US Pharm. 2015;40(8):25-29.
ABSTRACT: There have been recent updates to testosterone product labeling and additional warnings concerning increased adverse outcomes. With these latest safety updates and required boxed warnings, pharmacists are in an influential position to promote evidence-based counseling on testosterone replacement therapy (TRT) and to advocate for proper follow-up and monitoring. Additionally, pharmacists are in an accessible position within communities to answer patients’ questions regarding TRT efficacy and safety.
The FDA has made three changes to the labeling of testosterone products in recent years. In 2009, a required boxed warning was added to all transdermal gel products because of the risk of transference to women and children.1 Based on post-marketing reports, a warning of venous thromboembolism risk was added to all testosterone products in 2014.2 In March 2015, the FDA released a Drug Safety Communication (DSC) regarding testosterone replacement therapy (TRT).3 This communication was directed to all manufacturers of testosterone products with the mandate that the label include a warning of an increased risk of heart attack and stroke. The DSC also notified manufacturers of a requirement to clearly state the approved indication as: “Testosterone is FDA-approved as replacement therapy only for men who have low testosterone levels due to disorders of the testicles, pituitary gland, or brain that cause a condition called hypo-gonadism.”3
The FDA has stated that there appears to be improper prescribing of TRT. It is now aware that “testosterone is being used extensively in attempts to relieve symptoms in men who have low testosterone for no apparent reason other than aging.”3
Adult Male Hypogonadism
It is important to understand hypogonadism before reviewing the cardiovascular trials leading to these FDA updates. Male hypogonadism is a clinical syndrome of congenital or acquired origin manifesting as decreased testosterone levels with or without decreased spermatogenesis. It will present with signs and symptoms of androgen deficiency. Primary hypogonadism, a disease of testicular function, typically presents as low testosterone concentration with or without impaired spermatogenesis and with elevated luteinizing hormone (LH) and follicle-stimulating hormone (FSH) concentrations.4 Secondary hypogonadism, or hypogonadotropic hypogonadism, is a dysfunction of the hypothalamic-pituitary-gonadal axis presenting as low testosterone concentration with or without impaired spermatogenesis and with reduced or normal LH and FSH concentrations.4
Men typically seek treatment for hypogonadism based on the symptoms they experience with continued low testosterone levels. Per the Endocrine Society’s clinical guidelines for testosterone therapy in adult men with androgen deficiency syndromes, certain symptoms, such as decreased energy, depressed mood, and reduced muscle strength, are general and not indicative of male hypogonadism initially.4 More specific symptoms seen in hypogonadism include decreased sexual desire, loss of body hair, and breast discomfort, all of which may be alarming to patients. TRT has been shown to be beneficial in men experiencing these symptoms. However, the safety of long-term therapy has been questioned in various studies.
Cardiovascular issues with TRT were first noted in the Testosterone in Older Men with Mobility Limitations (TOM) trial and reported in the peer-reviewed journal Contemporary Clinical Trials in 2009.5 The TOM trial assessed the effects of testosterone administration on voluntary muscle strength in older men. The trial was terminated early because of the significantly higher incidence of cardiovascular events in the testosterone group.5
In 2014, two separate studies assessing cardiovascular adverse events with testosterone were published.6,7 The studies suggest that there are increased cardiovascular risks in elderly men, regardless of preexisting adverse cardiovascular history. Vigen et al reported the findings of a retrospective cohort study assessing elderly men in the Veterans Affairs healthcare system with low testosterone levels and coronary artery disease.6 Adjusting for the presence of coronary artery disease, this study found a statistically significant increase in adverse outcomes, including risk of stroke, heart attack, and death in the group receiving TRT compared to the group with no TRT (hazard ratio [HR] 1.29, 95% CI, 1.04-1.58).6 Finkle et al reported the results of a meta-analysis that found that men aged 65 years, regardless of cardiovascular history, had a twofold increased risk of heart attack (risk ratio [RR] 2.19, 1.27-3.77). It also found that men <65 years of age, with a preexisting history of heart disease, had a two- to threefold increased risk of heart attack (RR 2.90, 1.49-5.62).7
The studies by Finkle et al and Vigen et al have caused controversy in the field of andrology. Experts have published their objections to these findings, stating that the results from the two retrospective analyses contradict over 20 years of literature, as described in a letter to the editor in a subsequent edition of the Journal of the American Medical Association (JAMA).8 The Endocrine Society,9 American Urological Association,10 and the Sexual Medicine Society of North America11 have all released statements addressing this concern and noting the evidence supporting the contrary. Of note, there have been no prospective, randomized, controlled trials powered sufficiently to assess testosterone therapy and measure cardiovascular events as the primary outcome. There is currently a study being conducted by the National Institute on Aging to determine if testosterone treatment in men will improve their physical and sexual function, and decrease risk factors for cardiovascular disease and diabetes.12 However, the principal investigator, Peter J. Snyder, MD, has warned that the study is not designed to evaluate whether cardiovascular risk actually increases.13
With these recent updates on the safety of testosterone products and changes to the established boxed warning, pharmacists are in an influential position to promote evidence-based counseling on TRT and to advocate for proper follow-up and monitoring. The remainder of this article will focus on key information pharmacists should share with patients regarding the safety and efficacy of TRT.

