US Pharm. 2010;35(11):62-79.
Diabetes mellitus (DM) is more prevalent among patients with psychiatric illness compared with the general population. In addition to this, psychiatric patients are at greater risk for developing metabolic syndrome (MetS).1 MetS is characterized by abdominal or central obesity, atherogenic dyslipidemia (i.e., elevated triglycerides [TG] and reduced HDL cholesterol), insulin resistance (IR), and hypertension (HTN).2 For these reasons, together with the potential for metabolic adverse effects (AEs) associated with numerous psychotropic drugs--most notably atypical antipsychotics (AAs)--psychiatric patients must be closely monitored. Pharmacists have a unique role in the monitoring process because of their knowledge about medications and their visibility in the health care setting.
Association of Psychiatric Illness With DM and CVD
Depression, anxiety disorders, dementia, schizophrenia, and bipolar disorder (BD) occur more commonly in DM patients. DM may be involved in the development of the first three conditions.3 The exact mechanism by which DM may be linked to these conditions is not fully understood.
There appears to be a bidirectional relationship between DM and depression.4 About 7% of DM cases are thought to be attributed to depression.5 Depression is associated with a 60% to 65% increased risk of DM.5,6 Depression, which is postulated to be a state of chronic stress, has been associated with elevated cortisol levels and hypothalamic-pituitary-adrenal axis abnormalities.7-10 Additionally, depression has been associated with IR.9,11 Both depression and high cortisol levels are associated with an increased risk of MetS.12 A possible correlation has been found between a 10-point difference in Beck Depression Inventory score and a 1% (approximate) change in hemoglobin A1C (HbA1C).13 Depression also is associated with increased severity of diabetic complications.14
Central serotonergic function has been linked to both depressive symptomatology and IR.9 Further, depressed patients are more likely to be unengaged in self-care, noncompliant with dietary and weight-loss recommendations, or physically inactive--all of which can worsen obesity and IR.10,15 This risk is compounded by medications used to treat depression, which may contribute to weight gain (WG) and obesity and may be associated with increased risk of DM.4,10,15 Anxiety has been associated with an increase in inflammatory markers in DM patients, and up to 80% of DM patients experience anxiety secondary to their underlying endocrine abnormality.16,17
DM appears to double the risk of dementia.18 Numerous mechanisms have been proposed to explain this relationship. These include reduced insulin production; increased IR, resulting in peripheral hyperinsulinemia (HI); decreased insulin signaling in the brain, which may be related to increased tau phosphorylation and amyloid-beta (AB) levels (since insulin may interfere with AB breakdown); alterations in early insulin response; and hyperglycemia (HG)-related increased advanced glycation end products and oxidative stress, inflammation, and macrovascular and microvascular injury.18-22 DM appears to promote the formation of neurofibrillary tangles through involvement of free fatty acids, which are increased in obese patients.19,22
Insulin is involved in acetylcholine synthesis.18 Impaired insulin signaling may be due to neurotoxins known as AB-derived diffusible ligands.18 IR results in decreased insulin transport across the blood-brain barrier, reducing insulin levels and brain activity.19 Further, IR has been associated with increased tumor necrosis factor (TNF) levels, which may promote brain accumulation of AB.19 IR occurs in dementia as well as in MetS.19 Increased interleukin-6 levels have been found in dementia patients with functional impairment and in DM patients.23 An alteration in insulin-degrading enzyme, which is related to DM development, is associated with impaired degradation of AB protein.22 Lastly, lower insulin levels in the central nervous system have been associated with increased dementia, brain atrophy, and cognitive impairment.20
Increased rates of DM were identified in schizophrenia patients before the advent of antipsychotic medications, indicating that schizophrenia is associated with a state of IR or impaired fasting glucose tolerance.24 Psychiatric illness has a physiologic impact on carbohydrate metabolism, including effects on cortisol secretion, growth hormone production, and catecholamine secretion.3 There is a 10% to 15% prevalence of DM among schizophrenia patients, representing a relative risk (RR) of 2 to 3. The prevalence of DM among BD patients is 8% to 17%, representing an RR of 1.5 to 3.24
