, and ziprasidone do not show a relationship, and risperidone has mixed results.5
Weight Gain
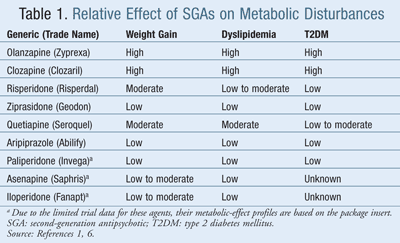
Weight gain associated with SGA use has been well documented in clinical trials. It typically occurs within the first 4 to 12 weeks of treatment and may not reach a plateau.6 Although the mechanism is not fully understood, the relative affinity for the 5-HT2 receptor has been analyzed. Medications that block the 5-HT2 receptor have been linked to increased food intake. Since clozapine and olanzapine appear to have the greatest affinity for the 5-HT2 receptor compared to other SGAs, this may partially explain why they are the worst offenders of weight gain.7 TABLE 1 provides a review of the relative metabolic side effects of the SGAs.
In a meta-analysis by Allison et al, patients taking clozapine (mean dose 468 mg/day) and olanzapine (mean dose 13.2 mg/day) at 10 weeks experienced weight gain from baseline of 4.45 kg and 4.15 kg, respectively.8 Patients taking risperidone (mean dose 5.9 mg/day) had an average weight gain of 2.10 kg.
During the CATIE trial (Clinical Antipsychotic Trials of Intervention Effectiveness), 1,493 patients were randomly assigned to receive olanzapine (7.5-30 mg/day), perphenazine (8-32 mg/day), quetiapine (200-800 mg/day), risperidone (1.5-6.0 mg/day), or ziprasidone (40-160 mg/day) for up to 18 months.9 Patients taking olanzapine gained an average of 0.9 kg per month and a greater proportion of these patients gained 7% or more of their body weight at 18 months or time of discontinuation compared to the other agents (30% vs. 7%-16%, P <.001). Patients taking quetiapine and risperidone showed minimal weight gain (0.5 kg and 0.36 kg increase from baseline, respectively), and those taking ziprasidone experienced weight loss (-0.72 kg).9
The Comparison of Atypicals in First Episode Psychosis (CAFE) study analyzed the metabolic side effects of 400 patients randomized to receive olanzapine, quetiapine, or risperidone over 52 weeks.10 At 52 weeks, mean weight gain from baseline was significantly higher in patients receiving olanzapine (24.2 ± 1.9 lb) versus risperidone (14.0 ± 1.9 lb) and quetiapine (12.1 ± 1.8 lb), P <.001.Similarly, an open-label randomized trial by Kahn et al comparing olanzapine, quetiapine, and ziprasidone showed mean weight gain of 13.9, 10.5, and 4.8 lb over 1 year from baseline, respectively.11 A meta-analysis by Jones et al analyzed the relative weight gain among other adverse effects of paliperidone versus all other SGAs except ziprasidone, asenapine, and iloperidone.12 The incidence of clinically significant weight gain (defined as ³7% from baseline) for those treated with paliperidone for 4 to 8 weeks was 1.75%, significantly less than any of the other agents studied.
