US Pharm. 2015;40(11):HS15-HS20.
ABSTRACT: A variety of factors can cause cognitive impairment in hospitalized patients, including surgery, critical illness, and medications. Furthermore, the development of cognitive impairment during hospitalization is associated with long-term cognitive dysfunction. Pharmacists should be aware of patient-specific risk factors and should actively monitor for medications that contribute to cognitive impairment. The role of pharmacotherapy in treating patients who develop cognitive impairment while hospitalized is limited; however, by minimizing the risk of this condition in hospitalized patients, pharmacists have the opportunity to reduce long-term cognitive dysfunction.
There are many reasons for the occurrence of cognitive impairment in patients during hospitalization, and this impairment can have a long-term impact post discharge.1,2 This review will discuss common causes of and risk factors for cognitive impairment in hospitalized patients, including surgery, critical illness, and medications. Pharmacists in the hospital setting should be aware of these issues to minimize patients’ exposure to any modifiable risk factors and to decrease the possibility of long-term cognitive dysfunction.
Delirium
Incidence: The incidence of delirium, a syndrome consisting of an acute change or fluctuation in mental status associated with inattention, cognitive impairment, and a change in level of consciousness, occurs in up to 80% of patients admitted to the ICU who require mechanical ventilation.3,4 Current guidelines on pain, agitation, and delirium in critically ill adults recommend an analgesia-first approach with routine assessments, since untreated pain and agitation impart negative physiological and psychological changes that contribute to poor clinical outcomes.3
Predictors: Practice guidelines recognize the complex interrelationship between pain, agitation, and delirium. Although the exact cause of delirium remains unclear, modulations in neurotransmission, brain anatomy, and physiological responses due to illness may contribute to a patient’s risk of delirium.3,5,6 Research has suggested a positive relationship between duration of delirium and anatomical changes in the brain’s white matter.1,7 Specific risk factors for ICU delirium include alcoholism, coma, history of hypertension, high severity of illness upon admission, and preexisting dementia.4 Additionally, untreated pain activates cytokines and catecholamines that promote neurotoxic processes, such as inflammation and tissue hypoxia.3,8
Drugs: Sedative-hypnotics and narcotics are commonly used in the ICU for agitation and analgesia. These medications may play a role in the disruption of neuronal transmission that affects cholinergic, dopamine, and gamma-aminobutyric acid type A receptors, contributing to an increased incidence of delirium in the ICU. Clinical studies that compared ICU patients receiving benzodiazepines with those receiving nonbenzodiazepines reported inconsistent results regarding an increased risk of delirium with benzodiazepine-based regimens.9-11
Prevention is considered the best way to manage ICU delirium. Delirium management involves treating the primary reason for ICU admission, decreasing the duration of symptoms, and reducing modifiable risk factors to promote adequate sleep.3 The use of antipsychotics is suggested for treating delirium, but the role of pharmacotherapy is limited.3,12 For many years, haloperidol was considered the drug of choice for delirium, but the most recent practice guidelines do not recommend its use, based on a lack of evidence.3 Practice guidelines instead suggest that atypical antipsychotics (e.g., quetiapine, olanzapine, ziprasidone) may reduce the duration of symptoms, but evidence supporting their efficacy remains unclear.3 Both typical and atypical antipsychotics are associated with serious adverse effects including, but not limited to, oversedation, hypotension, extrapyramidal symptoms, corrected QT-interval prolongation, and neuroleptic malignant syndrome. Therefore, it is important to closely monitor a patient’s clinical status to minimize the risk of these effects.