Despite the increased risk of suicide in psychiatric patients, the leading cause of death in patients with schizophrenia or BD is cardiovascular disease (CVD). This is especially true in younger patients, with the risk of CVD and stroke more than two to three times greater than in a nonpsychiatric population.24 MetS occurs in 37% to 63% of schizophrenia patients and in 30% to 49% of BD patients.24 Psychotropic medications used for managing mental illness can contribute to the development of MetS.25 A detailed review of the pharmacologic mechanisms associated with antipsychotic-induced metabolic abnormalities (MAs) recently was published.26
The risk factors that contribute to CVD (e.g., lack of insight, poor self-care, smoking, dyslipidemia, HTN, MetS, and visceral or abdominal obesity) also contribute to--or are present in conjunction with--DM.24 DM is associated with an increased body-mass index (BMI). Obesity constitutes a BMI ³30 kg/m2 or a waist circumference (WC) of 102 cm in men or 88 cm in women. In Asian patients, WC of >90 cm in men or 80 cm in women is considered central obesity, as is BMI ³25 kg/m2.27,28 Increased visceral adiposity and development of MetS are greater in schizophrenia patients and BD patients versus nonpsychiatric patients.29 Among antipsychotic users, younger patients with BMI <25, white patients, and patients experiencing a first episode of schizophrenia appear to be at greatest risk for WG.29 Other psychotropic classes are associated with WG (TABLE 1).29,30 Recently, selective serotonin reuptake inhibitors have been associated with development of abdominal obesity and hypercholesterolemia, which are elements of MetS. This relationship was not evident in older patients.31
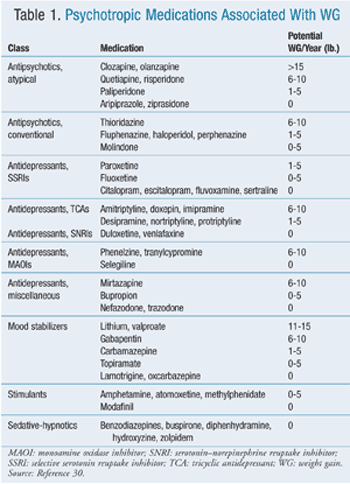
Drug-Induced WG
Among AAs, clozapine and olanzapine are associated with the greatest WG; quetiapine and risperidone with intermediate WG; and aripiprazole, paliperidone, and ziprasidone with the least WG.29 Long-term data for paliperidone and iloperidone are lacking; however, based on five 6-week trials in patients with schizophrenia or schizoaffective disorder, paliperidone was associated with a ³7% increase in body weight (BW) in 6% to 9% of patients (depending on dose) versus 1% to 9% of placebo patients.32 Iloperidone was associated with a ³7% WG in 12% to 18% of treated patients (depending on dose) versus 4% of placebo patients. Overall, based on four 4- to 6-week trials in schizophrenia patients, iloperidone was associated with a mean WG of 2.1 kg.33
There is an FDA class warning regarding these drugs' potential for inducing WG, thereby hastening the onset of or worsening DM.34 Low-potency conventional antipsychotics (CAs)--e.g., chlorpromazine, mesoridazine, thioridazine--also are associated with WG.35 Possible mechanisms for psychotropic-related WG include WG secondary to improved mood/affect; increased food cravings; alterations in resting metabolic state; sedation; increased cellular lipogenesis; and changes in neurotransmitters (e.g., 5-HT [serotonin] 1 and 2A/C receptor antagonism) and neuropeptides (e.g., leptin, ghrelin, cytokines such as TNF).29 Whereas antagonism of muscarinic (M) 3 and 5-HT1A receptors may lead to decreased pancreatic beta-cell responsiveness, 5-HT2A receptor antagonism suppresses glucose uptake into skeletal muscle; both factors appear to contribute to AA-induced HG.36 Medications with the greatest likelihood of increasing BW also are associated with increased IR and hypertriglyceridemia. Most WG occurs within the first 38 to 40 weeks after AA therapy is initiated, and WG reaches a plateau by about 9 months.35
Additionally, increased levels and patterns of circulating cortisol and catecholamines and immunologic alterations--including altered cytokine expression--that appear to be related to the underlying mental illness place the psychiatric patient at further risk for CVD.24
Psychiatric Drugs That Cause Glucose Intolerance (GI) or DM
Numerous medications have been associated with drug-induced HG.37 A connection has been found between antipsychotic agents and development of DM.38,39 Some studies have found a stronger association in patients who are younger or African American; others have reported older age and nonantipsychotic psychotropics as risk factors.36,39,40 A pattern similar to that for AA-associated WG has been seen for drug-induced DM, with drugs such as clozapine and olanzapine associated with greater risk of glucose abnormalities compared with other agents in the class.39 HI has been reported in 30% to 70% of patients receiving clozapine or olanzapine.41
