US Pharm. 2014;39(12):HS13-HS17.
ABSTRACT: Gastroparesis, a chronic condition marked by delayed gastric emptying in the absence of mechanical obstructions, occurs in nearly 2% of the population. Since gastroparesis is incurable, treatment has focused only on treating symptoms to improve quality of life. However, many patients continue to experience symptoms during therapy with currently approved medications. Azithromycin, which has been studied as an alternative to erythromycin, increases the risk of cardiovascular death from QT prolongation after only a 5-day treatment course. Azithromycin is not currently approved to treat gastroparesis, and its use for this purpose seems to have more risks than benefits. Pharmacists should intervene to prevent IV azithromycin from being ordered for gastroparesis treatment, especially when prescribed at an inappropriate dosing frequency.
Gastroparesis is a syndrome of delayed gastric emptying caused by factors other than mechanical obstructions in the stomach.1-11 The condition is estimated to affect 1.8% of the population; it is more prevalent in women (37.8 women vs. 9.6 men per 100,000 people) and is more common (88%) in younger women (mean age 41 years).1,6 Gastroparesis is incurable, and when its symptoms worsen, patients are typically admitted to the hospital for treatment. From 1995 to 2004, the number of hospital admissions for gastroparesis increased significantly, from 3,977 to 10,252 (158% increase) for primary gastroparesis and from 56,726 to 134,146 (136% increase) for secondary gastroparesis.3,6 Accordingly, healthcare costs for gastroparesis treatment have increased significantly since 2000.1
Signs and Symptoms
The most common signs and symptoms of gastroparesis are early satiety, nausea and vomiting, bloating, and epigastric pain. Pain and bloating occur in 20% and 8%, respectively, of gastroparesis patients.1 In a multicenter study from the National Institutes of Health Gastroparesis Clinical Research Consortium, abdominal pain was the dominant symptom in 18% of patients.6 Other symptoms include gastroesophageal reflux (GERD) and lack of appetite.1-10 Common symptoms of idiopathic gastroparesis are nausea (34%), pain (23%), and vomiting (19%); anxiety (36%) and depression (18%) also occur.1 Delayed gastric emptying occurs in 27% to 65% of patients with type 1 diabetes and in up to 30% of those with type 2 diabetes.1 Symptoms are aggravated by greasy foods, high-fiber foods (e.g., raw fruits and vegetables), and beverages that are carbonated or high in fat. Owing to a daily caloric intake that is less than 60% of that recommended, patients often have vitamin and mineral deficiencies.1 Complications (e.g., esophagitis, Mallory-Weiss tears, bezoars) may contribute to morbidity.5 Untreated or unsuccessfully treated gastroparesis often leads to weight loss, malnutrition, and hospitalization.1,5,9,10
Pathophysiology
Gastric contractility, which plays an important role in emptying solid food to the intestine, is modified by various intestinal peptides (e.g., motilin and cholecystokinin) and responds to physiological stimuli through the vagal nerve. When the vagal nerve in the stomach is damaged, the timing and strength of gastric contractility are disrupted, resulting in gastroparesis. Known causes of gastroparesis include diabetes (29%), certain types of surgery (13%), Parkinson’s disease (7.5%), pseudo-obstruction (4.8%), and miscellaneous factors (e.g., scleroderma, 6%).1,4,5,7 However, 35.6% of gastroparesis cases are idiopathic.
