Inflammatory bowel disease (IBD) is a relapsing-remitting gastrointestinal (GI) disorder involving areas from the mouth to the anus.1,2 Crohn's disease (CD) is a chronic form of IBD that can extend through the various layers of the GI tract, resulting in intestinal and extraintestinal complications. Another form of IBD is ulcerative colitis (UC), which primarily affects the rectum and colon; however, this article will address only CD.1,2
Annually in the United States, CD affects 5 in 100,000 people aged 15 to 30 years and 50 to 80 years. CD has its greatest onset in adolescence and young adulthood (age 15 to 25 years), and some studies have discovered a second peak in incidence between ages 50 and 80 years. Rates in men and women are similar, although some studies report a higher incidence in females.1,3,4 The equal sex ratio speaks against a marked occupational or hormonal influence on the disease.
The exact cause of CD is unknown. Theories concentrate on the role of infectious, genetic, and immunologic factors. It has been suggested that inflammation is caused by the stimulation of T cells and the overproduction of proinflammatory cytokines by bacterial products generated through GI-tract colonization. Another theory is that individuals who have family members with CD are at a greater risk for developing the disease. The coding for nucleotide-binding oligomerization domain 2 (NOD2), sometimes observed on chromosome 16, has been found to positively correlate with CD. NOD2 is a protein seen in white blood cells and GI epithelial cells and may be responsible for the recognition and destruction of bacterial products in the stomach. Other factors, such as diet, stress, smoking, and use of nonsteroidal anti-inflammatory drugs, may also contribute to the pathology of the disease.1,3-6
PATHOPHYSIOLOGY
Normal epithelium forms a protective barrier for the intestinal tract. Injury, bacterial products, or exogenous agents can compromise this barrier. Irritants interact with immune cells, facilitating an immune response. Further, proinflammatory cytokines (interleukin-12, tumor necrosis factor [TNF], and interferon gamma [IFN-G]) are usually elevated in CD; TNF is highly involved in this process, as it promotes cell death.1,3,5,7 Endothelial cells recruit leukocytes, fibroblasts, and epithelium into the mucosa from the vascular space, resulting in granulomas, a histopathologic hallmark of CD damage to the GI tract.1,8,9 Granulomatous lesions can extend through the intestinal wall and into the mesentery and regional lymph nodes. Chronic inflammation ultimately thickens the bowel wall and eventually narrows the lumen.
CLINICAL MANIFESTATIONS
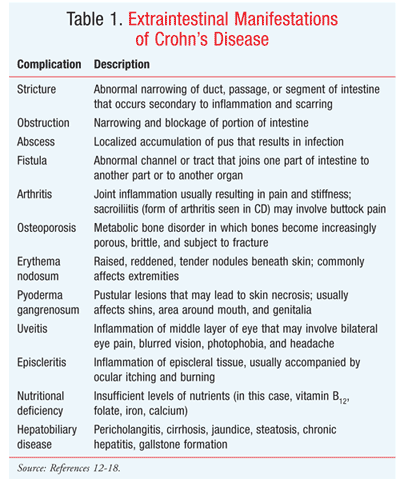
The terminal ileum is the most common site of CD. Discontinuous lesions, which consist of normal bowel separating segments of diseased bowel, may occur, increasing the probability of fistulas, obstruction, perforations, and stricture.2 Symptoms of CD include chronic or nocturnal diarrhea, abdominal pain, weight loss, chills, fever, and rectal bleeding.4,10 Cramps, distention, constipation, vomiting, and abscess formation may occur as well.8,10,11 CD also may be associated with extraintestinal manifestations, as specified in TABLE 1.12-18
DIAGNOSIS AND CLASSIFICATION
Endoscopy determines the location of inflammation and collects tissue biopsy for evaluation.5,19,20 Wireless capsule endoscopy is a recently developed procedure that is less invasive; the patient swallows a capsule that records images of the GI tract.13 Anti-Saccharomyces cerevisiae antibodies, which are present in more than 50% of patients, may also be helpful in diagnosis.11,15