The implications of weight gain are twofold. First, those who are overweight are at an increased risk for cardiovascular mortality. Second, patients with schizophrenia have poor adherence with medication use. Therefore, those who are aware of this side effect or experience weight gain firsthand may be more reluctant to maintain adherence, resulting in a decreased quality of life.6
Type 2 Diabetes Mellitus
The prevalence of T2DM in patients with schizophrenia is estimated to be three times higher than the general population even if they are drug naïve, suggesting a genetic predisposition.4 The mechanism of SGA-induced T2DM is likely multifactorial, with some studies suggesting a relationship between serotonergic antagonism and a reduction in insulin sensitivity. In addition, the case can be made that the diabetes is secondary to SGA-induced weight gain. However, in many case reports, hyperglycemia tended to occur within 6 weeks after the start of treatment with an SGA, and most new-onset cases were reversible upon discontinuation of therapy, suggesting an independent drug-related mechanism.6
Leslie and Rosenheck studied the incidence of new-onset T2DM in 56,849 patients in the Veterans Administration (VA)for at least 1 year.13 Patients were included if they were on a stable antipsychotic regimen (clozapine, olanzapine, risperidone, quetiapine, or conventional antipsychotic medications). Approximately 7% of the patients were diagnosed with diabetes at follow-up. While this study was not limited to use of SGAs, it highlights the association of new-onset diabetes. The hazard ratios for clozapine and olanzapine were significantly increased at 1.57 (confidence interval [CI], 1.31-1.89) and 1.15 (CI, 1.07-1.24), respectively, while there was a nonsignificant increase in the risperidone and quetiapine group.13
In phase I of the CATIE trial, olanzapine was associated with a significant increase in mean HbA1C (A1C) from baseline of 0.41% ± 0.09%, P = .01. Quetiapine and risperidone had a much lower increase of 0.05% ± 0.05% and 0.08% ± 0.04%, respectively, while there was a reduction in mean A1C in patients treated with ziprasidone of -0.10% ± 0.14%.9 Similar to weight gain, clozapine and olanzapine appear to have the greatest risk for diabetes as evidenced by epidemiologic studies and clinical trials.
Dyslipidemia
Lipid abnormalities have been shown to occur in patients treated with clozapine, olanzapine, quetiapine, and risperidone.14 These changes are largely related to concomitant weight gain. Therefore, agents associated with the greatest weight gain may have a greater propensity to cause dyslipidemia.4 A 14-week, double-blind clinical trial by Lindenmayer et al assessed changes in total cholesterol and blood glucose in 157 patients randomized to clozapine, olanzapine, risperidone, or haloperidol.15 There was a significant increase in mean total cholesterol at 8 weeks from baseline of 14.7 mg/dL (P <.02) and 12.3 mg/dL (P <.02) in patients taking clozapine and olanzapine, respectively. At 14 weeks, patients taking olanzapine had an increase in total mean cholesterol of 20.1 mg/dL (P <.02), while the increase was not significant in those taking risperidone or clozapine.15
In addition to changes in cholesterol, some SGAs cause hypertriglyceridemia, which is an independent risk factor for cardiovascular disease.6 In a retrospective analysis, mean triglyceride levels were significantly increased by 34%, 38%, and 19% from baseline in patients taking clozapine, olanzapine, and risperidone, respectively.16 Furthermore, a recent review by Chaggar et al concluded that olanzapine has the worst effect on one’s lipid panel, while quetiapine and clozapine also have a modest effect, and aripiprazole and ziprasidone appear to have a neutral to low effect.14
Preventing, Screening, and Managing the Metabolic Complications
Considering the metabolic abnormalities that SGAs may cause, proper prevention, screening, and management are essential to achieve optimum patient care. While the metabolic complications of SGAs are apparent, proper screening is lacking. A study by Morrato et al found that baseline lipid and serum glucose screenings in patients taking SGAs were low (10% and 27%, respectively) and that lipid screening for patients taking SGAs was only slightly higher (1.7%, P = .02) than that of the control group.17 Further, rates of serum glucose screening were the same in both the experimental and the control groups.17 To aid practitioners in addressing the issue of SGA-induced metabolic complications, a consensus panel was assembled in 2003 to develop treatment guidelines.18,19
Recommended Monitoring Protocol
