US Pharm. 2012;37(12):22-26.
ABSTRACT: In 2012, the American Cancer Society (ACS) revised its guidelines for colorectal cancer screening. The guidelines serve as a reference for recommended tests and schedules based upon risk factors for developing colorectal cancer. Screening tests are classified according to their ability to detect the presence of a cancer or to identify a patient with polyps that may become cancerous. Some tests, such as colonoscopy, require bowel preparation to cleanse the visualized site and improve evaluation. Several products are available for this purpose. If a patient is diagnosed with colorectal cancer, treatment with a combination of surgery, radiation, and/or systemic therapies can commence. Colorectal cancer is preventable, and adherence to screening guidelines can help reduce its impact on patients’ lives. The familiarity of pharmacists with these guidelines and their knowledge of screening tests can serve as a valuable resource and as encouragement for patients to follow ACS recommendations.
Guidelines for cancer screening have been available to practitioners and patients for decades to broaden awareness of cancer symptoms and to facilitate timely diagnosis and treatment. In June 2012, the American Cancer Society (ACS) released updated guidelines for early detection of colorectal cancer.1 This article will provide background information on the pathology and treatment of colorectal cancer and summarize the revised colorectal screening guidelines.
Epidemiology
The ACS estimated that, in 2012, colorectal cancer would be diagnosed in more than 103,000 Americans and would account for nearly 52,000 deaths.2 This ranks colorectal cancer as the third most common type of cancer and the third leading cause of cancer death in the United States.2 General risk factors for colorectal cancer include age older than 50 years, history of hereditary intestinal polyposis and nonpolyposis conditions, personal or family history of colorectal cancer, history of inflammatory bowel disease (i.e., ulcerative colitis or Crohn’s disease), history of Streptococcus bovis bacteremia, use of ureterosigmoidostomy, and presence of type 2 diabetes.1,3 Lifestyle factors that may contribute to the development of colorectal cancer include a diet high in animal fat, tobacco use, physical inactivity, obesity, and heavy alcohol use.1 Night-shift work for 3 or more days per week for at least 15 years has been shown to have a weak link to colorectal cancer.3 Overall, the lifetime risk of developing colorectal cancer is estimated to be 1 in 18.4
Pathology
Colorectal cancer arises in epithelial cells of the colon or rectum when multiple cumulative genetic mutations alter cell processes that normally restrict division, migration, and differentiation and confer malignant proliferative, invasive, and metastatic characteristics upon the cells.5 Once a tumor has gained malignant status, continued genetic instability leads to further alterations that can affect the cancer’s properties over time and impact sensitivity to treatment. Some of these mutations can serve as tumor markers for clinicians, such as activation of the latent gene encoding carcinoembryonic antigen, which can be measured in the blood to monitor treatment response or to detect recurrence.5
Most cases of colorectal cancer develop from polyps, growths that protrude from the mucosal surface into the gastrointestinal (GI) tract lumen.3 Three types of polyps occur: hamartoma (junior polyp), hyperplastic mucosal proliferation (hyperplastic polyp), and adenomatous polyp. Adenomatous is the only premalignant type, and fewer than 1% of these polyps become cancerous. Adenomatous polyps are found in approximately 30% of middle-aged patients and 50% of elderly patients, which highlights the importance of proper screening to identify affected individuals before the polyp becomes cancerous.
Factors affecting the probability of a polyp becoming cancerous include histologic features, size, and appearance.3 Adenomatous polyps may be tubular, villous, or tubulovillous, with villous adenomas the most likely to be cancerous. Adenomatous polyps may be either sessile (flat) or pedunculated (stalked), with sessile types being more likely to progress to cancer. Finally, polyps greater than 2.5 cm are five times more likely to be cancerous than those less than 1.5 cm (10% vs. 2%). Overall, once an adenomatous polyp forms, it takes at least 5 years of growth to reach clinical significance, suggesting the need to initiate screening and perform routine follow-up evaluation to identify polyps that are of concern before they become cancerous.3 As the developing polyp grows, it may extend into the muscular wall of the colon, where it can invade nearby blood and lymph vessels and thereby enable local or distant metastasis.1
Symptoms
The relatively slow progression of polyps into colorectal cancer enables patients to take proactive steps to reduce the risk of cancer and the impact it can have on their lives. Symptoms that should prompt a visit to a physician include a change in usual bowel habits lasting for more than a few days, a sensation of the need for a bowel movement that is not relieved by having a bowel movement, intestinal cramping or abdominal pain, prolonged weakness or fatigue, and unintended weight loss.1 Upon further evaluation, a patient also may be found to have liver-enzyme elevations and iron-deficiency anemia.4 Most of these symptoms are vague in terms of what their cause might be, likely causing many patients to fail to seek medical assistance. However, the possibility that these symptoms may represent a malignancy should be the impetus for an individual to seek a proper and prompt evaluation.