The Crohn's Disease Activity Index is used to assess the severity of the disease and to guide treatment selection.19 The classification of CD is described in TABLE 2.19,20

THERAPEUTIC MANAGEMENT OPTIONS
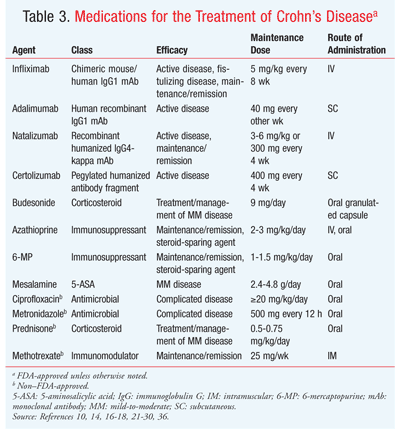
The objects of therapy for managing CD are to alleviate symptoms, resolve complications, and induce remission.1 Several classes of drugs have been used to effectively achieve these goals (TABLE 3).10,14,16-18,21-30
Aminosalicylates
Aminosalicylates represent first-line therapy for mild-to-moderate CD. These 5-aminosalicylic acid (5-ASA) compounds act on epithelial cells, moderating the release of lipid mediators, cytokines, and reactive oxygen species.19 Examples include sulfasalazine, mesalamine, olsalazine, and balsalazide. Studies of sulfasalazine 3 g to 5 g/day demonstrated an elevated rate of remission (40%-50%) at 16 weeks.21 Sulfasalazine contains a sulfa moiety that is responsible for most allergic reactions (e.g., Stevens-Johnson syndrome). Adverse effects include anorexia, headache, nausea, GI distress, and oligospermia. Other possible reactions include pancreatitis, agranulocytosis, and alveolitis.8
Mesalamine, a newer 5-ASA compound, does not contain a sulfa moiety and causes fewer allergic reactions. It demonstrates dose-related efficacy, but not dose-related adverse effects. Mesalamine is available in oral, rectal, and topical formulations; however, only the controlled-release capsule (Pentasa) and the delayed-release tablet (Asacol) are used for CD. Studies have found that these agents lack efficacy in CD that affects the small bowel and have a minimal impact on maintaining remission (≤50%).8,11,19
Glucocorticoids
Corticosteroids reduce inflammation and induce remission of active CD.8 They are commonly prescribed when 5-ASA compounds are ineffective.11 These agents work by suppressing interleukin transcription and arachidonic-acid metabolism and by stimulating apoptosis of lymphocytes in the gut.3,8 Adverse effects include moon face, acne, weight gain, and dyspepsia. Acute adrenal insufficiency may result from abrupt withdrawal.3,8 Corticosteroids may induce hypertension, diabetes, and osteoporosis; however, these conditions can be avoided with proper prophylaxis. Lack of efficacy in maintaining CD remission is a limitation of these agents.3,8,11 One study found 50% remission at 2 weeks, but not at 8 weeks.16
Immunosuppressants
Thiopurines have been shown to lessen the immune response, and they are effective in maintaining remission of moderate-to-severe CD and in treating fistulizing disease. They are traditionally employed after 5-ASA compounds have been tried. Azathioprine and 6-mercaptopurine are steroid-sparing agents that induce cell apoptosis. Adverse effects of these agents include flulike symptoms, hepatotoxicity, pancreatitis, and increased risk of lymphoma.3,8,11
By inhibiting cytokine synthesis, methotrexate is effective for inducing remission and preventing relapse in patients with CD. Adverse effects of methotrexate include nausea, vomiting, diarrhea, stomatitis, hepatotoxicity, and pneumonitis. Methotrexate therapy has a slow onset of action (3-6 months) and requires monitoring.1,9
Antimicrobials
Antimicrobial agents are helpful in the treatment of mild-to-moderate CD associated with fistulas and abscesses. Although fluoroquinolones and metronidazole have been used as therapy options, one study suggested that metronidazole is ineffective for treating small-bowel CD at doses ≤20 mg/kg/day.31 Metronidazole has been associated with yeast infections and neuropathy. Patients should avoid consuming alcohol while taking metronidazole to prevent severe nausea, vomiting, and toxicity (disulfiram-like reactions).3,11 Additionally, fluoroquinolones may cause yeast infections and rupture of tendons.