Prior to the initiation of SGA therapy, patients should be screened for diabetes, hypertension, dyslipidemia, and family history of cardiovascular disease. Weight, waist circumference, blood pressure, fasting plasma glucose, and fasting lipid panel should be encompassed in the screening.19 TABLE 2 summarizes the consensus panel’s routine monitoring recommendations for individuals taking SGAs.19

Prevention Strategies
The consensus statement recommends physical activity and nutrition counseling for all patients who are starting SGAs that have been associated with significant weight gain.18,19 Additionally, these strategies are recommended in all patients who are overweight or obese prior to the initiation of SGA therapy.18,19 If a patient’s total body weight increases ³5% from baseline after the initiation of an SGA, consideration should be given to switching to an alternative SGA associated with less weight gain.19
Patients already overweight who have either diabetes or risk factors for diabetes should be treated initially with aripiprazole or ziprasidone therapy when possible.18,19 These agents have been shown to cause less weight gain, dyslipidemia, and glucose intolerance/T2DM than other available SGAs. Similarly, patients who experience dyslipidemia, hyperglycemia, or glucose intolerance while taking an SGA should be switched to an alternative agent.3,18-20 When switching between SGAs, the therapeutic benefit of therapy should always be balanced with the potential metabolic complications. This is especially important to consider when treating patients who are refractory to a specific SGA, such as those patients taking clozapine. In the event that one SGA is discontinued and another initiated, it is recommended that the drugs be cross-titrated rather than abruptly discontinued.19
Management Strategies
Prior to the initiation of medications to attenuate the metabolic side effects of SGA therapy, clinicians should encourage nonpharmacologic therapy and lifestyle modifications. Many agents have been analyzed in managing the metabolic side effects of SGA therapy. While most have not demonstrated a significant clinical benefit, metformin has shown some promise. A randomized, controlled trial of 128 first-episode schizophrenia patients found that metformin 750 mg/day in combination with lifestyle modifications was superior to lifestyle modifications or metformin 750 mg/day alone at reducing body mass index, insulin resistance index, and waist circumference.21 In a meta-analysis of 32 randomized, double-blind, placebo-controlled studies examining 15 different medications used to attenuate weight gain, metformin had the greatest average weight loss of 2.94 kg (95% CI, -4.89 to -0.99) over an average of 13 weeks.22 In situations where a patient is to remain on a specific SGA despite metabolic complications, the health care provider may consider initiating metformin 750 mg/day in combination with lifestyle modifications.21,23
Prior to any treatment of metabolic complications, the use of an alternative SGA should be considered.18,24 In patients taking SGAs, when metabolic abnormalities progress to a disorder, the patient’s goals should be targeted and treated according to the appropriate guidelines specific to that disorder.24 For example, when a patient is diagnosed with diabetes while taking an atypical antipsychotic, he or she should be targeted to goal and treated according to the guidelines established by the American Diabetes Association (ADA) and/or the American Association of Clinical Endocrinologists (AACE).
Conclusion
While the introduction of SGAs has provided efficacious treatment options with reduced incidences of EPS, the metabolic complications can be problematic. When treating with SGAs, clinicians must consider patient-specific factors, balancing both the potential risk and the therapeutic benefit of each individual agent. Significant effort should be devoted to ensuring that patients receive proper screening prior to initiation and throughout the course of SGA therapy.
REFERENCES
1. Hasnain M, Fredrickson SK, Vieweg WV, et al. Metabolic syndrome associated with schizophrenia and atypical antipsychotics. Curr Diab Rep. 2010;10:209-216.
2. Lexi-Comp Online. Hudson, OH: Lexi-Comp, Inc.; 2010. www.lexi.com. Accessed August 17, 2010.
3. Crismon ML, Argo TR, Buckley PF. Schizophrenia. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacother-apy: A Pathophysiologic Approach. 7th ed. New York, NY: McGraw Hill; 2008:1099-1122.
4. Nasrallah HA. Atypical antipsychotic-induced metabolic side effects: insights from receptor-binding profiles. Mol Psychiatry. 2008;13:27-35.
5. Simon V, van Winkel R, De Hert M. Are weight gain and metabolic side effects of atypical antipsychotics dose dependent? A literature review. J Clin Psychiatry. 2009;70:1041-1050.
6. Tschoner A, Engl J, Laimer M, et al. Metabolic side effects of antipsychotic medication. Int J Clin Pract. 2007;61:1356-1370.
