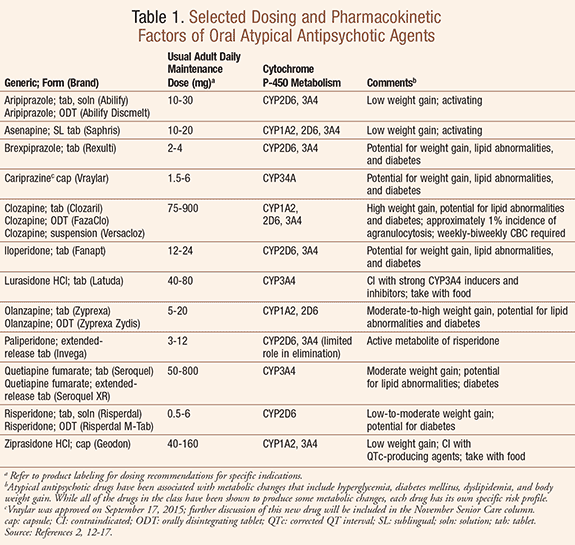
Brexpiprazole (Rexulti, Otsuka Pharmaceutical Co., Ltd.), an orally administered atypical antipsychotic agent, received FDA approval in July 2015 for two indications: 1) adjunctive treatment of major depressive disorder (MDD), and 2) treatment of schizophrenia. As with all other atypical antipsychotic agents (TABLE 1), brexpiprazole is not approved for the treatment of patients with dementia-related psychosis; the elderly with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death (see below, U.S. Boxed Warning).1 This article will review the drug’s mechanism, dosing, and safety and efficacy data, and consider the use of brexpiprazole and other antipsychotics in the vulnerable geriatric population.
Mechanism of Action
According to the manufacturer, the mechanism of action of brexpiprazole in the treatment of MDD or schizophrenia is unknown; its efficacy may be mediated through a combination of partial agonist activity at dopamine D2 and serotonin 5-HT1A receptors and antagonist activity at serotonin 5-HT2A receptors.1 As a point of reference with regard to antipsychotic drug development, the addition of serotonin antagonism to dopamine antagonism (classic neuroleptic mechanism) is thought to improve negative symptoms of psychosis and reduce the incidence of extrapyramidal side effects.2
Form and Recommended Dosing
Currently, brexpiprazole is available in round oral tablets in 0.25 mg (light brown), 0.5 mg (light orange), 1 mg (light yellow), 2 mg (light green), 3 mg (light purple), and 4 mg (white) strengths. Patients should be instructed that brexpiprazole can be administered with or without food.
Adjunctive Treatment of MDD: The recommended starting dose is 0.5 mg or 1 mg orally once daily; titrate to 1 mg once daily, then to the target dosage of 2 mg once daily. Increases should occur at weekly intervals based on clinical response and tolerability. The maximum recommended daily dosage is 3 mg. Reassess the patient periodically to determine the continued need and appropriate dosage for treatment.
Treatment of Schizophrenia: The recommended starting dose is 1 mg orally once daily on days 1 to 4. The recommended target dosage is 2 mg to 4 mg once daily. Titrate to 2 mg once daily on day 5 through day 7, then to 4 mg on day 8 based on clinical response and tolerability. The maximum recommended daily dosage is 4 mg. Reassess the patient periodically to determine the continued need and appropriate dosage for treatment.
Pharmacists should become familiar with additional dosage recommendations outlined in the full prescribing information for adjustments for hepatic impairment and renal impairment, and dosage modifications that are necessary for CYP2D6 poor metabolizers and for concomitant use with CYP inhibitors or inducers.
Clinical Efficacy Studies
Clinical trials of brexpiprazole support its efficacy as follows: At the recommended target dosage of 2 mg/day as an adjunct to antidepressant medication for the treatment of MDD (based on scoring of the Montgomery-Åsberg Depression Rating Scale [MADRS], which measures the degree of depressive symptomatology); and at the recommended target dosage of 2-4 mg/day for the treatment of schizophrenia (scoring of the Positive and Negative Syndrome Scale [PANSS], which measures positive symptoms, negative symptoms, and general psychopathology of schizophrenia).1 Head-to-head comparisons with other available agents among patients with MDD and schizophrenia—including geriatric patients >65 years—are needed.3 The efficacy clinical studies of brexpiprazole were conducted without including any patients aged >65 years; thus, a determination could not be made as to whether geriatric patients in this age bracket respond differently than younger individuals with regard to efficacy.
