US Pharm. 2010;35(11):49-61.
Traditionally, detoxification products have been used to remove the buildup of toxins thought to accumulate in the body through ingestion and exposure. While detoxification products serve the same purpose today, they are also used as an adjunct to rapid-weight-loss regimens. Because of possible laxative effects, some detoxification products have a potential for abuse.
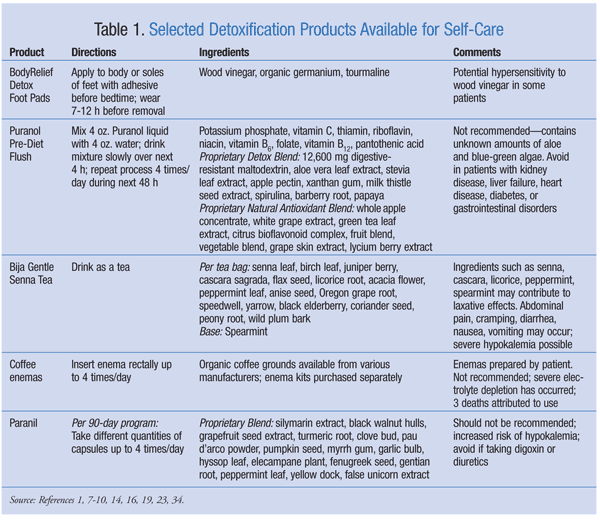
Detoxification products are available in a variety of delivery forms, including topical patches, liquids, enemas, and capsules. Selected products and their ingredients are listed in TABLE 1. Detoxification products fall under the category of supplements and are not required by the FDA to have proof of efficacy or standardized ingredients. Public awareness of side effects and interactions related to ingredients in detoxification products is minimal, and often health care professionals are not consulted when these products are used. It is important that health care professionals, especially pharmacists, take a more active role in counseling patients about detoxification products.
Topical Detoxification
Detoxification foot pads are used to purify the body and remove toxins and impurities through the skin. Products commonly contain powdered wood or bamboo vinegars (pyroligneous acid), germanium, and tourmaline. After use, brown coloration on the pad indicates absorption of toxins. Complete detoxification is indicated when the pads no longer change color after use.
The active ingredient in detoxification foot pads is crystallized pyroligneous acid, a product of ethanol oxidation and the destructive distillation of wood.1 There are no proven effects of topical application of germanium.2 Small studies have reported that tourmaline, a source of infrared radiation, may confer circulatory benefits; however, no conclusive testing has been done to evaluate the form or quantity of tourmaline required to achieve an effect.3,4
To date, the effectiveness of detoxification foot pads has not been proven. While there are no clinical studies to disprove the claims, several reporting agencies performed informal testing to make consumers aware of a possible scam in relation to marketing claims. The claim that detoxification foot pads help the body expel impurities and draw out toxins is based on the physiology of the eccrine sweat glands on the soles of the feet. Eccrine glands naturally secrete water, salts, and traces of metabolic waste in the form of urea, uric acid, and ammonia.5 This process occurs regardless of the use of detoxification foot pads. The marketers of Kinoki Foot Pads have been charged with deceptive advertising by the Federal Trade Commission.6
Consumers should be aware that skin irritation and allergic dermatitis have been reported with topical use of pyroligneous acid.1 No adverse reactions have been reported with topical use of germanium, and there is insufficient information on topical use of tourmaline to suggest adverse reactions.2,4
Liquid Detoxification
Detoxification products are also available in liquid form. Although many liquid products exist for colon cleansing, several manufacturers are pushing toward a new trend: predieting. The purpose of predieting detoxification liquids is to remove toxins from the body in order to maximize the effects of future dieting. One product, Puranol Pre-Diet Flush (Windmill Health Products), is claimed to “prepare your body to be ready for a diet and optimize the amount of weight you can lose.”7 The manufacturer also mentions that the product will “cleanse your body of built-up waste and toxins that hinder the success of your diet.”7
Puranol has two line extensions known as “proprietary blends.”8 Ingredients, cautions, drug interactions, and directions appear in TABLE 1.
