US Pharm. 2016;41(2):39-42.
ABSTRACT: Ketoacidosis is a serious medical emergency requiring hospitalization. It is most commonly associated with diabetes and alcoholism, but each type is treated differently. Some treatments for ketoacidosis, such as insulin and potassium, are considered high-alert medications, and others could result in electrolyte imbalances. Several cardiovascular complications are associated with ketoacidosis as a result of electrolyte imbalances, including arrhythmias, ECG changes, ventricular tachycardia, and cardiac arrest, which can be prevented with appropriate initial treatment. Acute myocardial infarction can predispose patients with diabetes to ketoacidosis and worsen their cardiovascular outcomes. Cardiopulmonary complications such as pulmonary edema and respiratory failure have also been seen with ketoacidosis. Overall, the mortality rate of ketoacidosis is low with proper and urgent medical treatment. Hospital pharmacists can help ensure standardization and improve the safety of pharmacotherapy for ketoacidosis. In the outpatient setting, pharmacists can educate patients on prevention of ketoacidosis and when to seek medical attention.
Metabolic acidosis occurs as a result of increased endogenous acid production, a decrease in bicarbonate, or a buildup of endogenous acids.1 Ketoacidosis is a metabolic disorder in which regulation of ketones is disrupted, leading to excess secretion, accumulation, and ultimately a decrease in the blood pH.2 Acidosis is defined by a serum pH <7.35, while a pH <6.8 is considered incompatible with life.1,3 Ketone formation occurs by breakdown of fatty acids. Insulin inhibits beta-oxidation of fatty acids; thus, low levels of insulin accelerate ketone formation, which can be seen in patients with diabetes. Extremely elevated blood glucose levels lead to osmotic diuresis, which results in excess secretion of cortisol and catecholamines, further promoting fatty-acid oxidation and ketone formation. Increased levels of glucagon, which is stimulated by hypoglycemia and insulin deficiency, leads to lipolysis, resulting in additional free fatty-acid production and ketogenesis.2
Types of Ketoacidosis
Two common types of ketoacidosis are diabetic ketoacidosis (DKA) and alcoholic ketoacidosis (AKA). The mortality rate of DKA is between 1% and 10%, with the most common cause of death being infection or cardiovascular-related events such as circulatory collapse and hypokalemia.4,5 Death resulting from AKA has been reported, but the overall mortality rate is low with urgent treatment.6,7 Both DKA and AKA are considered medical emergencies and require urgent medical care.4,6 TABLE 1 provides a comparison of the two conditions.
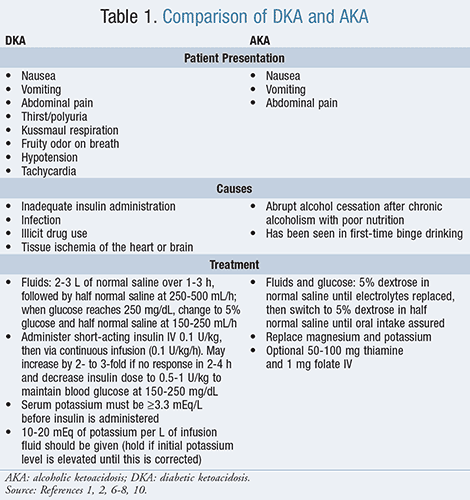
Diabetic Ketoacidosis
Ketoacidosis most commonly occurs in insulin-dependent diabetes with omission of insulin or during acute illness, which increases insulin requirements, both leading to the breakdown of fatty acids and ketone formation.2,8 Insulin depletion increases counterregulatory hormone release such as glucagon, catecholamines, cortisol, and growth hormone. Excess glucagon and catecholamines with low levels of insulin promote ketogenesis.9 DKA typically presents with hyperglycemia, arterial pH <7.3, serum ketones, and a high anion gap acidosis.2 Serum potassium may appear elevated despite a total body decrease, and hypovolemia and hyperglycemia mask the depletion of sodium, chloride, phosphorus, and magnesium. Patients may complain of nausea, vomiting, excessive thirst, frequent urination, and abdominal pain, which develop over 24 hours. Hyperglycemia can lead to osmotic diuresis and tachycardia, while volume depletion with peripheral vasodilation can result in hypotension. Patients may have a fruity odor on their breath and experience deep and labored breathing (Kussmaul respiration) secondary to the metabolic acidosis.8