Safety: Clinical Data
Adverse Effects (AEs): The most commonly encountered AEs noted in the product labeling were akathisia and weight gain; other reported AEs include, but are not limited to, somnolence, tremor, dizziness, anxiety, and restlessness.1,3 Pharmacists should report suspected adverse reactions to the FDA (1-800-FDA-1088; www.fda.gov/medwatch).
Geriatric Population: The selection of a dosage for an elderly patient should be done cautiously; the manufacturer recommends usually starting the dose at the low end of the dosing range, which would reflect the greater frequency of decreased hepatic, renal, and cardiac function, concomitant diseases, and other drug therapy in the geriatric population. The pharmacokinetics of once-daily oral administration of brexpiprazole (up to 3 mg/day for 14 days) as an adjunct therapy in the treatment of elderly subjects (70 to 85 years old, N=11) with MDD were comparable to those observed in adult subjects with MDD, based on the results of a safety, tolerability, and pharmacokinetics trial.1
U.S. Boxed Warning
Brexpiprazole comes with a black box warning, in the United States, of increased risk of mortality in elderly patients with dementia-related psychosis and a potential increase in the risk of suicidal thoughts and behaviors in patients aged <24 years. Elderly patients with dementia-related psychosis treated with antipsychotic drugs are at an increased risk of death; brexpiprazole is not approved for the treatment of patients with dementia-related psychosis. Antidepressants increased the risk of suicidal thoughts and behaviors in patients aged <24 years in short-term studies. The manufacturer advises that patients be monitored closely for clinical worsening and for emergence of suicidal thoughts and behaviors. Further, the safety and efficacy of brexpiprazole has not been established in pediatric patients.
Advocacy for Patients and Caregivers
In light of the plethora of drug information available to the general public through a variety of sources, including the easily accessible Internet, pharmacists are needed more than ever to interpret reliable and reputable information in their role as patient and caregiver advocate for the benefit and protection of the elderly. It is the responsibility of pharmacists to familiarize themselves with the FDA-approved indications for a particular medication, to be fully aware of the populations in whom efficacy clinical trials were conducted, to stay up-to-date on any new indications by referring to updated manufacturer’s package labeling and current medical literature, and, with a critical eye, to stay alert and sensitive to pharmaceutical product marketing—including educational presentations—that may imply that a drug has been found to be specifically effective in the elderly population in whom it has not been tested.4
Of particular note is that while safety testing of a drug may have been conducted in the elderly (those aged >65 years), efficacy clinical trials may not have been conducted in this population, and therefore, clear guidance regarding effective use in the geriatric specialty population as compared with younger individuals is not available.
Additional Geriatric Considerations
In the U.S., many elderly patients receive antipsychotic medications for nonpsychotic behavior.2 In light of the above-mentioned black box warning, brexpiprazole, as with other atypical antipsychotic agents, should be avoided for behavioral problems of dementia.1,5 However, in the absence of approved agents or a convincing clinical-trials evidence base for treating patients with behavioral symptoms, a general therapeutic approach is recommended.2,6
• Before initiating antipsychotic medication, investigate any reversible cause (e.g., anticholinergic agent, infection, source of pain, change in the environment)
• Identify target symptoms for treatment before therapy begins; target symptoms include wandering, restlessness, physical aggression, screaming, disinhibition, misidentification, sleep disturbances, delusions, hallucinations, and, very frequently, depression
• Drug therapies for the identified target symptoms should be slowly initiated and slowly advanced
• Periodic attempts should be made to wean the patient off the medication intervention when symptoms are controlled.
Updated Beers Criteria
2015 Beers Criteria for Potentially Inappropriate Medication Use in Older Adults, an update to the 2012 list, addresses medications to avoid in delirium and in dementia/cognitive impairment, including7-9:
• The addition of antipsychotics to the list of drugs to avoid in delirium
• Updated language with regard to avoiding the use of antipsychotics for behavioral problems of dementia and/or delirium
• Use of antipsychotics in the elderly only if nonpharmacologic options have failed or are not possible AND if the patient poses the threat of harm to self or others.