Viral infection resulting in vagal-nerve damage in the stomach is believed to cause gastroparesis.1 Diabetic patients with autonomic neuropathy resulting from glucose toxicity or altered postprandial hormone release may have gastric dysrhythmia, antral hypomotility, antral dilation, and decreased gastric tone, which impair the stomach’s ability to empty solids. Injury to the vagal nerve during surgery (especially laparoscopic fundoplication) also results in gastroparesis.2-11 Gastroparesis can occur in patients with neurologic or smooth-muscle disorders, anorexia or bulimia, GERD, endocrine disorders, autoimmune diseases, cancers, and upper-gastrointestinal (GI) surgery.5 Other causes include radiotherapy, systemic lupus erythematosus, neurologic disorders (e.g., Parkinson’s disease, stroke, multiple sclerosis, spinal injury), chemotherapy, and total parenteral nutrition.1 Additionally, the following agents may delay gastric emptying and reduce intestinal contractility: alcohol, anticholinergics, calcium channel blockers, clonidine, dopamine agonists, histamine2 receptor antagonists, lithium, nicotine, opioid analgesics, progesterone, and proton pump inhibitors.5,7
Diagnosis
In addition to a thorough medical history and physical examination, a number of diagnostic tests may be used to confirm a diagnosis of gastroparesis (TABLE 1). Scintigraphy is considered the gold standard for diagnosing gastroparesis.1,2,4,5-7,9,11

Treatment
Since gastroparesis is not curable, the primary treatment goal is to improve the patient’s quality of life through symptom management.2,4-7,9-11 First, a patient’s fluid, electrolyte, and nutritional deficiencies should be corrected. Second, the underlying causes of gastroparesis should be identified and corrected, if possible. Lifestyle adjustments and medications may be used to improve symptoms. Mild gastroparesis can be managed through proper nutrition and weight control. Medications in combination with dietary and lifestyle modifications may be required to control symptoms of moderate-to-severe gastroparesis.1,5,11
Appropriate diet and eating habits are important in minimizing the symptoms of gastroparesis, especially nausea and vomiting. Meals should be small, and six per day may be consumed. Food should be chewed well, and no carbonated liquids should be drunk with a meal. To facilitate emptying of food to the small intestine, the patient should walk or sit for at least 2 hours after a meal. Foods that are high in fat or fiber should be avoided, since they are difficult to break down and move to the intestine. In the case of severe symptoms, liquids or pureed foods should be consumed.1,2,4,11 Alcohol and smoking should be avoided, as they delay gastric emptying.7 For diabetic gastroparesis, blood glucose levels should be controlled, since hyperglycemia delays gastric emptying and interferes with erythromycin efficacy.7
Medications
Medications used to treat gastroparesis include antiemetics and motilin receptor agonists (TABLE 2).
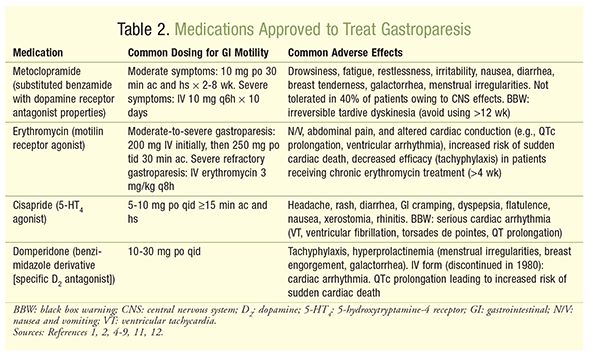
Antiemetics should be used prophylactically to prevent or reduce nausea and vomiting, especially in diabetic patients, in whom vomiting can lead to ketoacidosis and subsequent hospitalization. Metoclopramide is a prokinetic agent used most frequently to improve gastric emptying by increasing antral contractions, coordinating antral-duodenal motility, and reducing fundic relaxation.10 However, its prokinetic properties are limited to the proximal gut. The drug also has an antiemetic effect through its brainstem dopamine (D2) receptor antagonism and vagal and brainstem 5-hydroxytryptamine-3 receptor antagonism. Long-term utilization decreases its prokinetic efficacy (tachyphylaxis), but the antiemetic action is not affected.7 If a patient cannot tolerate oral metoclopramide, then the drug may be given SC or IV.1 A serious side effect of metoclopramide is irreversible tardive dyskinesia, which prevents its long-term use (avoid using >12 weeks).1,2,4-9,11