Screening Tests and Bowel Preparation
The goal of colon cancer screening is to identify and remove polyps in an asymptomatic individual before malignant transformation occurs or to identify cancer early in the course of disease, when cure is most likely.1 A variety of screening tests may be performed. These tests may be categorized according to whether they can detect both polyps and cancerous lesions (flexible sigmoidoscopy, colonoscopy, double-contrast barium enema, CT scan of colon) or identify only the presence of cancer (fecal occult blood test [FOBT], fecal immunochemical test [FIT]).1 These tests vary with respect to their degree of invasiveness, patient convenience, preparation requirements, amount of colon evaluated, and test limitations (TABLE 1).
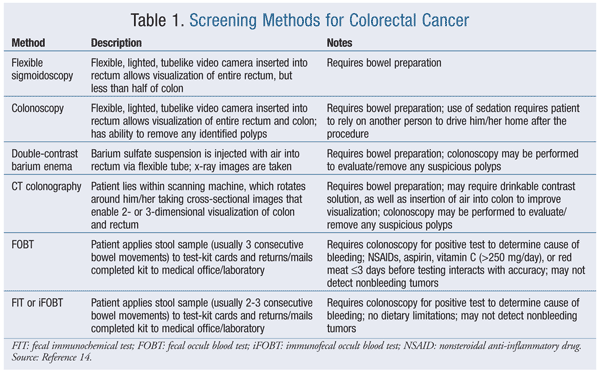
Common bowel-preparation techniques use polyethylene glycol (PEG)–based osmotic laxatives to pull fluid into the gut and stimulate repeated bowel movements that cleanse the site and allow improved visualization during evaluation.6-9 Electrolytic salts of potassium and sodium, along with the volume of water added to solubilize the active ingredient and prevent water and electrolyte imbalances, are additional components of these products. Newer products combine PEG with a stimulant laxative or rely upon osmotic actions of sulfated salts to trigger a cleansing diarrhea.9,10 (See TABLE 2 for a comparison.) Methods of bowel preparation also may be classified according to the amount of fluid (low or standard volume) the patient must imbibe.
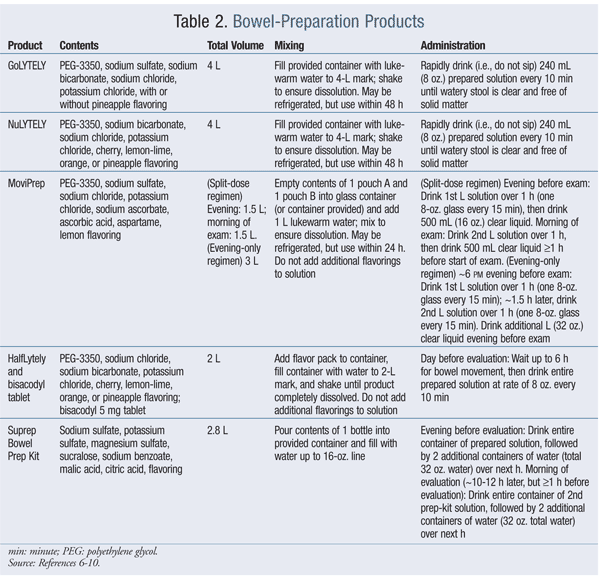
Patients must precisely follow the directions for the specific product to ensure an adequate result. The pharmacist can be a valuable source of information when dispensing these prescription-only products to the patient. These products can cause other effects in addition to voluminous diarrhea; side effects such as nausea, vomiting, bloating, abdominal distention, and electrolyte disturbances may occur, as well as seizures, cardiac arrhythmias, and ischemic colitis (although rare). Bowel-preparation kits generally are contraindicated in patients with GI obstruction, bowel perforation, gastric retention, ileus, toxic colitis, toxic megacolon, or known allergies to any of the active or inactive components. Patients with impaired gag reflex are at risk for aspiration, and these products should be used with caution under close observation.6-10 The ACS guidelines and a discussion between the patient and physician help determine which test should be employed, and when. With some tests, such as FOBT, FIT, and CT of the colon (also called virtual colonoscopy), a follow-up colonoscopy is required in the event of a positive result in order to determine the reason for occult blood in the stool or to further evaluate suspicious findings. Although virtual colonoscopy continues to gain in popularity, concerns over the degree of sensitivity, as well as the potential need for a follow-up colonoscopy, lessen its value compared with traditional colonoscopy.4
ACS Screening Guidelines
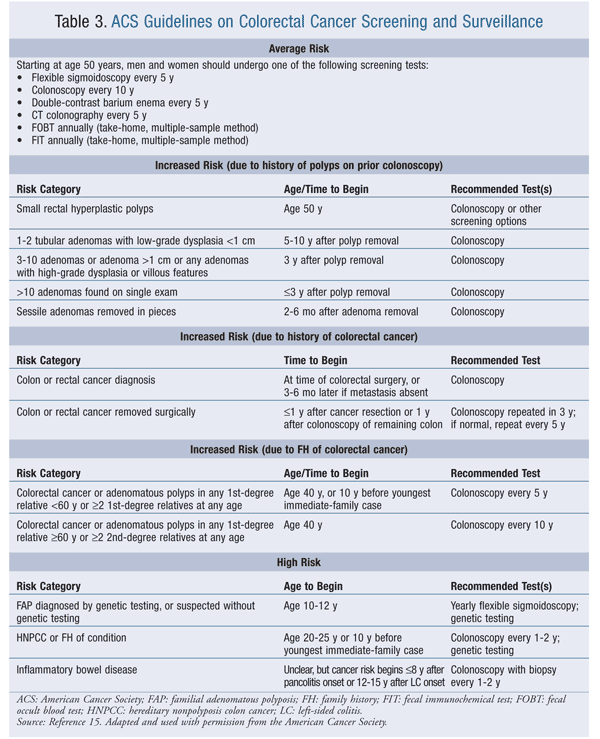