Treatment of DKA: The mainstay of treatment for DKA is IV insulin and fluids, and it is best treated via a protocol-driven approach in the ICU.1 Immediately, an IV bolus of 0.1 U/kg of short-acting insulin should be administered, followed by continuous insulin infusion with 0.1 U/kg/h and increased if no response in 2 to 4 hours. The insulin dose can be decreased to 0.05 to 0.1 U/kg to maintain plasma glucose of 150 to 250 mg/dL and prevent hypoglycemia. Once the patient’s acidosis has resolved and he or she is stable and eating, IV insulin administration can be switched to long-acting insulin injections with a 2- to 4-hour overlap of continuous insulin infusion and subcutaneous (SC) insulin injections to prevent DKA relapse. Short-acting insulin should also be given with regard to meals. If initial potassium concentration is <3.3 mEq/L, insulin should be held until the potassium level is increased. In addition to insulin, an initial bolus of 2 to 3 L of normal saline should be given over 1 to 3 hours. When the patient is hemodynamically stable and urine output is occur-ring, switching to half normal saline will help decrease the risk of hyperchloremia.1
Insulin inhibits ketogenesis, promotes ketone use, and enhances bicarbonate production.1 However, hyperglycemia resolves faster than ketoacidosis, so when plasma glucose reaches 250 mg/dL, dextrose is added to maintain the plasma glucose between 150 and 250 mg/dL while insulin infusion is continued. Total body potassium is depleted in DKA, and as long as initial potassium concentrations are not elevated, 10 to 20 mEq of potassium should be added to each liter of IV fluid. Sodium bicarbonate is not recommended unless pH is <7.0, as this rapid reversal in acidosis may result in impaired cardiac function. Phosphate and magnesium replacement should be individualized based on laboratory values.8
Alcoholic Ketoacidosis
Alcoholics can also develop ketoacidosis during abrupt alcohol cessation in the presence of poor nutrition, in which the body does not have enough glucose to serve as a source of energy. During this state the body increases fatty-acid metabolism, which is enhanced by a decrease in insulin secretion and an increase in glucagon. AKA usually presents with an elevated anion gap acidosis and elevated serum lactate concentration. Some patients may have a normal blood pH due to mixed acid-base disorders owing to vomiting and respiratory alkalosis.10 Patient symptoms include nausea, vomiting, and abdominal pain.6
Treatment of AKA: The hallmark treatment for AKA is glucose and fluids.2,6,7 Glucose administration promotes insulin secretion, stopping lipolysis and free fatty-acid formation, thus halting ketogenesis. Initially, 5% dextrose in normal saline should be administered and switched to 5% dextrose in half normal saline once electrolytes are replaced. Once oral intake is assured, fluids can be discontinued.6 Potassium and magnesium administration should be individualized and guided by laboratory results. Thiamine and folate may be used if vitamin deficiency is confirmed or in those at risk for deficiency to prevent Wernicke encephalopathy.7,6 Insulin is not used, as it may cause hypoglycemia due to low glycogen levels and normal or low glucose levels on presentation.2,6,7 As with DKA, sodium bicarbonate is only indicated in severe acidemia with a pH <7.0. Phosphate replacement is only used when phosphorus levels are <1 mg/dL. In such cases there is usually another contributor to the acidosis, such as coingestions.6
Cardiovascular Complications
There are various cardiovascular complications in ketoacidosis secondary to electrolyte disturbances and catecholamine release. Pulmonary edema can also result, and patients with acute myocardial infarction (MI) may be predisposed to ketoacidosis, which in turn further causes damage to the myocardium.11
Ketoacidosis results in electrolyte imbalances, especially reductions in potassium, magnesium, and phosphorus, which can result in cardio-vascular complications if not carefully replaced (TABLE 2). Initially, potassium may present as elevated due to the intracellular shift to the surrounding plasma secondary to low levels of insulin.11 During treatment with fluids and insulin, the extracellular potassium can shift intracellularly, causing dangerously low potassium levels.12 Potassium concentrations are very important because both hypokalemia and hyperkalemia can cause arrhythmias. Normal serum range of potassium is 3.5 to 5 mEq/L. Serum levels <3 mEq/L can result in arrhythmias and cardiac arrest, whereas levels >5.5 mEq/L can cause ECG changes with subsequent ventricular tachycardia or fibrillation.11