Postoperative Cognitive Dysfunction
Postoperative cognitive dysfunction (POCD) is a mild neurocognitive disorder involving a subtle impairment of memory, concentration, and information processing differing from that of delirium and dementia.1 The International Study of Post-Operative Cognitive Dysfunction (ISPOCD) describes cognitive dysfunction as an abnormality that is detected by using four specific neuropsychological tests post surgery.13,14 A summary of the ISPOCD findings can help healthcare providers estimate the degree of impairment after surgery. General guidelines for estimating the degree of impairment appear in TABLE 1.15
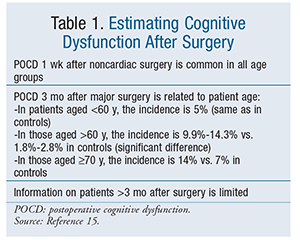
Incidence: The incidence of POCD is usually reported based on the type of surgery performed. A review of 62 studies that assessed POCD after cardiac surgery demonstrated an average decline in cognitive function of 51% (range 7%-70%) over a period of 1 to 21 days after surgery.16 A review of 46 studies that assessed POCD 7 to 21 days after noncardiac surgery found a decline in 41% to 71% of patients in studies without controls; studies with controls evidenced a decline of 6.8% to 31%.17 Patients aged >60 years were considered a higher risk and had a decline of 25.8% to 32.7%, compared with a decline of 19.2% in patients aged 40 to 59 years.17
Predictors: In an attempt to minimize or avoid the occurrence of cognitive impairment, several predictors have been identified from both cardiac and noncardiac surgeries. Predictors from a recent study in which patients had undergone coronary artery bypass surgery included female gender, increasing age, higher bleeding risk, and increased serum creatinine.18,19 Other reported predictors from cardiac and noncardiac surgeries are listed in TABLE 2.1,20-24 Correlations to the duration or type of anesthesia (regional vs. general) or to the type of anesthetic (halothane or isoflurane) have not been made.1,25

Drugs: The mechanism of action, adverse drug reactions, and administration of multiple agents are linked to cognitive dysfunction.26 The impairment is more pronounced in elderly patients, who are considered a high-risk population. Drugs that are recognized to commonly cause cognitive impairment during hospitalization include anticholinergics, benzodiazepines, and narcotics. Other drugs that are commonly used in hospitalized patients and are linked to confusion, impaired memory, or delirium in the elderly are listed in TABLE 3; alternative agents are also included.3,27-30
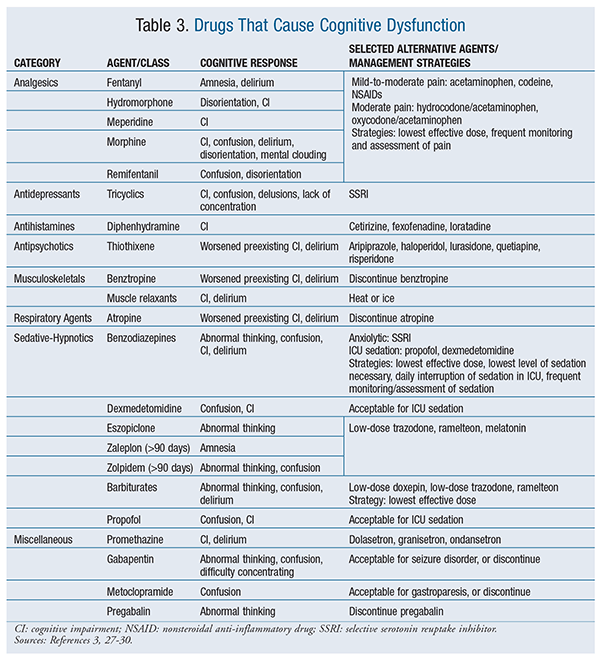
It is important to consider clinical situations in the ICU or postoperative setting that warrant the use of agents that cause cognitive dysfunction without an alternative agent. In these scenarios, strategies to minimize cognitive impairment are necessary. For clinicians, using the lowest effective dose and performing frequent monitoring and assessment of a patient’s pain, agitation, or cognition will likely reduce the duration of medication-induced cognitive impairment. Additionally, maintaining a light level of sedation and performing daily interruption of sedation in the ICU are recommended to minimize the risk of long-term cognitive dysfunction.3
Long-Term Cognitive Dysfunction
Patients who develop cognitive impairment during hospitalization are at risk for long-term cognitive dysfunction post hospital discharge. A 16% to 17% incidence of POCD 3 months after hospital discharge, with no association with type of anesthesia (regional or general) or type of surgery, has been reported.31 The development of ICU delirium is consistently associated with poorer clinical outcomes, including a longer duration of mechanical ventilation, longer length of hospitalization, higher subsequent long-term disability impacting activities of daily living, and higher mortality.32-37 Long-term cognitive impairment after critical illness in 821 patients was reported to be 40% at 3 months after discharge and 34% at 12 months; in patients aged <49 years with no preexisting problems, 34% had impairment 3 months after discharge and 20% had impairment at 12 months.8
The Role of the Pharmacist
Although the role of pharmacotherapy in treating patients with cognitive impairment is limited, the opportunity for the pharmacist to be actively involved in prevention confers the potential to reduce a patient’s risk of cognitive impairment during hospitalization and long-term cognitive dysfunction post hospital discharge. The pharmacist should consider the clinical implications of duplicate therapy, drug-disease interactions, and/or drug-drug interactions when he or she is evaluating a patient’s medication profile. See the CASE STUDY.