Inadequate treatment of MAs in psychiatric patients may be due to lack of knowledge on the part of treating psychiatrists. The Atypical Antipsychotic Therapy and Metabolic Issues National Survey found that only 59% of psychiatrists recognized WG as a potential AE of AAs, and just 51% acknowledged DM as a potential complication of AAs. Even fewer psychiatrists recognized dyslipidemia (22%) or metabolic decompensation--e.g., diabetic ketoacidosis (DKA)--(2%) as possible AEs. However, 85% stated that they had changed a patient's regimen because of concerns about adverse metabolic consequences.42
Some cases of AA-induced DM appear to be related to significant WG, but up to one-quarter may not be.36 This points to the possibility of two different mechanisms that may contribute to drug-induced DM: a direct cellular effect on glucose utilization by blocking glucose transporter protein in cell membranes, and an indirect effect on carbohydrate metabolism via the neuroendocrine system. Antipsychotics may directly contribute to development of DM by altering beta cells' capacity to secrete sufficient insulin.43
A review was recently published on postulated mechanisms behind AA-induced DM.44 Insulin metabolism and carbohydrate regulation may be affected, which may explain the rapid fluctuations in blood glucose (BG) noted in some patients despite the absence of massive WG.30 AAs may lead directly to production of atherogenic dyslipidemia, which has been associated with IR. In patients who develop dyslipidemia and IR, there may be a rapid increase in fasting TG levels upon therapy initiation and an equally rapid decrease upon discontinuation.43 Likewise, AAs may induce MAs in the absence of significant WG.36 AAs, but not CAs, appear to impair the action of insulin on adipocytes, which has a negative impact on the lipid profile.36
AAs have been associated with DKA and hyperglycemic hyperosmolar syndrome. It is thought that M3 cholinergic receptor antagonism may play a role in the development of these life-threatening conditions. These conditions occur rapidly upon initiation of antipsychotic therapy, often within the first week.45 At greatest risk are younger patients, those with a lower BMI, and female patients. Although these conditions can be fatal, especially if the antipsychotic is not withdrawn, they usually resolve upon discontinuation.43 Positive rechallenge has been noted, however.45
Managing Psychotropic Drug-Associated BG Alterations
Various interventions have been tried to help manage the WG and MAs associated with psychotropic medications. A consensus statement has been published on AAs and the development of obesity and DM.46 Weight checks are recommended at baseline, monthly for the first 3 months, and quarterly thereafter, with additional checks performed at every visit.46 Lifestyle modifications such as nutritional counseling, behavioral interventions, and increased physical activity are recommended for overweight or obese psychiatric patients taking AAs.46
Interventions to prevent WG should be started when a patient's BMI is >25 kg/m2. Active weight-loss intervention should be undertaken for those with a BMI of 30 kg/m2; BMI between 25 kg/m2 and 29.9 kg/m2 in the presence of two or more risk factors; or WC >88 cm in women or 102 cm in men, along with two or more risk factors (e.g., HTN, dyslipidemia, CVD, DM, sleep apnea). Lifestyle interventions should be tried for 6 months before re-evaluation to determine the need for more aggressive treatment.29
Early (i.e., within the first 4-12 weeks of therapy) WG is a predictor of long-term substantial WG, even though WG may plateau around week 40. For most AAs, a clear dose-WG relationship has not been firmly established, although evidence points to a direct correlation for clozapine.47 Patients with a BMI <25 kg/m2 appear to be at greater risk for developing WG from AAs.29
If additional interventions are desired or if lifestyle interventions alone are unsuccessful, one strategy is to switch to an AA with a lower likelihood of WG.29,45,47 Care must be exercised, however, since switching antipsychotics carries a risk of loss of symptom control.24,45 Different strategies have been used for switching, including abrupt discontinuation of the offending agent or gradual tapering followed by slow uptitration of a new drug with a more benign AE profile.45 The Comparison of Antipsychotics for Metabolic Problems study is examining the issue of antipsychotic switching in schizophrenia patients.48
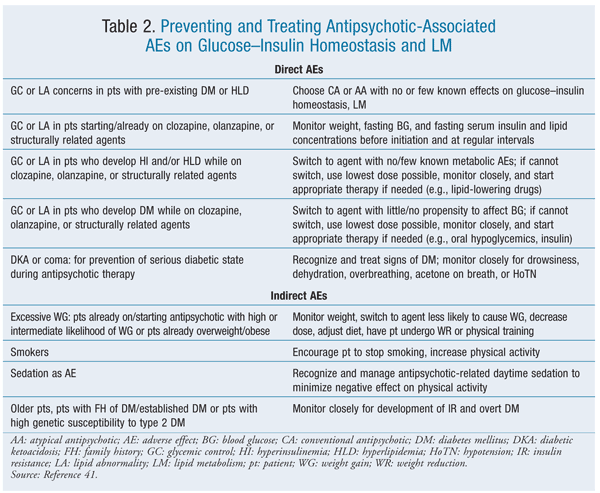
Guidelines have been developed for the prevention and treatment of antipsychotic-associated AEs on glucose-insulin homeostasis and lipid metabolism (TABLE 2).41