Aloe, blue-green algae, milk thistle, and green tea are ingredients in Puranol products that may be associated with specific adverse effects, as well as unproven claims of efficacy. As a leaf extract contained in an unknown quantity, aloe possibly is effective for constipation. It is a stimulant laxative that contains up to 30% anthraquinones.9 Anthraquinones are transformed into anthrones, which irritate the mucous membranes of the colon. Anthrones also increase fluid and electrolyte secretion into the gut, thereby depleting electrolyte levels. The adverse effects of aloe when used as a cathartic agent include cramping, diarrhea, muscle weakness, and weight loss. The use of aloe leaf extract has been linked to acute hepatitis.9 Aloe also is known to cause hypokalemia and hypoglycemia.9 Patients should be advised not to use aloe if they have diabetes, gastrointestinal conditions, liver disease, or renal failure. Aloe should not be given to patients who take digoxin, as the drug's effectiveness may be increased and toxicity is likely.10
In 2002, the FDA requested that all oral products with aloe be reformulated (i.e., aloe removed), owing to safety concerns.11 However, products containing aloe are still available.
Another ingredient, blue-green algae, has been used for its detoxifying effects. Blue-green algae have many uses, including weight loss, improved digestion, and bowel health. Algae are plantlike bacteria that consist of 65% crude protein, high concentrations of B vitamins, phenylalanine, and some traces of iron.10 The phenylalanine is thought to reduce appetite and provide a feeling of fullness. Algae also are known to stimulate the immune system if consumed in certain quantities.10
There is no reliable evidence that blue-green algae are effective for detoxifying the body or reducing weight.10 Blue-green algae should not be recommended; this supplement is prone to contamination with microbes and heavy metals, which could cause serious adverse reactions related to poisoning.10 Although adverse effects are rare with pure algae, they may include upset stomach, vomiting, diarrhea, and edema.10 Interactions with immunosuppressant therapy also have been noted. Uncontaminated algae free of microcystin, a toxin produced by the bacteria, may be recommended for use.10 Products must be tested for purity, but Puranol does not claim to participate in purity testing and is not certified by the United States Pharmacopeia.
Milk thistle is known to have laxative effects.12 It also is used to clear the liver of toxic chemicals. The mechanism of action of milk thistle involves its active component silymarin.12 Silymarin alters the hepatocyte cell membrane to prevent toxins from entering the liver. It also has antioxidant effects on the liver and may aid in its regeneration. Although milk thistle has few interactions and is generally safe, there is not enough evidence to support its benefits.12
Green tea is used for weight loss and cognitive alertness.13 Although it varies by type of tea and brewing time, the amount of caffeine is approximately 20 to 45 mg per cup of brewed tea. There is insufficient evidence that green tea can detoxify the body or promote weight loss. Adverse effects of caffeine include nausea, vomiting, bloating, dizziness, and confusion.13 Green tea can interact with and increase the effectiveness of other central nervous system (CNS) stimulants, such as amphetamines, oral decongestants, and cocaine.8 Green tea should not be used by individuals with bleeding disorders, anemia, or anxiety.13
Puranol products contain selected vitamins and minerals that are beyond the scope of this article. A blend of apple, barberry, grape, citrus bioflavonoid, lemon, lycium berry, xanthan gum, and papaya is another component. There is no conclusive evidence to link any of these ingredients to detoxifying effects.8
Uncommon Liquid Detoxification Products
Laxative Teas: Senna teas are used to cleanse the body of toxins. Bija Gentle Senna Tea (Flora, Inc.) is one example.14 Ingredients are given in TABLE 1.
Senna is used for constipation, weight loss, and cleansing.15 Senna is a stimulant laxative (anthranoid) that causes the colon to produce secretions and increase motility. Abdominal pain, nausea, vomiting, bloating, and diarrhea have been associated with its use.13 If excessive amounts of senna tea are ingested (>1 L/day), liver failure can occur.16
Cascara is another stimulant laxative found in Bija Gentle Senna Tea. For effectiveness, cascara must be obtained from aged, dried bark.16 The bark contains anthraglycosides, which act as an intestinal stimulant. Specifically, cascara causes an increase in large-intestine motility and allows evacuation to occur.17 The adverse effects of cascara include abdominal cramping and flatulence. Additionally, if cascara bark is not properly aged, it can cause severe vomiting. Cascara can cause severe hypokalemia, and it should not be used longer than 2 weeks under any circumstances.16 Patients may become dependent upon cascara and aloe for their laxative effects.
Although the FDA had approved cascara in medications, approval was revoked in 2002 owing to safety concerns.16 However, cascara-containing products, as well as aloe agents, continue to be marketed in the U.S.