Treatment Guidelines
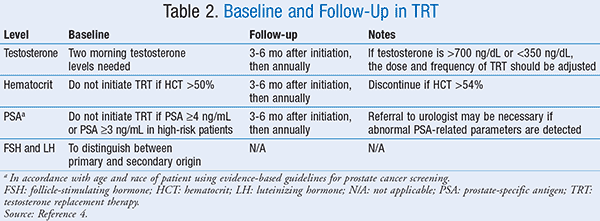
Along with signs and symptoms (TABLE 1),4 male hypogonadism is confirmed by two morning testosterone levels on separate occasions. Repeating the testosterone level is necessary because testosterone levels fluctuate throughout the day. Therefore, one low reading may not be indicative of chronically low levels. As shown in TABLE 2,4 LH and FSH levels are drawn once to determine primary or secondary origin. To assess the patient’s risk of prostate cancer, a prostate-specific antigen (PSA) is recommended at baseline, in 3 to 6 months, and then annually. Hematocrit levels are recommended on the same schedule as PSA to evaluate the risk for developing hypoxia and sleep apnea. Additional laboratory tests may be recommended for other medical conditions in the individual patient. Pharmacists can discuss these monitoring parameters with their patients during counseling sessions to help improve awareness and compliance with proper monitoring.
Counseling
When initially counseling a patient starting TRT, it is essential that he understand that this therapy is used to treat hypogonadism. It is not effective as mono-therapy for erectile dysfunction. However, there are data suggesting a benefit of TRT in hypogonadal males who have failed phosphodiesterase type 5 (PDE5) inhibitor treatment.14
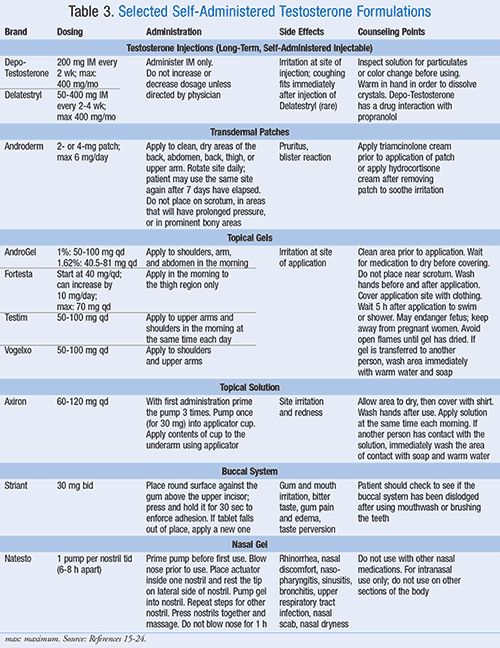
TRT products should always be used as directed by the physician.15,16 It is important to advise the patient to clean the area prior to administration.17-22 With gel and solution products, the patient must take precautions to avoid transference to women and children, such as washing hands after application and covering the application site with clothing; the products may also cause fetal harm.15-24 Other general counseling points include the following:
- Watch for potential signs and symptoms of a thromboembolism (e.g., shortness of breath, chest pain, swelling of extremities)
- If a patient misses a dose, apply the dose immediately. However, if it is close to the next dose, the patient should skip the missed dose and apply at the next scheduled time. DO NOT double-up on dosing
- If the patient is diabetic, monitor blood glucose levels closely while taking TRT. Contact physician if blood glucose levels are not within therapeutic range despite the use of diabetic medication
- Contact physician if the patient experiences increased fatigue, prolonged erection, irregular breathing, or skin color changes
- Caution patients who are using oxyphenbutazone. Using testosterone in conjunction with oxyphenbutazone may increase the medication serum levels
- Counsel patients regarding the possible adverse effects including gynecomastia, sleep apnea, dyslipidemia, hypercalcemia, and an increased prevalence of acne
- A summary of counseling points by dosage form and brand name for self-administered testosterone formulations is included in TABLE 3.15-24