The ACS guidelines for colorectal cancer screening are stratified according to a person’s risk for the disease (TABLE 3). Patients deemed to be at increased risk for developing colorectal cancer include those with a personal history of colorectal cancer or adenomatous polyps, personal history of inflammatory bowel disease, strong family history of colorectal cancer or polyps, or family history of hereditary conditions associated with colorectal cancer (familial adenomatous polyposis, hereditary nonpolyposis colon cancer).1 Individuals not meeting criteria for increased risk are considered to be at average risk for developing colorectal cancer, and routine screening is modified to reflect this reduced risk.11 Although only about 50% of people older than 50 years adhere to the ACS guidelines, the benefits of colorectal screening cannot be ignored: Patients diagnosed in the early stage of disease have a 5-year survival rate of approximately 90%, while this value drops to 12% for patients diagnosed with metastases reflective of advanced disease.1
Diagnosis
If screening results in positive detection of a tumor, staging and classification are performed to determine treatment and prognosis. Classification involves the tumor-node-metastasis (TNM) method, in which T1-T4 indicates the depth of tumor penetration into the bowel wall, N0-N2 represents the involvement of regional lymph nodes, and M0-M1 denotes the absence or presence of distant metastases. Staging (I-IV) is based upon how high the tumor scores in these three areas, with early stages describing smaller tumors with more local involvement and later stages denoting larger tumors that have spread deeper into the mucosa and more broadly to regional lymph nodes and distant areas of the body (i.e., liver, lungs, bone, and brain).3,12
Treatment
Surgery, radiation, and systemic therapy are among the options available to manage patients with colorectal cancer. Stage and individual patient characteristics are the key determinants in treatment selection. The extent and strategy of surgical resection depend upon the tumor’s location and involvement with adjacent structures. Radiation may be administered before or after surgery to improve resectability or to reduce the probability of regional recurrence within the pelvic area. Systemic therapy relies heavily on fluorouracil combined with leucovorin, with a number of treatment regimens that vary by dose and schedule. Additional systemic options include capecitabine, irinotecan, oxaliplatin, and the monoclonal antibodies cetuximab, panitumumab, and bevacizumab.3,13 Systemic therapy is typically administered as adjuvant treatment following surgical resection in order to eliminate any remaining micrometastases.13
Conclusion
Colorectal cancer is a common form of malignancy with identifiable risk factors. Treatment can improve overall survival, especially when the disease is diagnosed at an early stage. Screening methods are available that facilitate early disease recognition. As highly accessible health care practitioners, pharmacists represent a resource for patients who have questions about screening recommendations or require a catalyst to seek indicated care. The revised guidelines developed by the ACS may be used by pharmacists, other health care providers, and patients requiring information regarding colorectal cancer screening. Pharmacists who are familiar with these guidelines can assist patients in making informed decisions about screening for colorectal cancer and can have a profound impact on their health.