Adjunctive pharmacotherapeutic agents also have been tried, although none is approved for managing psychotropic-induced WG or MetS. A goal of therapy is to lose 5% to 10% of initial BW over 3 to 6 months.45 Drugs utilized are orlistat, an enteric inhibitor of pancreatic enzymes; sibutramine, a reuptake inhibitor of norepinephrine, serotonin, and dopamine; topiramate, a mood stabilizer/anticonvulsant; H2 receptor antagonists (i.e., nizatidine, ranitidine, famotidine); amantadine, an antiviral/antiparkinsonian; antidepressants (e.g., fluoxetine, fluvoxamine); phenylpropanolamine, a sympathomimetic; dextroamphetamine, a psychostimulant; rosiglitazone, a thiazolidinedione; modafinil, an analeptic; and metformin.47,49
Two recent papers describe these modalities.47,49 Most trials have enrolled small numbers of patients and have yielded conflicting outcomes, resulting in insufficient support for their use. Results have been somewhat favorable for topiramate, reboxetine, and amantadine, but have been more nebulous for sibutramine and largely negative for H2 receptor antagonists and fluoxetine.47
These drugs are not indicated for the prevention or treatment of psychotropic-induced WG or metabolic disturbances. Diet and exercise are more beneficial than the use of adjunctive pharmacotherapy, which should be reserved for patients who have failed lifestyle modifications alone.29,47 Of the abovementioned agents, the most promising is metformin, although results are mixed.36,50
Metformin, a biguanide hypoglycemic agent, is useful for preventing or delaying the onset of type 2 DM in high-risk patients.51 Metformin has been used alone, in combination with lifestyle modifications, or with sibutramine. Doses studied have ranged from 500 to 2,250 mg/day. Patients experiencing metabolic complications secondary to AA therapy who are younger or who are placed on biguanide shortly after initiating antipsychotic therapy appear to benefit most from metformin.36
Guidelines for the use of metformin in managing AA-induced WG and glucose-metabolism dysregulation were recently published.36 A point system was developed in which a score of ³4 points renders a patient a potential candidate for metformin therapy. The point system is divided into baseline features. These include family history of DM or CVD; current BMI within normal range (18.5-24.9 kg/m2), but a positive history of overweight or obesity; BMI in the overweight or obese range; presence of HG or GI; and features developing after antipsychotic initiation (including progression of normal baseline BMI to overweight or obese status and new-onset HG or GI).36 Metformin may be discontinued if no benefit is noted after 6 to 8 months.36
Health outcomes are generally poorer for patients with DM and concomitant psychiatric illness. This is largely attributed to noncompliance with BG monitoring and failure to adhere to pharmacologic and behavioral treatment plans.3 However, this assumption has been challenged by a study that concluded that glucose control was better in DM patients receiving psychiatric services for schizophrenia than in DM patients without mental illness.52
Monitoring Patients
The American Diabetes Association (ADA)--in conjunction with the American Psychiatric Association, the American Association of Clinical Endocrinologists, the North American Association for the Study of Obesity, and the Mount Sinai Conference--has published recommendations for monitoring patients taking antipsychotics.46,53 When therapy is initiated, the ADA advises monitoring weight and BMI at baseline; at 4, 8, and 12 weeks; and quarterly thereafter. Patients should monitor their weight and track any changes. WG of >5% over baseline may indicate the need to switch to an agent less likely to cause WG. Fasting glucose, lipid levels, and blood pressure (BP) should be monitored at baseline and at 3 months. Thereafter, BG and BP may be monitored yearly and a lipid profile taken every 5 years if they remain within normal limits. If abnormal glucose or lipid levels are present, the patient should undergo further evaluation even if asymptomatic. WC and family or personal history should be determined at baseline and annually thereafter.46
The Mount Sinai guidelines recommend monitoring weight, lipid profile, and glycemic status.53 Patients should be weighed and have their WC determined at baseline or upon change of therapy and at every subsequent visit for the first 6 months. If WG of ³1 BMI is present or if the WC is >35 inches in women or >40 inches in men, a weight-loss plan should be initiated and a switch to an agent less likely to cause WG should be considered. Patients should be weighed at every visit until their weight has stabilized, and every 3 months thereafter. Patients should self-monitor their weight and report any significant gains to their health care provider.