In addition to the preparations mentioned in TABLE 3, two other formulations of testosterone must be administered in the healthcare provider’s office, Aveed and Testopel.25,26 Thus, the community pharmacist may not have the opportunity to counsel patients on these dosage forms. Aveed (testosterone undecanoate) is a long-term injectable. Patients must be monitored for 30 minutes at the physician’s office after administration for any adverse reactions. Dosing for Aveed is 750 mg initially, then 750 mg 4 weeks later, then repeat every 10 weeks.25
Testopel is a testosterone pellet inserted by a clinician just under the skin in the hip or another fatty area.26 Patients are advised to speak to their physician if they have prolonged irritation or rash at the sign of insertion. Testopel has similar drug interactions and adverse events as other forms of TRT, such as decreased insulin requirements and prolonged erection.
Role of the Pharmacist
There have been recent updates on testosterone product labeling and additional warnings concerning increased adverse outcomes. Pharmacists play a key role in managing TRT in a safe and effective manner, as they are in an accessible position within communities to counsel patients and answer questions. With proper counseling of patients on TRT dosage forms and side effects and reminders to have their test results monitored appropriately, TRT can be used to improve hypogonadism and optimally reach the goal of normal testosterone levels.
Pharmacists should emphasize the following counseling points when discussing TRT with a patient:
- Keep away from women and children
- Wash hands before and after application (gels, solutions)
- Cover application area with a T-shirt (gels, solutions)
- Have testosterone, hematocrit (HCT), and PSA levels checked annually once stable
- Contact your physician immediately if you have severe reactions to the testosterone.
Pharmacists should always be aware of any other medications the patient is currently taking and possible drug interactions. Drug interactions noted in the products’ package inserts include insulin, anticoagulants, and corticosteroids. Pharmacists are the last healthcare professional the patient may see before taking his TRT. Therefore, pharmacists play a vital role in the improvement of their patients’ hypogonadism and the safety and efficacy of their therapy.
REFERENCES
1. FDA. Topical testosterone gel products (marketed as AndroGel 1% and Testim 1%): secondary exposure of children to topical testosterone products. Postmarket Reviews—Volume 2, Number 3, 2009. www.fda.gov/Drugs/DrugSafety/DrugSafetyNewsletter/ucm189806.htm. Accessed March 28, 2015.
2. FDA. Testosterone products: FDA/CDER statement—risk of venous blood clots. June 20, 2014. www.fda.gov/Safety/MedWatch/SafetyInformation/SafetyAlertsforHumanMedicalProducts/ucm402054.htm. Accessed March 28, 2015.
3. FDA Drug Safety Communication: FDA cautions about using testosterone products for low testosterone due to aging; requires labeling change to inform of possible increased risk of heart attack and stroke with use. March 3, 2015. www.fda.gov/Drugs/DrugSafety/ucm436259.htm. Accessed March 28, 2015.
4. Bhasin S, Cunningham GR, Hayes FJ, et al. Testosterone therapy in men with androgen deficiency syndromes: an Endocrine Society clinical practice guideline. J Clin Endocrinol Metab. 2010;95(6):2536-2559.