Conclusion
Cognitive impairment that occurs during hospitalization affects the patient’s well-being and quality of life even following hospital discharge. For pharmacists working in the hospital setting, being aware of the contributing risk factors can help minimize the development of cognitive impairment and delirium in patients who are hospitalized.
REFERENCES
1. Monk TG, Price CC. Postoperative cognitive disorders. Curr Opin Crit Care. 2011;17:376-381.
2. Salluh JI, Wang H, Schneider EB, et al. Outcome of delirium in critically ill patients: systematic review and meta-analysis. BMJ. 2015;350:h2538.
3. Barr J, Fraser GL, Puntillo K, et al. Clinical practice guidelines for the management of pain, agitation, and delirium in adult patients in the intensive care unit. Crit Care Med. 2013;41:263-306.
4. American Psychiatric Association. The Diagnostic and Statistical Manual of Mental Health Disorders, Fifth Edition. Arlington, VA: American Psychiatric Association; 2013.
5. Zaal IJ, Slooter AJ. Delirium in critically ill patients: epidemiology, pathophysiology, diagnosis and management. Drugs. 2012;72:1457-1471.
6. Rompaey B, Schuurmans MJ, Shortridge-Baggett LM, et al. Risk factors for intensive care delirium: a systematic review. Intensive Crit Care Nurs. 2008;24:98-107.
7. Morandi A, Rogers BP, Gunther ML, et al. The relationship between delirium duration, white matter integrity, and cognitive impairment in intensive care unit survivors as determined by diffusion tensor imaging: the VISIONS prospective cohort magnetic resonance imaging study. Crit Care Med. 2012;40:2182-2189.
8. Pandharipande PP, Girard TD, Jackson JC, et al. Long-term cognitive impairment after critical illness. N Engl J Med. 2013;369:1306-1316.
9. Riker RR, Shehabi Y, Bokesch PM, et al. Dexmedetomidine vs midazolam for sedation of critically ill patients: a randomized trial. JAMA. 2009;301:489-499.
10. Pandharipande PP, Pun BT, Herr DL, et al. Effect of sedation with dexmedetomidine vs lorazepam on acute brain dysfunction in mechanically ventilated patients: the MENDS randomized controlled trial. JAMA. 2007;298:2644-2653.
11. Jakob SM, Ruokonen E, Grounds RM, et al. Dexmedetomidine vs midazolam or propofol for sedation during prolonged mechanical ventilation: two randomized controlled trials. JAMA. 2012;307:1151-1160.
12. Jones SF, Pisani MA. ICU delirium: an update. Curr Opin Crit Care. 2012;18:146-151.
13. Rasmussen LS, Larsen K, Houx P, et al. The assessment of postoperative cognitive function. Acta Anaesthesiol Scand. 2001;45:275-289.
14. Moller JT, Cluitmans P, Rasmussen LS, et al. Long-term postoperative cognitive dysfunction in the elderly ISPOCD1 study. ISPOCD investigators. International Study of Post-Operative Cognitive Dysfunction. Lancet. 1998;351:857-861.
15. Seymour DG, Severn AM. Cognitive dysfunction after surgery and anaesthesia: what can we tell the grandparents? Age Ageing. 2009;38:147-150.
16. Rudolph JL, Schreiber KA, Culley DJ, et al. Measurement of post-operative cognitive dysfunction after cardiac surgery: a systematic review. Acta Anaesthesiol Scand. 2010;54:663-677.
17. Newman S, Stygall J, Hirani S, et al. Postoperative cognitive dysfunction after noncardiac surgery: a systematic review. Anesthesiology. 2007;106:572-590.