Bija Gentle Senna Tea contains licorice root. Licorice has been associated with detoxification effects, specifically weight loss.18 Its ability to rid the body of impurities is unproven. Adverse effects include water retention, increased sodium absorption, and pseudohyperaldosteronism.18 Hypertension and severe edema have been associated with long-term use of licorice. Licorice should not be used with senna or cascara, as excessive potassium depletion may occur.18
Coffee Enemas: Enemas are used to evacuate waste products from the colon. Coffee enemas are used to rid the body of toxins and promote liver activity.19 This therapy was developed by the German physician Max Gerson to treat patients with cancer.20 As part of the therapy, patients are instructed to use coffee enemas four times daily.20 Patients must also take various supplements and follow a special diet consisting of fruits and vegetables. Additionally, the Gonzalez regimen advocates the use of coffee enemas twice daily in the treatment of pancreatic cancer.21 Patients typically prepare the enemas themselves, using specially formulated coffee grounds and enema kits. The kits and coffee are sold by various manufacturers; alternatively, the coffee may be brewed at home.
The mechanism of coffee enemas is complex. The coffee is absorbed through the colon wall and enters the liver.22 It is believed that coffee enemas act on the liver by dilating blood vessels.22 The increase in blood flow causes the liver to more efficiently remove toxins. It is theorized that the bile duct becomes dilated and that this action increases the liver's ability to metabolize toxic substances related to cancer growth and treatment.20 However, there is no evidence to support the beneficial activity of coffee enemas.22 When coffee is administered rectally, severe depletion of electrolytes may occur.21 Septicemia also has been associated with coffee enemas. To date, three deaths resulting from coffee-enema use have been documented.19 Coffee enemas are not recommended for patient use.
Capsule Detoxification
Paranil (DrNatura) is a detoxification product widely used to purify the colon and liver.23 This product is one component of a detoxification regimen known as the Colonix Internal Cleansing Program; two other products, a fiber powder and a senna tea, are also part of this program. Paranil is the main component purported to be responsible for colon and liver purification. The manufacturer's directions for use are complicated.
According to the manufacturer, “Paranil® capsules contain 18 detoxifying herbs that have been successfully used for many years by naturopaths and other health professionals to purify the colon and liver. It is powerful yet safe and well tolerated by adults.”23 Paranil's ingredients are listed in TABLE 1. Major drug interactions may occur with ingredients found in this product; these are discussed below.
Prickly ash may increase the risk of bleeding when used with anticoagulants and antiplatelets, and it has minor interactions with antacids, H2 blockers, and proton-pump inhibitors.24 Elecampane moderately increases the effect of CNS depressants.25 Unicorn root may cause stupor, colic, and vertigo, as well as minor interactions with antacids, H2 blockers, and proton-pump inhibitors.26 Fenugreek has a moderate interaction with anticoagulants and antidiabetic medications.
Garlic has multiple drug interactions, including moderate interactions with anticoagulants, oral contraceptives, cyclosporine, and CYP450 2E1 and 3A4 substrates.27 Additionally, garlic has major interactions with isoniazid, nonnucleoside reverse transcriptase inhibitors, and saquinavir.28 Patients taking these medications should avoid garlic. Grapefruit has major interactions with benzodiazepines, buspirone, calcium channel blockers, carbamazepine, carvedilol, clomipramine, cyclosporine, CYP450 3A4 substrates, dextromethorphan, estrogens, sildenafil, fexofenadine, statins, itraconazole, etoposide, and cisapride.29 Hyssop and male fern have no known drug interactions.30,31 Myrrh has moderate interactions with antidiabetic drugs and warfarin.32 Peppermint has moderate interactions with cyclosporine and CYP450 1A2, 2C19, 2C9, and 3A4.33 Yellow dock may have drug interactions with digoxin and potassium-depleting diuretics, resulting in hypokalemia.34
Other Considerations: Abuse
Eating disorders such as anorexia nervosa have been associated with the abuse of detoxification products, especially laxatives.35 One subtype of anorexia nervosa is characterized by binging, purging, or both. Often, people suffering from eating disorders feel gratification when they use detoxifying products.35 Many of these individuals are attracted to laxatives and enemas because of claims of detoxification and weight loss. In one study, more than 18.8% of subjects with eating disorders claimed to abuse laxatives.35 Patients who suffer from anorexia and its various subtypes, such as bulimia, may present with symptoms related to the overuse of laxatives and detoxification products. Specifically, patients may experience electrolyte imbalance, severe hypokalemia, and edema.35 The use of detoxification products can worsen the condition of patients with eating disorders.
Conclusion
Many products on the market claim to have the ability to detoxify the body. Among the products available to consumers are topical products and oral products in both liquid and tablet form. However, these products are not sanctioned by the FDA, meaning that there is no evidence-based clinical literature to support their use. Additionally, patients should be advised that detoxifying agents are not effective for removing toxins and may, in fact, be dangerous.