Erythromycin is a macrolide antibiotic that also has motilin receptor agonist activity, which improves gastric-emptying rates by stimulating enteric contractility. It causes pyloric relaxation and is the most potent gastric-emptying stimulant. However, erythromycin often causes cramping and abdominal pain, and its clinical effectiveness lessens (tachyphylaxis) after 4 weeks.6 Tachyphylaxis, which is believed to be caused by downregulation of motilin receptors, can be prevented by using a low dosage (125 mg twice or thrice daily).6,7 The suspension form is preferred because of its superior absorption. Erythromycin is used as an adjunct to other prokinetic agents, but it effectuates some improvement in small-bowel motility and, to a lesser degree, colonic motility.1,4-11
Cisapride improves gastric emptying by stimulating the stomach through the 5-hydroxytryptamine-4 receptors (release of acetylcholine from cholinergic nerves within the GI tract). It stimulates antral contractions, improves antroduodenal coordination and accelerates gastric emptying. Initially, the FDA approved cisapride for nocturnal heartburn; however, the drug was withdrawn from the market in 2000 owing to numerous drug interactions, serious cardiac arrhythmias, and associated deaths. Currently, cisapride is available only via a limited-access protocol from the manufacturer and is allowed for use only with strict restrictions for special conditions.1,4,5,9,10,12
Domperidone, a D2 antagonist, is highly effective at reducing GI symptoms and hospitalization by controlling nausea and vomiting and accelerating gastric emptying of solid food. Because of its antiemetic properties, domperidone reduces 67% of symptoms of diabetic gastroparesis. It has no central nervous system side effects, but it may cause hyperprolactinemia in women because of its antidopaminergic effect.1,4,5,7,8-11 Domperidone is not FDA-approved, but it is available through an investigational New Drug Application with local institutional review board approval, which requires patient informed consent and dispensing by an FDA-authorized pharmacy.
Controversies Surrounding Use of Azithromycin
Erythromycin use is limited by many drug interactions and adverse effects including QT-interval prolongation, which has been linked to sudden cardiac death.13 As a result, azithromycin has been studied as a potential alternative to erythromycin for treating gastroparesis because it has fewer adverse effects and drug interactions than does erythromycin (TABLE 3).12,14

Azithromycin is an azalide, a subclass of macrolide antibiotic derived from erythromycin, with a methyl-substituted nitrogen atom incorporated into the lactone ring. Azithromycin is assumed to have prokinetic characteristics (improved gastric emptying) similar to those of erythromycin. In one study, small-bowel manometric data of 30 patients with chronic digestive problems or documented refractory gastroparesis revealed that, when the same dosage (250 mg IV) was administered, azithromycin and erythromycin had a similar effect on antral activity.13 However, in another study, the prokinetic effect of azithromycin in vitro on rabbit duodenum was not well-sustained compared with that of clarithromycin. Thus, azithromycin appears not to be a useful therapeutic prokinetic agent.15
In 2010, a study using gastric-emptying scintigraphy showed that azithromycin’s effect on accelerating gastric emptying in adult patients with gastroparesis is equivalent to that of erythromycin (mean gastric emptying half-life of azithromycin = 10.4 ± 7.2 min vs. 11.9 ± 8.4 min for erythromycin, P = 0.3; normal half-life is 45-90 min).16 A review of articles on azithromycin used for gastroparesis treatment from 1966 to 2012 found that just two observational studies (performed during testing procedures) support this use. Since the prokinetic effects and safety profile of azithromycin are controversial, further research is required to evaluate the drug’s long-term efficacy and safety, and randomized, blinded, controlled studies should be performed before azithromycin is considered for gastroparesis treatment.14,17
