US Pharm. 2016;41(10):26-29.
ABSTRACT: Following the legislation that allowed the FDA to approve biosimilars in the United States, biological products that are similar to the reference product in terms of safety, purity, and potency are gradually entering the market. To date, only four biosimilars have been approved in the U.S. (Zarxio, Inflectra, Erelzi, and Amjevita), although many agents are currently undergoing trials and may soon become available. Pharmacists can facilitate the use of these products by engaging further with prescribers, patients, insurance companies, and the FDA.
Biological products have become a large part of healthcare delivery in the United States. Even though they account for <1% of all prescriptions dispensed in the U.S., they constitute 28% of prescription-drug spending.1 The number of billable therapeutic biologics increased from 80 in 2013 to about 96 in 2014, including older medications such as collagenase that were approved as far back as 1965.2 It is expected that the biologics market will increase at a rate of more than 20% per year, and that by 2025 more than 70% of New Drug Approvals would be biological products.3 The increasing use of these products underlines the need for cost control, especially for those agents that have enjoyed a lengthy period of exclusive use.
Biologics are commonly used to treat autoimmune disorders, inflammatory diseases, and cancers. They can also be used in diagnostics; e.g., to predict genetic propensity to conditions such as Parkinson’s disease.4
Defining Biosimilars
Biosimilars are one of two types of biological products that have been designed to provide an alternative to currently marketed biologics, the other being known as interchangeables. An interchangeable biological product is biosimilar to an FDA-approved reference product and meets additional standards for interchangeability. An interchangeable biological product may be substituted for the reference product by a pharmacist without the intervention of the healthcare provider who prescribed the reference product.5
The Affordable Care Act made way for these biological products in 2010, allowing the FDA the authority to approve and regulate biosimilar products for marketing in the U.S.5 The Biologics Price Competition and Innovation Act of 2009 (BCPI Act), which was a part of this legislation, created a new abbreviated approval pathway under the Public Health Service Act for biological products that are shown to be biosimilar to or interchangeable with an FDA-approved biological product known as a reference product. The BCPI Act also provides for a 12-year exclusivity period from the date of first licensing of the reference product before approval of any biosimilar can occur. Furthermore, the FDA will not accept or consider potential applications for biosimilars until the fifth year after the approval of the reference product.5
In order for a biosimilar to be approved, it must have the same mechanism of action, route of administration, dosage form, and strength as the reference product. It can only be licensed for use for the same indications and conditions that have been approved for the reference product. Furthermore, there must be no clinically meaningful differences between the biosimilar and the reference product in terms of the safety, purity, and potency of the product. There may, however, be minor differences in clinically inactive components.6 The manufacturing facilities of the biosimilar must also meet FDA standards.
Express Scripts estimated that the U.S. could save $250 billion between 2014 and 2024 if 11 of the likeliest biosimilars reached the market.7 This article discusses the biosimilars that have been approved by the FDA as well as some that may obtain approval in the near future.
Approved U.S. Biosimilars
As of September 2016, only four biosimilars had been granted approval from the FDA: Zarxio (biosimilar to Neupogen), Inflectra (biosimilar to Remicade), Erelzi (biosimilar to Enbrel), and Amjevita (biosimilar to Humira).8-16 In contrast, biosimilars have been available in Europe since 2006.7
Zarxio (Biosimilar to Neupogen): Zarxio (filgrastim-sndz) was approved in March 2015, making it the first biosimilar to received U.S. approval.8 It is manufactured by Sandoz and is the biosimilar to Neupogen (filgrastim by Amgen). Indicated for the treatment of neutropenia, it is approved for use in patients with9:
- Cancer receiving myelosuppressive chemotherapy
- Acute myeloid leukemia receiving induction or consolidation chemotherapy
- Cancer undergoing bone marrow transplantation
- Ongoing autologous peripheral blood progenitor cell collection and therapy
- Severe chronic neutropenia.