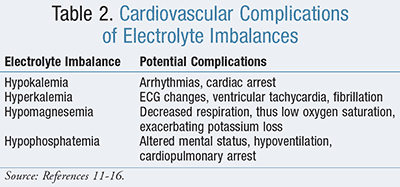
Magnesium levels should also be monitored closely, as hypomagnesemia can exacerbate potassium loss by impairing cellular uptake of potassium and increasing kidney secretion.12 Magnesium also serves as a muscle relaxant and hypomagnesemia can result in decreased respiration and low oxygen saturation, which can be very dangerous in individuals with underlying asthma or chronic obstructive pulmonary disease (COPD).13
An additional electrolyte imbalance that may occur is elevated phosphorus levels. Although phosphorous levels may initially be normal or elevated, this electrolyte level is very sensitive to standard treatment for ketoacidosis. In fact, there have been multiple case reports of complications due to drastic reductions in phosphorus levels.14-16 One case report showed that within 1 treatment day the phosphorus level of a ketoacidosis patient went from 4.9 mg/dL to 0.6 mg/dL upon ketoacidosis treatment. The normal levels of phosphorus are 2.5 to 4.5 mg/dL. This change in phosphorus level resulted in continued altered consciousness. Once phosphorus was supplied, the patient’s mental status returned to baseline on day 5 of ketoacidosis treatment.14 Depleted phosphorus levels can also result in skeletal muscle weakness and hypoventilation, leading to cardiopulmonary arrest.12 Aggressive phosphate replacement can result in hypocalcemia, so it is important to monitor both phosphorus and calcium levels.17
Catecholamine release during ketoacidosis has direct effects on the cardiovascular system.11 Reduction in serum pH normally results in increased ventilation and Kussmaul respiration. Although cardiac contractility can be depressed, catecholamine release results in normal inotropic function.1 During mild acidosis (pH 7.2-7.35), catecholamine release is increased and countered by increased cardiac output via increased chronotropic and inotropic function. Once the pH level drops below 7.2, there is direct cardiac depression due to the hydrogen ions. This ultimately leads to reduced cardiac output and potentially severe shock.11 Extremely elevated blood glucose levels result in osmotic diuresis, and this may lead to volume contraction. This state is associated with elevated cortisol levels and catecholamine secretion, which further stimulates free fatty-acid production and ketogenesis.2
Cardiopulmonary complications such as pulmonary edema and acute respiratory distress syndrome have been reported in patients with ketoacidosis.5,11 Arterial vasodilation in the periphery as well as central venous constriction can be present during ketoacidosis, and this vasoconstriction could aid in the development of pulmonary edema even in minor volume overload.11 Alveolar ventilator compensation is seen in DKA, which may precipitate low oxygen saturation and acute respiratory distress syndrome, especially in those with underlying respiratory disease or acute respiratory infections. This typically occurs during treatment of DKA rather than upon presentation, so it is important to monitor for any laboratory or clinical signs of respiratory failure while the patient is undergoing treatment.5
Acute Myocardial Infarction in Patients With Diabetes
Acute MI is seen with higher frequency in patients with diabetes and is associated with greater morbidity and mortality than in patients without diabetes.9 This link is believed to be due to increased atherosclerosis in patients with diabetes, who tend to be at an increased risk for cardiovascular-related factors such as hypertension and dyslipidemia.9,11
Acute MI is a precipitating factor for DKA in approximately 4% of patients with diabetes who experience acute MI.11 DKA precipitated by acute MI increases the mortality rate in these patients to nearly 85%. During DKA events, the myocardium is denied glucose uptake because of high levels of ketones and free fatty acids, leading to myocardial ischemia. This can further exacerbate the cardiovascular damage that has already occurred from acute MI. Additional tissue damage occurs in the heart due to increased levels of free radicals. As previously mentioned, during states of acidosis there is an increase in catecholamine release. This increase prevents any reserve insulin secretion, exacerbating lipolysis and cardiac tissue uptake of free fatty acids, thereby further injuring the myocardium with toxic fatty acids.11