Patients who already have DM, who present with HG prior to AA initiation, or who need a change of antipsychotic should be referred to a medical provider. In the absence of DM or HG, patients at high risk for glucose abnormalities should test their glucose in 4 months. If the patient is hyperglycemic, a referral is recommended; if not, glucose should be checked annually. Results that call for more frequent BG monitoring include WG of ³1 BMI unit, fasting BG >110 mg/dL, random BG >200 mg/dL, or HbA1C >6.1%.53
If a baseline lipid profile reveals no abnormalities, lipids may be tested every 2 years. If lipids are elevated at baseline or upon a change in therapy, the patient should be referred to a medical provider, or diet and lipid-lowering therapy should be considered. In this population, the lipid profile should be monitored every 6 months if LDL >130 mg/dL.53 Caution is advised in administering AAs with statins, as cases of rhabdomyolysis have been reported.45
Guidelines have indicated that either a fasting glucose or HbA1C may be used for monitoring blood sugar.51,53 Despite this, monitoring of metabolic profiles in patients taking AAs remains insufficient.54,55 Another problem the psychiatric patient faces, besides insufficient screening and management of MAs, is the inadequate management of depression.56
Conclusion
Management of metabolic complications, including DM and MetS, in the psychiatric patient is complex. Pharmacists are uniquely suited to guide patients in this regard, as it has been shown that they can reduce CVD risk, even in DM patients with psychiatric conditions.57 There is a monitoring gap in clinical practice that the pharmacist can help fill.54 In patients with severe mental illness, the provision of regularly scheduled psychiatric care may positively impact glycemic control.58,59 It is important to take a holistic approach to the care of these patients, addressing both their psychiatric and physical health care needs.60,61 In keeping with this holistic approach, treatment of the underlying psychiatric condition should not be neglected. For example, only about one-quarter of depressed DM patients are appropriately diagnosed and treated for their depression.56
It is projected that, by 2020, depression will be the second leading cause of disease worldwide. Depression is especially problematic in older adults, as it complicates the course and outcome of other illnesses.62 The pharmacist's role is likely to expand as the United States faces an obesity epidemic and an aging population often afflicted with psychiatric conditions such as dementia, depression, and late-onset schizophrenia.
REFERENCES
1. Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults. Executive summary of the Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III). JAMA. 2001;285:2486-2497.
2. Newcomer JW. Metabolic syndrome and mental illness. Am J Manag Care. 2007;13(suppl 7):S170-S177.
3. Myers KA, Edwards AL. Diabetic patients with psychiatric illness. Curr Diabetes Rev. 2010;6:222-227.
4. Lyketsos CG. Depression and diabetes: more on what the relationship might be. Am J Psychiatry. 2010;167:496-497.
5. Campayo A, de Jonge P, Roy JF, et al. Depressive disorder and incident diabetes mellitus: the effect of characteristics of depression. Am J Psychiatry. 2010;167:580-588.
6. Mezuk B, Eaton WW, Albrecht S, Golden SH. Depression and type 2 diabetes over the lifespan: a meta-analysis. Diabetes Care. 2008;31:2383-2390.
7. Wolkowitz OM, Epel ES, Reus VI. Stress hormone-related psychopathology: pathophysiological and treatment implications. World J Biol Psychiatry. 2001;2:115-143.
8. Boyle SH, Surwit RS, Georgiades A, et al. Depressive symptoms, race, and glucose concentrations: the role of cortisol as mediator. Diabetes Care. 2007;30:2484-2488.
9. Okamura F, Tashiro A, Utsumi A, et al. Insulin resistance in patients with depression and its changes in the clinical course of depression: a report on three cases using the minimal model analysis. Intern Med. 1999;38:257-260.