In line with the FDA requirements for the approval of biosimilars, Zarxio is available in the same formulations as Neupogen, prefilled syringes of 300 mcg/0.5 mL and 480 mcg/0.8 mL.9 The recommended starting dosage of Neupogen is 5 mcg/kg/day‚ administered as a single daily SC injection‚ by short IV infusion (15 to 30 minutes)‚ or by continuous IV infusion.9 Zarxio should be administered following the same dosing schedule. It should not be administered to patients with a history of serious allergic reactions to human granulocyte colony-stimulating factors (G-CSFs) such as filgrastim or pegfilgrastim products.9 Patients using Zarxio may typically complain of aching in the bones or muscles and redness, swelling, or itching at the injection site.9 More severe adverse effects include rupture of the spleen; serious allergic reactions leading to rashes, shortness of breath, wheezing, and/or swelling around the mouth and eyes; fast pulse and sweating; and acute respiratory distress syndrome.9
Zarxio saves patients about 15%, with the prices set at $275.66 for a 300-mcg syringe and $438.98 for a 480-mcg syringe as compared to Neupogen at $324.30 and $516.45, respectively.10
Inflectra (Biosimilar to Remicade): In April 2016, the FDA approved the second biosimilar in the U.S., Inflectra (infliximab-dyyb), a biosimilar to Janssen Biotech’s Remicade (infliximab), which was originally licensed in 1998.11 Inflectra is a tumor necrosis factor (TNF) blocker approved for the following indications12:
- Moderate-to-severe Crohn’s disease in adult and pediatric patients aged ≥6 years; Inflectra has not been studied in children <6 years of age
- Moderate-to-severe ulcerative colitis
- Moderate-to-severe rheumatoid arthritis in combination with methotrexate
- Active ankylosing spondylitis
- Active psoriatic arthritis
- Chronic severe plaque psoriasis.
Inflectra is available as an injection of 100 mg of infliximab-dyyb in a 20-mL vial for IV infusion. The recommended dosing is 5 mg/kg given as three injections at 0, 2, and 6 weeks, then every 8 weeks for all indications except in rheumatoid arthritis, whereby the recommended dose is 3 to 10 mg/kg up to every 4 weeks, and in active ankylosing spondylitis, where the maintenance regimen is 5 mg/kg every 6 weeks thereafter.12
Inflectra is contraindicated at doses higher than 5 mg/kg in patients with moderate-to-severe heart failure or a known hypersensitivity to infliximab products. Patients commonly experience respiratory infections such as sinus infections, sore throat, headache, coughing, and stomach pains. Infusion reactions such as fever, chills, chest pains, low or high blood pressure, shortness of breath, rash, and itching may occur 2 hours after infusion. Other serious adverse effects may include liver injury, blood problems, lupuslike syndrome, psoriasis, and, in rare cases, nervous system disorders.12
The product labeling for Inflectra includes a black box warning relating to the increased risk of serious infections that can lead to hospitalizations or death, including tuberculosis, bacterial sepsis, invasive fungal infections, and infections due to other opportunistic infections. Additionally, patients may develop lymphoma and other malignancies that may be fatal in children and adolescent patients treated with TNF-blocking agents.12
Inflectra is manufactured by Celltrion for Hospira.12 When contacted, Hospira stated that it was not able to release the price of Inflectra at this point.
Erelzi (Biosimilar to Enbrel): On August 30, 2016, the FDA approved the TNF blocker etanercept-szzs (Erelzi, Sandoz), which is a biosimilar to etanercept (Enbrel, Amgen).13 Erelzi is approved for14:
- Rheumatoid arthritis
- Polyarticular juvenile idiopathic arthritis (JIA) in patients aged ≥2 years
- Psoriatic arthritis
- Ankylosing spondylitis
- Plaque psoriasis.