The cardiac muscle is susceptible to minor changes in the pH of extracellular fluid in regard to ion exchange with intracellular fluid. In states of acidosis, there is an increase in hydrogen ions, and this increase affects multiple organelles within the myocardium. Low pH reduces the calcium concentration, resulting in less tension generated by myofibrils. In fact, tissue acidosis in myocardial ischemia develops just before or at the onset of contractility failure. In cases of extreme acidosis, necrosis can occur in the myocardium.18
Role of the Pharmacist
The treatment for ketoacidosis involves insulin and potassium replacement, which are both considered high-alert medications per the Institute for Safe Medication Practices (ISMP).6,8,19 High-alert medications are those deemed to have a greater risk of causing significant harm to a patient if used in error. Extra precautions to help reduce medication errors are necessary for these drugs, such as high-alert auxiliary labels placed by the pharmacist and automated alerts. Administering these medications in a timely manner is important to prevent patient complications. Pharmacists should be directly involved in the standardized process for ordering, storing, and preparing the drugs administered for ketoacidosis to ensure accuracy and help prevent medication errors. A pharmacist can also be a valuable source of drug information for these medications when questions arise among medical staff.
In the community and ambulatory care settings, pharmacists can also be valuable assets to educate patients on prevention of DKA and AKA. These pharmacists often see patients on a more frequent basis and can play a pivotal role in educating patients on the signs and symptoms of acidosis, and when to seek urgent medical care. Furthermore, these pharmacists have access to important information about patient medication compliance, and when an intervention may be necessary to prevent the development of ketoacidosis. Patients with diabetes should be educated to not stop their insulin abruptly without consulting their physician and to monitor for signs and symptoms of ketoacidosis in times of acute illness. Alcoholics should be educated on their risk of complications such as ketoacidosis and referred for alcohol abuse counseling.
Conclusion
DKA and AKA are serious metabolic emergencies that can result in cardiovascular complications due to electrolyte disturbances with potassium, magnesium, and phosphorus. Acute cardiovascular changes can also occur due to catecholamine release. Pulmonary edema and respiratory failure are secondary conditions that can occur as a result of ketoacidosis, and myocardium ischemia can be further exacerbated by ketoacidosis during acute MI. It is important to immediately start treatment for patients with ketoacidosis because successful management can prevent potentially life-threatening cardiovascular complications.
REFERENCES
1. DuBose TD Jr. Acidosis and alkalosis. In: Kasper D, Fauci A, Stephen Hauser S, et al, eds. Harrison’s Principles of Internal Medicine. 19th ed. New York, NY: McGraw-Hill; 2015. http://accesspharmacy.mhmedical.com/content.aspx?bookid=1130&Sectionid=79726883. Accessed October 6, 2015.
2. Kaufman DC, Kitching AJ, Kellum JA. Acid-base balance. In: Hall JB, Schmidt GA, Kress KP, eds. Principles of Critical Care. 4th ed. New York, NY: McGraw-Hill; 2015. http://accessmedicine.mhmedical.com/content.aspx?bookid=1340&Sectionid=80025262. Accessed October 6, 2015.
3. Clot-Silla E, Argudo-Ramirez A, Fuentes-Arderiu X. Letter to the editor: measured values incompatible with human life. J Int Fed Clin Chem Lab Med. www.ifcc.org/media/70148/eJIFCC_v22_02_03.pdf. Accessed October 6, 2015.
4. Kishore P. Diabetic ketoacidosis. Merck Manual Professional Version. June 1, 2014. www.merckmanuals.com/professional/endocrine-and-metabolic-disorders/diabetes-mellitus-and-disorders-of-carbohydrate-metabolism/diabetic-ketoacidosis-dka#. Accessed January 20, 2016.