5. Basaria S, Coviello A, Travison T, et al. Adverse events associated with testosterone administration. N Engl J Med. 2010;363(2):109-122.
6. Vigen R, O’Donnell CI, Barón AE, et al. Association of testosterone therapy with mortality, myocardial infarction, and stroke in men with low testosterone levels. JAMA. 2013;310(17):1829-1836.
7. Finkle W, Greenland S, Ridgeway G, et al. Increased risk of non-fatal myocardial infarction following testosterone therapy prescription in men. PLoS One. 2014;9(1):e85805.
8. Morgentaler A, Traish A, Kacker R. Deaths and cardiovascular events in men receiving testosterone. JAMA. 2014;311(9):961-962.
9. The Endocrine Society. The risk of cardiovascular events in men receiving testosterone therapy: an Endocrine Society statement. February 7, 2014. https://www.endocrine.org/~/media/endosociety/Files/Advocacy%20and%20Outreach/Position%20Statements/Other%20Statements/The%20Risk%20of%20Cardiovascular%20Events%20in%20Men%20Receiving%20Testosterone%20Therapy.pdf. Accessed May 13, 2015.
10. American Urological Association. AUA position statement on testosterone therapy. February 2014. https://www.auanet.org/about/testosterone-therapy.cfm. Accessed May 13, 2015.
11. Sexual Medicine Society of North America. Position statements. Testosterone therapy and cardiovascular risks. www.smsna.org/V1/about/position-statements. Accessed May 13, 2015.
12. The Testosterone Trial. About the T-Trial. http://rt5.cceb.upenn.edu/portal/page/portal/T-Trial%20Portal/T-Trial%20Public%20Page%20-%20About. Accessed May 10, 2015.
13. Lamb T. The benefits and risks of popular testosterone replacement therapy for older men are not fully known. Drug Injury Watch. May 1, 2014. www.drug-injury.com/druginjurycom/2014/05/cardiovascular-risks-side-effects-androgel-testosterone-axiron-trt-testim-heart-attacks-stokes-drug-safety-unknown.html. Accessed May 10, 2015.
14. Shabsigh R, Kaufman JM, Steidle C, Padma-Nathan H. Randomized study of testosterone gel as adjunctive therapy to sildenafil in hypogonadal men with erectile dysfunction who do not respond to sildenafil alone. J Urol. 2008;179(5 suppl):S97-S102.
15. Depo-Testosterone (testosterone cypionate injection) package insert. Kirkland, Quebec: Pfizer Canada Inc; 2015.
16. Delatestryl (testosterone enanthate injection) package insert. Lexington, MA: Indevus Pharmaceuticals, Inc; 2007.17. Androderm (testosterone transdermal system) package insert. Corona, CA: Watson Pharma, Inc; 2001.
18. AndroGel (testosterone gel) package insert. Marietta, GA: Solvay Pharmaceuticals, Inc; 2004.
19. Fortesta (testosterone gel) package insert. Malvern, PA: Endo Pharmaceuticals, Inc; 2014.
20. Testim (testosterone gel) package insert. Chesterbrook, PA: Auxilium Pharmaceuticals, Inc; 2014.
21. Vogelxo (testosterone gel) package insert. Maple Grove, MN: Upsher-Smith Pharmaceuticals, Inc; 2014.
22. Axiron (testosterone topical Solution) package insert. Indianapolis, IN: Eli Lilly and Company; 2014.
23. Striant (testosterone buccal system) package insert. Livingston, NJ: Columbia Laboratories, Inc; 2003.
24. Natesto (testosterone nasal gel) package insert. Malvern, PA: Endo Pharmaceuticals, Inc; May 2015.
25. Aveed (testosterone undecanoate injection) package insert. Malvern, PA: Endo Pharmaceuticals, Inc; 2014.
26. Testopel (testosterone pellets) package insert. Rye, NY: Bartor Pharmacal; 2013.
To comment on this article, contact rdavidson@uspharmacist.com.