18. Habib S, Khan A, Afridi MI, et al. Frequency and predictors of cognitive decline in patients undergoing coronary artery bypass graft surgery. J Coll Physicians Surg Pak. 2014;24:543-548.
19. Boodhwani M, Rubens FD, Wozny D, et al. Predictors of early neurocognitive deficits in low-risk patients undergoing on-pump coronary artery bypass surgery. Circulation. 2006;114(1 suppl):I461-I466.
20. Newman MF, Kirchner JL, Phillips-Bute B, et al. Longitudinal assessment of neurocognitive function after coronary-artery bypass surgery. N Engl J Med. 2001;344:395-402.
21. Dupuis G, Kennedy E, Lindquist R, et al. Coronary artery bypass graft surgery and cognitive performance. Am J Crit Care. 2006;15:471-478.
22. Monk TG, Weldon BC, Garvan CW, et al. Predictors of cognitive dysfunction after major noncardiac surgery. Anesthesiology. 2008;108:18-30.
23. Suchyta MR, Hopkins RO, White J, et al. The incidence of cognitive dysfunction after ARDS. Am J Respir Crit Care Med. 2004;169:847-853.
24. MacLullich AM, Beaglehole A, Hall RJ, Meagher DJ. Delirium and long-term cognitive impairment. Int Rev Psychiatry. 2009;21:30-42.
25. Mathews SB, Arnold SE, Epperson CN. Hospitalization and cognitive decline: can the nature of the relationship be deciphered? Am J Geriatr Psychiatry. 2014;22:465-480.
26. Lund BC, Canahan RM, Egge JA, et al. Inappropriate prescribing predicts adverse drug events in older adults. Ann Pharmacother. 2010;44:957-963.
27. American Geriatrics Society 2012 Beers Criteria Update Expert Panel. American Geriatrics Society updated Beers criteria for potentially inappropriate medication use in older adults. J Am Geriatr Soc. 2012;60:616-631.
28. Pasina L, Djade CD, Lucca U, et al. Association of anticholinergic burden with cognitive and functional status in a cohort of hospitalized elderly: comparison of the anticholinergic cognitive burden scale and anticholinergic risk scale: results from the REPOSI study. Drugs Aging. 2013;30:103-112.
29. Drug Facts and Comparisons. Facts & Comparisons [database]. St. Louis, MO: Wolters Kluwer Health, Inc; March 2005. Accessed June 22, 2015.
30. Potentially harmful drugs in the elderly: Beers list. Pharmacist’s Letter. 2012;30:Detail-Document #300501. http://pharmacistsletter.therapeuticresearch.com/pl/ArticleDD.aspx?nidchk=1&cs=faculty&s=PL&pt=2&fpt=56&dd=300501&pb=PL&segment=6935&searchid=53439469. Accessed September 25, 2015.
31. Evered L, Scott DA, Silbert B, Maruff P. Postoperative cognitive dysfunction is independent of type of surgery and anesthetic. Anesth Analg. 2011;112:1179-1185.
32. Ely EW, Gautam S, Margolin R, et al. The impact of delirium in the intensive care unit on hospital length of stay. Intens Care Med. 2001;27:1892-1900.
33. Ely EW, Shintani A, Truman B, et al. Delirium as a predictor of mortality in mechanically ventilated patients in the intensive care unit. JAMA. 2004;291:1753-1762.
34. Lat I, McMillian W, Taylor S, et al. The impact of delirium on clinical outcomes in mechanically ventilated surgical and trauma patients. Crit Care Med. 2009;37:1898-1905.
35. Miller RR III, Ely EW. Delirium and cognitive dysfunction in the intensive care unit. Curr Psychiatry Rep. 2007;9:26-34.
36. Morandi A, Jackson JC, Ely EW. Delirium in the intensive care unit. Int Rev Psychiatry. 2009;21:43-58.
37. Brummel NE, Jackson JC, Pandharipande PP, et al. Delirium in the ICU and subsequent long-term disability among survivors of mechanical ventilation. Crit Care Med. 2014;42:369-377.
To comment on this article, contact rdavidson@uspharmacist.com.