5. Konstantinov NK, Rohrscheib M, Agaba EI, et al. Respiratory failure in diabetic ketoacidosis. World J Diabetes. 2015;6(8):1009-1023.
6. Woods WA, Perina DG. Alcoholic ketoacidosis. In: Tintinalli JE, Stapczynski J, Ma O, et al, eds. Tintinalli’s Emergency Medicine: A Comprehensive Study Guide. 8th ed. New York, NY: McGraw-Hill; 2016. http://accessmedicine.mhmedical.com/content.aspx?bookid=1658&Sectionid=109443879. Accessed January 20, 2016.
7. Yip L. Ethanol. In: Hoffman RS, Howland MA, Lewin NA, et al, eds. Goldfrank’s Toxicologic Emergencies. 10th ed. New York, NY: McGraw-Hill; 2015. http://accesspharmacy.mhmedical.com/content.aspx?bookid=1163&Sectionid=65098321. Accessed October 6, 2015.
8. Powers AC. Diabetes mellitus: management and therapies. In: Kasper D, Fauci A, Hauser S, et al, eds. Harrison’s Principles of Internal Medicine. 19th ed. New York, NY: McGraw-Hill; 2015. http://accesspharmacy.mhmedical.com/content.aspx?bookid=1130&Sectionid=79752952. Accessed January 20, 2016.
9. Jacoby R, Nesto R. Acute myocardial infarction in the diabetic patient: pathophysiology, clinical course and prognosis. J Am Coll Cardiol. 1992;20:736-744.
10. Cho KC. Electrolyte & acid-base disorders. In: Papadakis MA, McPhee SJ, Rabow MW, eds. Current Medical Diagnosis & Treatment 2015. New York, NY: McGraw-Hill; 2014. http://accessmedicine.mhmedical.com/content.aspx?bookid=1019&Sectionid=57668613. Accessed October 6, 2015.
11. Gandhi MJ, Suvarna TT. Cardiovascular complications in diabetic ketoacidosis. Int J Diab Dev Countries. 1995;15:132-133. http://diabetes.org.in/journal/1995_oct-dec/article5.pdf. Accessed October 6, 2015.
12. Kerr J, Moore M, Frank D. Management of diabetic ketoacidosis and other hyperglycemia emergencies. Critical Connections. December 6, 2012. www.sccm.org/Communications/Critical-Connections/Archives/Pages/Management-of-Diabetic-Ketoacidosis-and-Other-Hyperglycemic-Emergencies.aspx. Accessed October 6, 2015.
13. Van der Plas AA, Schilder JC, Marinus J, van Hilten JJ. An explanatory study evaluating the muscle relaxant effects of intramuscular magnesium sulphate for dystonia in complex regional pain syndrome. J Pain. 2013;14(11):1341-1348.
14. Murakami T, Yoshida M, Funazo T, et al. Prolonged disturbance of consciousness caused by severe hypophosphatemia: a report of two cases. Intern Med. 2014;53:2227-2232.
15. Megarbane B, Guerrier G, Blancher A, et al. A possible hypophosphatemia-induced life-threatening encephalopathy in diabetic ketoacidosis: a case report. Am J Med Sci. 2007;333(6):384-386.
16. Liu PY, Jeng CY. Severe hypophosphatemia in a patient with diabetic ketoacidosis and acute respiratory failure. J Chin Med Assoc. 2004;67(7):355-359.
17. Gosmanov AR, Gosmanova EO, Dillard-Cannon E. Management of adult diabetic ketoacidosis. Diabetes Metab Syndr Obes. 2014;7:255-264.
18. Poole-Wilson PA. Acidosis and contractility of heart muscle. Ciba Found Symp. 1982;87:58-76.
19. ISMP list of high-alert medications in acute care settings. Institute for Safe Medication Practices. 2014. www.ismp.org/tools/institutionalhighAlert.asp. Accessed January 21, 2016.
To comment on this article, contact rdavidson@uspharmacist.com.