REFERENCES
1. Hazardous Substances Data Bank. Acetic acid: human health effects.
http://toxnet.nlm.nih.gov/cgi-
2. Hazardous Substances Data Bank. Germanium: human health effects.
http://toxnet.nlm.nih.gov/cgi-
3. Yoo B, Park C, Oh T, et al. Investigation of jewelry powders radiating far-infrared rays and the biological effects on human skin. J Cosmet Sci. 2002;53:175-184.
4. Liang J, Zhu D, Meng J, et al. Performance and application of far infrared rays emitted from rare earth mineral composite materials. J Nanosci Nanotechnol. 2008;8:1203-1210.
5. Marieb EN, Hoehn K. The integumentary system. In: Marieb E, Hoehn K, eds. Human Anatomy and Physiology. 8th ed. San Francisco, CA: Benjamin Cummings; 2010:155.
6. Federal Trade Commission. FTC charges marketers of Kinoki Foot Pads with deceptive advertising; seeks funds for consumer redress.
www.ftc.gov/opa/2009/01/xacta.
7. Dietary Supplements Labels Database. Windmill Health Puranol™ 48 Hour Pre-diet Flush.
http://dietarysupplements.nlm.
8. Natural Medicines Comprehensive Database. Puranol 48 Hour Pre Diet Flush by Windmill Health Products.
www.naturaldatabase.com/(S(
9. Natural Medicines Comprehensive Database. Aloe.
www.naturaldatabase.com/(S(
10. Natural Comprehensive Medicine Database. Blue-green algae.
www.naturaldatabase.com/(S(
11. Status of certain additional over-the counter drug category II and III active ingredients. Fed Regist. 2002;67:31125-31127.
12. Natural Medicines Comprehensive Database. Milk thistle.
www.naturaldatabase.com/(S(
13. Natural Medicines Comprehensive Database. Green tea.
www.naturaldatabase.com/(S(
14. Natural Medicines Comprehensive Database. Bija Gentle Senna Tea information.
www.naturaldatabase.com/(S(
15. Natural Medicines Comprehensive Database. Senna.
www.naturaldatabase.com/(S(
16. Vanderperren B, Rizzo M, Angenot L, et al. Acute liver failure with renal impairment related to the abuse of senna anthraquinone glycosides. Ann Pharmacother. 2005;39:1353-1357.
17. Natural Medicines Comprehensive Database. Cascara.
www.naturaldatabase.com/(S(
18. Natural Medicines Comprehensive Database. Licorice.
www.naturaldatabase.com/(S(
19. Natural Medicines Comprehensive Database. Detoxification [coffee enema].
www.naturaldatabase.com/(S(
20. National Cancer Institute. Gerson therapy (PDQ®).
www.cancer.gov/cancertopics/
21. National Cancer Institute. Gonzalez regimen (PDQ®).
www.cancer.gov/cancertopics/
22. Ernst E. Colonic irrigation and the theory of autointoxication: a triumph of ignorance over science. J Clin Gastroenterol. 1997;24:196-198.
23. DrNatura. Colonix will make you look and feel better.
www.drnatura.com/colonix_
24. Natural Medicines Comprehensive Database. Northern prickly ash.
www.naturaldatabase.com/(S(
25. McGuffin M, Hobbs C, Upton R, Goldberg A, eds. American Herbal Products Association's Botanical Safety Handbook. Boca Raton, FL: CRC Press; 1997.
26. Natural Medicines Comprehensive Database. Aletris [unicorn root].
www.naturaldatabase.com/(S(
27. Legnani C, Frascaro M, Guazzaloca G, et al. Effects of a dried garlic preparation on fibrinolysis and platelet aggregation in healthy subjects. Arzneimittelforschung. 1993;43:119-122.
28. Natural Medicines Comprehensive Database. Garlic.
www.naturaldatabase.com/(S(
29. Natural Medicines Comprehensive Database. Grapefruit.
www.naturaldatabase.com/(S(
March 5, 2010.
30. Natural Medicines Comprehensive Database. Hyssop.
www.naturaldatabase.com/(S(
31. Natural Medicines Comprehensive Database. Male fern.
www.naturaldatabase.com/(S(
32. Natural Medicines Comprehensive Database. Myrrh.
www.naturaldatabase.com/(S(
33. Natural Medicines Comprehensive Database. Peppermint.
www.naturaldatabase.com/(S(
34. Brinker F. Herb Contraindications and Drug Interactions. 2nd ed. Sandy, OR: Eclectic Medical; 1998.
35. Kovacs D, Palmer RL. The associations between laxative abuse and other symptoms among adults with anorexia nervosa. Int J Eat Disord. 2004;36:224-228.
To comment on this article, contact rdavidson@uspharmacist.com.