It was assumed that azithromycin has a lesser incidence of QTc-interval prolongation than erythromycin.12,13 However, in a cohort study, azithromycin increased the risk of cardiovascular death compared with amoxicillin during a 5-day course of antibiotic treatment.18 On March 12, 2013, the FDA issued a warning that “azithromycin can cause abnormal changes in the electrical activity of the heart that may lead to a potentially fatal irregular heart rhythm. Patients at particular risk…include those with known risk factors such as existing QT interval prolongation, low blood levels of potassium or magnesium, a slower than normal heart rate, or use of certain drugs used to treat abnormal heart rhythms.”19 However, a recent historical cohort study of Danish adults aged 18 to 64 years that linked data on filled prescriptions, cause of death, and patient characteristics concluded that azithromycin use was not associated with an increased risk of death from cardiovascular causes in this general population of young and middle-aged adults.20
To date, the FDA has not approved azithromycin for gastroparesis treatment. Additionally, there are no controlled studies supporting off-label use of azithromycin for treating gastroparesis. However, in one case in which the author of this article was involved (no case study published owing to lack of follow-up), a physician in a teaching hospital used azithromycin 250 mg IV every 24 hours to treat a patient with gastroparesis because erythromycin was back-ordered. After 5 days, the physician changed the dosage to 125 mg every 12 hours based on a recommendation from a GI resident (for reasons unknown). After pharmacy intervention on the inappropriate interval (appropriate interval: every 24 hours based on azithromycin’s long half-life [68-72 hours]12), the physician changed the order back to the original dosage.
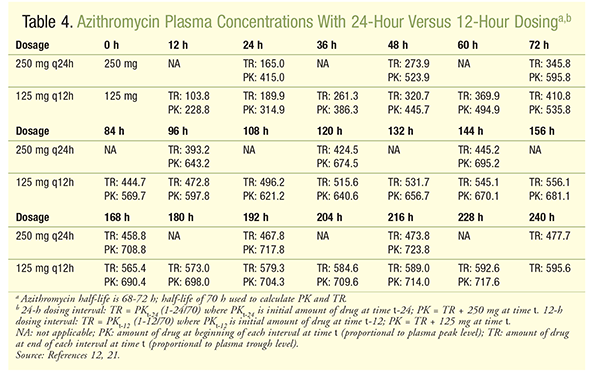
In addition to the adverse effects associated with normal-interval dosing (every 24 hours), the administration of IV azithromycin every 12 hours results in more drug accumulation in the system (TABLE 4).12,21 After 5 days (120 hours), the amount of drug remaining in the blood with the 12-hour dosing interval is 21.5% higher than that for the 24-hour interval; at 240 hours (10 days, 3.5 half-lives, 90.625% of steady state21), the amount increases to 24.7% higher than that for the 24-hour interval. In other words, trough levels of azithromycin after 5 days and 10 days are much higher with the 12-hour dosing interval compared with the 24-hour dosing interval. Since QT-interval prolongation was shown to be concentration-dependent for erythromycin and azithromycin in rats and in healthy participants in a randomized, placebo-controlled, parallel-group trial,22,23 the higher plasma concentration with 12-hour dosing could increase the risk of cardiovascular death in patients.
Conclusion
The use of IV azithromycin to treat gastroparesis is questionable and risky because of the lack of reliable randomized, blinded, controlled studies to confirm its long-term efficacy and safety. It is even riskier to administer azithromycin at an interval of every 12 hours instead of every 24 hours; the drug continues to accumulate with each 12-hour interval, leading to an increased risk of QTc-interval prolongation. Additionally, because inappropriate use of azithromycin results in long-term antibiotic resistance, IV azithromycin should be reserved for the treatment of community-acquired pneumonia or other serious bacterial infections. Given the uncertain efficacy, unknown long-term safety, and increased risk of cardiovascular death associated with short-term azithromycin use, the pharmacist should intervene to prevent IV azithromycin from being ordered to treat gastroparesis, especially at the inappropriate interval of every 12 hours.
REFERENCES
1. Hasler WL. Gastroparesis. Curr Opin Gastroenterol. 2012;28:621-628.
2. Waseem S, Moshiree B, Draganov PV. Gastroparesis: current diagnostic challenges and management considerations. World J Gastroenterol. 2009;15:25-37.