7. Reynolds GP, Kirk SL. Metabolic side effects of antipsychotic drug treatment—pharmacological mechanisms. Pharmacol Ther. 2010;125:169-179.
8. Allison DB, Mentore JL, Heo M, et al. Antipsychotic-induced weight gain: a comprehensive research synthesis. Am J Psychiatry. 1999;156:1668-1696.
9. Lieberman JA, Stroup TS, McEvoy JP, et al. Effectiveness of antipsychotic drugs in patients with chronic schizophrenia. N Engl J Med. 2005;353:1209-1223.
10. Patel JK, Buckley PF, Woolson S, et al. Metabolic profiles of second-generation antipsychotics in early psychosis: findings from the CAFE study. Schizophr Res. 2009;111: 9-16.
11. Kahn RS, Fleischhacker WW, Boter H, et al. Effectiveness of antipsychotic drugs in first-episode schizophrenia and schizophreniform disorder: an open randomised clinical trial. Lancet. 2008;371:1085-1097.
12. Jones MP, Nicholl D, Trakas K. Efficacy and tolerability of paliperidone ER and other oral atypical antipsychotics in schizophrenia. Int J Clin Pharmcol Ther. 2010;48:383-399.
13. Leslie DL, Rosenheck RA. Incidence of newly diagnosed diabetes attributable to atypical antipsychotic medications. Am J Psychiatry. 2004;161:1709-1711.
14. Chaggar PS, Shaw SM, Williams SG. Effect of antipsychotic medications on glucose and lipid levels. J Clin Pharmacol. 2010 Apr 21 [Epub ahead of print].
15. Lindenmayer JP, Czobor P, Volavka J, et al. Changes in glucose and cholesterol levels in patients with schizophrenia treated with typical or atypical antipsychotics. Am J Psychiatry. 2003;160:290-296.
16. Wirshing DA, Boyd JA, Meng LR, et al. The effects of novel antipsychotics on glucose and lipid levels. J Clin Psychiatry. 2002;63:856-865.
17. Morrato EH, Druss B, Hartung DM, et al. Metabolic testing rates in 3 state Medicaid programs after FDA warnings and ADA/APA recommendations for second-generation antipsychotic drugs. Arch Gen Psychiatry. 2010;67:17-24.
18. Kiraly B, Gunning K, Leiser J. Primary care issues in patients with mental illness. American Family Physician. 2008;78:355-362.
19. American Diabetes Association; American Psychiatric Association; American Association of Clinical Endocrinologists; North American Association for the Study of Obesity. Consensus development conference on antipsychotic drugs and obesity and diabetes. Diabetes Care. 2004;27:596-601.
20. Lamberti JS, Olson D, Crilly JF, et al. Prevalence of the metabolic syndrome among patients receiving clozapine. Am J Psychiatry. 2006;163:1273-1276.
21. Wu RR, Zhao JP, Jin H, et al. Lifestyle intervention and metformin for treatment of antipsychotic-induced weight gain: a randomized controlled trial. JAMA. 2008;299:185-193.
22. Maayan L, Vakhrusheva J, Correll CU. Effectiveness of medications used to attenuate antipsychotic-related weight gain and metabolic abnormalities: a systemic review and meta analysis. Neuropsychopharmacology. 2010;35:1520-1530.
23. Dixon L, Perkins D, Calmes C. Guideline watch (September 2009): practice guideline for the treatment of patients with schizophrenia. In: American Psychiatric Association Practice Guidelines for the Treatment of Psychiatric Disorders. Arlington, VA: American Psychiatric Association; 2004.
24. Hasnain M, Vieweg WV, Fredrickson SK, et al. Clinical monitoring and management of the metabolic syndrome in patients receiving atypical antipsychotic medications. Prim Care Diab. 2009;3:5-15.
To comment on this article, contact rdavidson@uspharmacist.com.