The recommended dosage for patients with rheumatoid arthritis and psoriatic arthritis is 50 mg SC once weekly with or without methotrexate. In patients with JIA who weigh >63 kg, the normal dose is 0.8 mg/kg weekly, with a maximum of 50 mg per week. The most commonly reported adverse effects are infections and injection-site reactions. The use of Erelzi is contraindicated in sepsis.14
Amjevita (Biosimilar to Humira): Amjevita (adalimumab-atto, Amgen) was approved by the FDA as a biosimilar to AbbVie’s Humira (adalimumab) on September 23, 2016.15 Amjevita is indicated for the treatment of 15,16:
- Moderate-to-severe rheumatoid arthritis
- Active psoriatic arthritis
- Active ankylosing spondylitis
- Moderate-to-severe Crohn’s disease
- Moderate-to-severe ulcerative colitis
- Moderate-to-severe plaque psoriasis
- Polyarticular JIA in children aged ≥4 years.
The recommended dosage for patients with rheumatoid arthritis, psoriatic arthritis, and ankylosing spondylitis is 40 mg SC every other week.16
Amjevita has been associated with infections and injection-site reactions. Like its reference product, Amjevita has a black box warning for lymphoma and other malignancies. It is important that pharmacists dispense the agent with a patient medication guide that describes important information about its uses and risks.15,16
Future Biosimilars
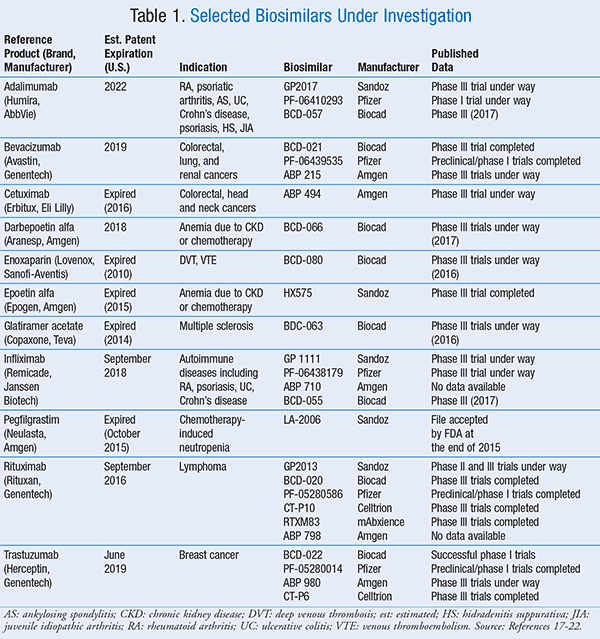
With a number of biological products soon to lose patent protection, more biosimilars are expected to enter the market. TABLE 1 lists some of the trials being conducted on biosimilars, and if successfully completed, these products may become available in the near future.17-22
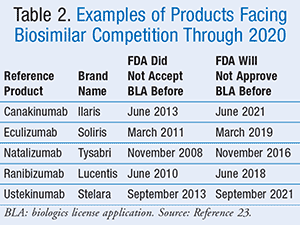
A number of other biologics face the prospect of biosimilar competition based upon the exclusivity conditions outlined by the Affordable Care Act. The exclusivity period lasts for 12 years from the date of first licensure. Some of these products whose exclusivity periods are about to expire and will soon face competition from biosimilars are listed in TABLE 2.23
How Does This Affect Pharmacists?
As with any new developments, there are likely to be some challenges, particularly since biosimilars, unlike generics, cannot be automatically substituted for branded products. While biosimilars mean savings for patients, pharmacists may need to facilitate the use of these products by engaging further with prescribers, insurance companies, and the FDA. This is beneficial on many fronts, including:
1) Insurance claims processing: Insurance companies may only cover a biosimilar, while a prescriber may write the reference product. In this case, the prescriber will have to be contacted to change the prescription so that payment can be processed.
2) Postmarketing data: Since the FDA approval process for biosimilars is accelerated, postmarketing data are vital for the safe and effective use of these products. Pharmacists are therefore encouraged to record adverse events and report them to either the FDA or prescribers.
3) Educating practitioners: Pharmacists can provide useful information on the efficacy and safety of biosimilars, making them more comfortable with changing from a branded innovator product to a biosimilar. Since it is important that patients do not switch back and forth between products, pharmacists can help in selecting the most appropriate biosimilar for a particular patient.