3. Wang YR, Fisher RS, Parkman HP. Gastroparesis-related hospitalizations in the United States: trends, characteristics, and outcomes, 1995-2004. Am J Gastroenterol. 2008;103:313-322.
4. Gastroparesis. Bethesda, MD: National Digestive Diseases Information Clearinghouse; 2012. NIH Publication No. 12-4348.
5. Hilas O. Management of gastroparesis. US Pharm. 2011;36(12):HS15-HS18.
6. Camilleri M, Bharucha AE, Farrugia G. Epidemiology, mechanisms, and management of diabetic gastroparesis. Clin Gastroenterol Hepatol. 2011;9:5-12.
7. Patrick A, Epstein O. Review article: gastroparesis. Aliment Pharmacol Ther. 2008;27:724-740.
8. Bonino JA, McCallum RW. Pharmacological management of chronic gastroparesis. Practical Gastroenterol. June 2007:44-59.
9. Rayner CK, Horowitz M. New management approaches for gastroparesis. Nat Clin Pract Gastroenterol Hepatol. 2005;2:454-462.
10. Buckles DC, Forster J, McCallum RW. The treatment of gastroparesis in the age of the gastric pacemaker: a review. MedGenMed. 2003;5:5.
11. Johnson DA. Gastroparesis: new guidelines. Medscape Multispecialty. www.medscape.com/viewarticle/804214. Accessed November 7, 2014.
12. Lexicomp Online [online drug database]. http://online.lexi.com/lco/action/doc/retrieve/docid/patch_f/1768824,%20_f/1831759. Accessed August 22, 2014.
13. Moshiree B, McDonald R, Hou W, Toskes PP. Comparison of the effect of azithromycin versus erythromycin on antroduodenal pressure profiles of patients with chronic functional gastrointestinal pain and gastroparesis. Dig Dis Sci. 2010;55:675-683.
14. Potter TG, Snider KR. Azithromycin for the treatment of gastroparesis. Ann Pharmacother. 2013;47:411-415.
15. Chiragh S, Begum A, Karim S. Prokinetic effect of clarithromycin and azithromycin—in vitro study on rabbit duodenum. Biomedica. 2006;22:130-134.
16. Larson JM, Tavakkoli A, Drane WE, et al. Advantages of azithromycin over erythromycin in improving the gastric emptying half-time in adults with gastroparesis. J Neurogastroenterol Motil. 2010;16:407-413.
17. Ghoshal UC. Pharmacotherapy for gastroparesis: an attempt to evaluate a safer alternative. J Neurogastroenterol Motil. 2010;16:350-352.
18. Ray WA, Murray KT, Hall K, et al. Azithromycin and the risk of cardiovascular death. N Engl J Med. 2012;366:1881-1890.
19. FDA drug safety communication: azithromycin (Zithromax or Zmax) and the risk of potentially fatal heart rhythms. www.fda.gov/Drugs/DrugSafety/ucm341822.htm. Accessed August 9, 2014.
20. Svanström H, Pasternak B, Hviid A. Use of azithromycin and death from cardiovascular causes. N Engl J Med. 2013;368:1704-1712.
21. DiPiro JT, Spruill WJ, Wade WE, et al, eds. Concepts in Clinical Pharmacokinetics. 4th ed. Bethesda, MD: American Society of Health-System Pharmacists; 2005:32,49.
22. Ohtani H, Taninaka C, Hanada E, et al. Comparative pharmacodynamic analysis of Q-T interval prolongation induced by the macrolides clarithromycin, roxithromycin, and azithromycin in rats. Antimicrob Agents Chemother. 2000;44:2630-2637.
23. Zithromax (azithromycin for injection) product information. New York, NY: Pfizer Inc; July 2014.
To comment on this article, contact rdavidson@uspharmacist.com.