10. Golden SH, Williams JE, Ford DE, et al. Depressive symptoms and the risk of type 2 diabetes: the Atherosclerosis Risk in Communities study. Diabetes Care. 2004;27:429-435.
11. Ramasubbu R. Insulin resistance: a metabolic link between depressive disorder and atherosclerotic vascular diseases. Med Hypotheses. 2002;59:537-551.
12. Vogelzangs N, Suthers K, Ferrucci L, et al. Hypercortisolemic depression is associated with the metabolic syndrome in late-life. Psychoneuroendocrinology. 2007;32:151-159.
13. Van Tilburg MAL, McCaskill CC, Lane JD, et al. Depressed mood is a factor in glycemic control in type 1 diabetes. Psychosom Med. 2001;63:551-555.
14. de Groot M, Anderson R, Freedland KE, et al. Association of depression and diabetic complications: a meta-analysis. Psychosom Med. 2001;63:619-630.
15. Brown LC, Majumdar SR, Newman SC, Johnson SA. History of depression increases risk of type 2 diabetes in younger adults. Diabetes Care. 2005;28:1063-1067.
16. Brennan AM, Fargnoli JL, Williams CJ, et al. Phobic anxiety is associated with higher serum concentrations of adipokines and cytokines in women with diabetes. Diabetes Care. 2009;32:926-931.
17. Tuncay T, Masabak I, Gok DE, Kutlu M. The relationship between anxiety, coping strategies and characteristics of patients with diabetes. Health Qual Life Outcomes. 2008;6:79.
18. Kroner Z. The relationship between Alzheimer's disease and diabetes: type 3 diabetes? Altern Med Rev. 2009;14:373-379.
19. Craft S. The role of metabolic disorders in Alzheimer disease and vascular dementia: two roads converged. Arch Neurol. 2009;66:300-305.
20. Burns JM, Donnelly JE, Anderson HS, et al. Peripheral insulin and brain structure in early Alzheimer disease. Neurology. 2007;69:1094-1104.
21. Rönnemaa E, Zethelius B, Sundelöf J, et al. Glucose metabolism and the risk of Alzheimer's disease and dementia: a population-based 12 year follow-up study in 71-year-old men. Diabetologia. 2009;52:1504-1510.
22. Akomolafe A, Beiser A, Meigs JB, et al. Diabetes mellitus and risk of developing Alzheimer disease: results from the Framingham Study. Arch Neurol. 2006;63:1551-1555.
23. Zuliani G, Guerra G, Ranzini M, et al. High interleukin-6 plasma levels are associated with functional impairment in older patients with vascular dementia. Int J Geriatr Psychiatry. 2007;22:305-311.
24. Holt RIG, Peveler RC. Diabetes and cardiovascular risk in severe mental illness: a missed opportunity and challenge for the future. Practical Diabetes Int. 2010;27:79-84.
25. Wofford MR, King DS, Harrell TK. Drug-induced metabolic syndrome. J Clin Hypertens (Greenwich). 2006;8:114-119.
26. Reynolds GP, Kirk SL. Metabolic side effects of antipsychotic drug treatment--pharmacological mechanisms. Pharmacol Ther. 2010;125:169-179.
27. Tan CE, Ma S, Wai D, et al. Can we apply the National Cholesterol Education Program Adult Treatment Panel definition of the metabolic syndrome to Asians? Diabetes Care. 2004;27:1182-1186.
28. Anuurad E, Shiwaku K, Nogi A, et al. The new BMI criteria for Asians by the regional office for the Western Pacific Region of WHO are suitable for screening of overweight to prevent metabolic syndrome in elder Japanese workers. J Occup Health. 2003;45:335-343.
29. Citrome L, Vreeland B. Schizophrenia, obesity, and antipsychotic medications: what can we do? Postgrad Med. 2008;120:18-33.
30. Vieweg WVR, Levy JR, Fredrickson SK, et al. Psychotropic drug considerations in depressed patients with metabolic disturbances. Am J Med. 2008;121:647-655.
31. Raeder MB, Bjelland I, Vollset SE, Steen VM. Obesity, dyslipidemia, and diabetes with selective serotonin reuptake inhibitors: the Hordaland Health Study. J Clin Psychiatry. 2006;67:1974-1982.
32. Invega (paliperidone) product information. Titusville, NJ: Ortho-McNeil-Janssen Pharmaceuticals, Inc; January 2010.
To comment on this article, contact rdavidson@uspharmacist.com.