Since there are new developments in this field, pharmacists are encouraged to keep abreast of these changes by tracking FDA policies and state regulations. Pharmacists who have the most up-to-date knowledge will instill confidence in patients when discussing these new agents. Overall, pharmacists can provide an objective perspective and educate prescribers and patients regarding biosimilars and their availability.
REFERENCES
1. Sarpatwari A, Avorn J, Kesselheim A. Progress and hurdles for follow-on biologics. N Engl J Med. 2015;372:2380-2382.
2. Center for Drug Evaluation and Research. CDER therapeutic biologic products. CDER billable biologic product list; 2014. www.fda.gov/downloads/ForIndustry/UserFees/PrescriptionDrugUserFee/UCM164641.pdf. Accessed April 23, 2016.
3. Erickson BE. Untangling biosimilars. Chem Eng News. 2010;88:25-27.
4. Morrow T, Felcone LH. Defining the difference: what makes biologics unique. Biotechnol Health. 2004;1(4):24-29.
5. FDA overview of biosimilar products. February 18, 2016. http://fdabiosimilars.e-paga.com. Accessed May 10, 2016.
6. FDA. Information for healthcare professionals (biosimilars). www.fda.gov/Drugs/DevelopmentApprovalProcess/HowDrugsareDevelopedandApproved/ApprovalApplications/TherapeuticBiologicApplications/Biosimilars/ucm241719.htm. Accessed May 10, 2016.
7. Reuters. Novartis launches first U.S. ‘biosimilar’ drug at 15 percent discount. September 3, 2015. www.reuters.com/article/us-novartis-drug-idUSKCN0R30C220150903. Accessed May 10, 2016.
8. FDA approves first biosimilar product Zarxio. March 6, 2015. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm436648.htm. Accessed May 11, 2016.
9. Sandoz. Zarxio product information. www.zarxio.com/info/hcp/about-zarxio.jsp. Accessed May 11, 2016.
10. Hirschler B. Novartis launches first biosimilar in US. Pharmacoecon Outcomes News. 2015;736(1):5.
11. FDA approves Inflectra, a biosimilar to Remicade. April 5, 2016. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm494227.htm. Accessed May 12, 2016.
12. Inflectra (infliximab-dyyb) prescribing information. Lake Forest, IL: Hospira; April 2016.
13. FDA approves Erelzi, a biosimilar to Enbrel. August 30, 2016. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm518639.htm. Accessed September 1, 2016.
14. Erelzi (etanercept-szzs) prescribing information. Princeton, NJ: Sandoz Inc; August 2016.
15. FDA approves Amjevita, a biosimilar to Humira. September 23, 2016. www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm522243.htm. Accessed September 23, 2016.
16. Amjevita (adalimumab-atto) prescribing information. Thousand Oaks, CA: Amgen Inc; September 2016.
17. Rugo HS, Linton KM, Cervi P, et al. A clinician’s guide to biosimilars in oncology. Cancer Treat Rev. 2016;21;46:73-79.
18. Sandoz. Sandoz: Unrivalled biosimilars pipeline. www.sandoz-biosimilars.com/en/aboutus/biosimilars-pipeline.shtml. Accessed May 17, 2016.
19. Pfizer Inc. Pfizer biosimilars. www.pfizerbiosimilars.com/pfizer-pipeline. Accessed May 17, 2016.
20. Amgen Inc. Amgen pipeline. www.amgenpipeline.com/~/media/amgen/full/www-amgenpipeline-com/charts/amgenpipelinedownload.ashx. Accessed May 17, 2016.
21. Biocad. Biocad biosimilars. http://biocadglobal.com/pipelines/Biosimilar. Accessed May 17, 2016.
22. Amgen. 2015 Trends in Biosimilars Report. www.amgenbiosimilars.com/~/media/amgen/full/www-amgenbiosimilars-com/downloads/2015_trends_in_biosimilars_report.ashx?la=en. Accessed May 20, 2016.
23. Bourgoin AF, Nuskey B. White paper: an outlook on U.S. biosimilar competition. Drugs Today (Barc). 2013;49(6):399-410.
To comment on this article, contact rdavidson@uspharmacist.com.