Patient Counseling and Medication Guide
The brexpiprazole (Rexulti) Medication Guide, also approved by the FDA in July 2015, is a helpful educational tool when counseling patients and caregivers regarding associated potential AEs. These AEs, which can also occur with other atypical antipsychotic agents, include metabolic problems (e.g., weight gain, hyperglycemia, diabetes mellitus, dyslipidemia), cerebrovascular problems, neuroleptic malignant syndrome (NMS), and tardive dyskinesia.1,10,11 Teaching patients how to avoid becoming overheated and dehydrated, particular risks in the elderly, are outlined in this teaching aid as well.
Conclusion
Many elderly patients receive antipsychotic medications for inappropriate nonpsychotic behavior; further, use of antipsychotics in elderly patients with dementia is associated with an increased risk of mortality and cerebrovascular accidents. With a new atypical antipsychotic agent recently FDA-approved for adjunctive treatment of MDD and for the treatment of schizophrenia, pharmacists as clinicians and educators to patients, caregivers, and healthcare providers can guide appropriate use—and caution against inappropriate use—of this agent in the geriatric specialty population.
REFERENCES
1. Rexulti full prescribing information. Tokyo, Japan: Otsuka Pharmaceutical Co., Ltd; July 2015. www.otsuka-us.com/Products/Documents/Rexulti.PI.pdf. Accessed September 18, 2015.
2. Semla TP, Beizer JL, Higbee MD. Geriatric Dosage Handbook. 19th ed. Hudson, OH: Lexicomp; 2014:669-670,1475-1476.
3. Citrome L. Brexpiprazole for schizophrenia and as adjunct for major depressive disorder: a systematic review of the efficacy and safety profile for this newly approved antipsychotic - what is the number needed to treat, number needed to harm and likelihood to be helped or harmed? Int J Clin Pract. 2015;69(9):978-997.
4. Brill S. The miracle industry: America’s most admired lawbreaker. Huffington Post. http://highline.huffingtonpost.com/miracleindustry/americas-most-admired-lawbreaker. Accessed September 15, 2015.
5. Reuben DB, Herr KA, Pacala JT, et al. Geriatrics at Your Fingertips: 2015. 17th Edition. New York, NY: American Geriatrics Society; 2015:275-276.
6. Farlow MR. Alzheimer’s disease. In: Fillit HM, Rockwood K, Woodhouse K, eds. Brocklehurst’s Textbook of Geriatric Medicine and Gerontology. 7th ed. Philadelphia, PA: Saunders Elsevier; 2010:411-420.
7. Fick D, Beizer J, Brandt N, et al. American Geriatrics Society (AGS) 2015 Beers Criteria for potentially inappropriate medication use in older adults. Presented at: American Geriatrics Society Annual Meeting; May 16, 2015; National Harbor, MD.
8. Aparasu RR, Chatterjee S, Mehta S, Chen H. Risk of death in dual-eligible nursing home residents using typical or atypical antipsychotic agents. Med Care. 2012;50(11):961-969.
9. Rigler SK, Shireman TI, Cook-Wiens GJ; et al. Fracture risk in nursing home residents initiating antipsychotic medications. J Am Geriatr Soc. 2013;61(5):715-722.
10. Zagaria ME. Antipsychotic-associated metabolic and cardiovascular risks: monitoring and risk-reduction strategies. Amer J for Nurse Practitioners. 2011;15(3/4):63-66.
11. Zagaria ME. Common adverse effects of antipsychotic agents in the elderly. US Pharm. 2010;35(11):22-26.
12. Woods SW. Chlorpromazine equivalent doses for the newer atypical antipsychotics. J Clin Psychiatry. 2003; 64(6):663-667.
13. Atypical antipsychotic pharmacokinetics. MPR. Revised August 2015. www.empr.com/clinical-charts/atypical-antipsychotic-pharmacokinetics/article/197025. Accessed September 12, 2015.
14. Epocrates.com. Epocrates Plus Version 15.8. Updated September 16, 2015. Accessed September 16, 2015.
15. FDA. FDA approves new drug to treat schizophrenia and bipolar disorder. http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm463103.htm. Accessed September 21, 2015.
16. Vraylar prescribing information. Zejtun, Malta: Actavis; 2015. http://pi.actavis.com/data_stream.asp?product_group=2028&p=pi&language=E. Accessed September 23, 2015.
17. Clark MA, Finkel R, Rey JA, et al, eds. Pharmacology. 5th ed. Baltimore, MD: Lippencott Williams & Wilkins; 2012:161-168.
To comment on this article, contact rdavidson@uspharmacist.com.