REFERENCES
1. American Cancer Society. Colorectal cancer early detection.
www.cancer.org/acs/groups/cid/documents/webcontent/003170-pdf.pdf.
Accessed November 14, 2012.
2. American Cancer Society. Cancer Facts & Figures 2012. Atlanta, GA: American Cancer Society; 2012.
3. Mayer RJ. Gastrointestinal tract cancer. In: Longo DL, Fauci AS, Kasper DL, et al, eds. Harrison’s Principles of Internal Medicine. 18th ed. New York, NY: McGraw Hill Medical; 2012.
4. George TJ. Colorectal cancer. In: Abraham J, Gulley JL, Allegra CJ, eds. The Bethesda Handbook of Clinical Oncology. 3rd ed [online]. Philadelphia, PA: Lippincott Williams & Wilkins; 2010.
5. Moasser MM. Neoplasia. In: McPhee SJ, Hammer GD, eds. Pathophysiology of Disease: An Introduction to Clinical Medicine. 6th ed [online]. New York, NY: McGraw Hill Medical; 2010.
6. GoLytely (PEG-3350 and electrolytes) product information. Braintree, MA: Braintree Laboratories, Inc; November 2000.
7. NuLytely (PEG-3350, sodium chloride, sodium bicarbonate, potassium
chloride) product information. Braintree, MA: Braintree Laboratories,
Inc; February 2008.
8. MoviPrep (ascorbic acid, PEG-3350; potassium chloride, sodium
ascorbate, sodium chloride, sodium sulfate) product information.
Raleigh, NC: Salix Pharmaceuticals, Inc; April 2011.
9. HalfLytely (bisacodyl, PEG-3350, potassium chloride, sodium
bicarbonate, sodium chloride) product information. Braintree, MA:
Braintree Laboratories, Inc; July 2010.
10. SuPrep Bowel Prep Kit (sodium sulfate, potassium sulfate,
magnesium sulfate) product information. Braintree, MA: Braintree
Laboratories, Inc; August 2010.
11. National Comprehensive Cancer Network. NCCN Clinical Practice
Guidelines in Oncology. Colorectal cancer screening. Version 2.2012.
www.nccn.org/professionals/physician_gls/pdf/colorectal_screening.pdf.
Accessed November 14, 2012.
12. Edge SB, Byrd DR, Compton CC, et al, eds. AJCC Cancer Staging Manual. 7th ed. New York, NY: Springer; 2010.
13. National Comprehensive Cancer Network. NCCN Clinical Practice
Guidelines in Oncology. Colon cancer. Version 3.2012.
www.nccn.org/professionals/physician_gls/pdf/colon.pdf. Accessed October
9, 2012.
14. American Cancer Society. Colorectal cancer screening tests.
www.cancer.org/cancer/colonandrectumcancer/moreinformation/colonandrectumcancerearlydetection/colorectal-cancer-early-detection-screening-tests-used.
Accessed November 14, 2012.
15. American Cancer Society. Colorectal cancer early detection.
American Cancer Society recommendations for colorectal cancer early
detection.
www.cancer.org/cancer/colonandrectumcancer/moreinformation/colonandrectumcancerearlydetection/colorectal-cancer-early-detection-acs-recommendations.
Accessed November 14, 2012.
To comment on this article, contact rdavidson@uspharmacist.com.