Biologicals
Biological agents target specific mediators of the immune system. Current FDA-approved monoclonal antibodies (mAb) for the treatment of CD include infliximab (Remicade); adalimumab (Humira); natalizumab (Tysabri); and certolizumab (Cimzia).11,14,15,17,18,21-29,
Infliximab: Infliximab, a chimeric mouse/human mAb, was FDA-approved in 2003 for reducing signs and symptoms and maintaining remission in moderate-to-severe CD. It also is useful in the treatment of fistulizing CD. Infliximab neutralizes the biological activity of TNF-alpha by binding to the cytokine and preventing it from binding to its receptors. Infliximab is immunogenic and may be associated with infusion reactions and decreased efficacy.11,14,17,32
Results from the ACCENT (A Crohn's Disease Clinical Trial Evaluating Infliximab in a New Long-Term Treatment Regimen) I trial indicate that patients with CD who responded to an initial dose (5 mg/kg) of infliximab and maintained treatment every 8 weeks were more likely to be in remission at 30 and 54 weeks. These patients also discontinued corticosteroid therapy and maintained longer periods of remission.16 Use of infliximab has raised questions about abscess formation due to rapid healing of fistulas. The ACCENT II trial evaluated these concerns and found no basis for them.14
Adalimumab: Adalimumab, a human recombinant agent introduced in 2007, is associated with an overall decrease in immunogenicity, improved safety, and similar efficacy.11,22-25 CHARM (Crohn's Trial of the Fully Human Antibody Adalimumab for Remission Maintenance) assessed the efficacy of adalimumab in moderate-to-severe CD. The researchers found that 47% and 41% of patients receiving 40 mg subcutaneously (SC) weekly and 40% and 36% of those receiving 40 mg SC every other week (EOW) maintained remission at 26 weeks and 56 weeks, respectively.22
The CLASSIC (Clinical Assessment of Adalimumab Safety and Efficacy Studied as Induction Therapy in Crohn's Disease) I and II trials evaluated the use of adalimumab for induction and maintenance therapy in CD. The CLASSIC I trial found adalimumab to be superior to placebo for moderate-to-severe CD in patients naïve to anti-TNF therapy. The CLASSIC II trial yielded remission rates of 83% and 79% for up to 56 weeks in patients naïve to anti-TNF therapy who received 40 mg SC weekly or 40 mg SC EOW, respectively.23,24
The GAIN (Gauging Adalimumab Efficacy in Infliximab Nonresponders) trial investigated the use of adalimumab in infliximab-refractory CD. The investigators concluded that adalimumab had some efficacy in infliximab-refractory or -intolerant patients.25
Natalizumab: Natalizumab, a drug commonly used to manage multiple sclerosis, gained FDA approval in January 2008 for induction and remission maintenance therapy for CD. The ENACT (Efficacy of Natalizumab as Active Crohn's Therapy) I and II trials were conducted to evaluate patients' response to treatment. Patients who responded to treatment during the initial trial progressed to the second trial. ENACT II showed higher rates of sustained response (61%) and remission (44%) at 36 weeks. This trial also reported one death from progressive multifocal leukoencephalopathy, an opportunistic viral infection of the brain that may result in severe disability or death.26
ENCORE (European National Crohn's Observational Registry) was a postmarketing surveillance registry used to collect long-term safety data. Sixty percent of patients receiving natalizumab demonstrated response and 48% retained clinical response at 12 weeks, and the drug was well tolerated.27
Certolizumab: Certolizumab pegol is a pegylated humanized antibody fragment that was FDA-approved in April 2008. The PRECISE (PEGylated Antibody Fragment Evaluation in Crohn's Disease Safety and Efficacy) 1 trial found that patients treated with certolizumab demonstrated clinical response at 6 weeks (22%) and 26 (12%) weeks.18,28,29 There was no significant difference in remission rates. Advantages of this therapy include longer half-life, SC administration, and once-monthly dosing.11,18,28,29
Other Therapies
Omega-3 fatty acids, probiotics, changes in diet, and surgery have been used in the treatment of CD.11 The goal of surgery is to control symptoms that do not respond to medications. Although it is commonly indicated for obstruction, fistula, and abscess formation, surgery is not a curative option.11
Emerging Therapies
Sargramostim (Leukine) is a recombinant human granulocyte-macrophage colony-stimulating factor that triggers the proliferation and differentiation of neutrophils, eosinophils, monocytes, and macrophages. It also may block the growth of tumor cells. Sargramostim was initially formulated with edetate disodium, which led to its removal from the market in 2000; however, it has been reformulated to a liquid solution that does not contain this compound.
A humanized anti-IFN-G antibody, fontolizumab may have clinical activity and tolerability in moderate-to-severe CD.30 Further clinical trials are necessary to determine the agent's clinical applicability.
Alicaforsen is a biologic response modifier that inhibits intercellular adhesion molecule-1 (ICAM-1). ICAM-1 seems to play a role in intestinal inflammation. Alicaforsen is currently undergoing phase III clinical trials for the treatment of CD. Initial results indicate that this agent may be more useful for UC than for CD.30
Top-Down Versus Step-Up Therapy
Traditional management of CD involves step-up therapy, a method in which less-aggressive agents (e.g., 5-ASA compounds, antimicrobials) are used prior to instituting agents that are more aggressive (e.g., immunosuppressants, biologicals). Conversely, top-down therapy employs biologicals and other aggressive therapies first. One study concluded that top-down therapy is superior to sequential step-up therapy for reducing exposure to glucocorticosteroids and achieving long-lasting remission.33 However, the case for top-down treatment strategies in the treatment of CD remains controversial. There is a risk of overtreatment of patients with mild cases of CD; also, there are no discernible markers to identify disease severity. Further studies must be conducted, as many top-down strategies have yet to be evaluated.34,35
CONCLUSION
Emerging therapies for the management of CD offer patients and health care providers more options for combating this disease.18 Recent studies offer more information concerning the most effective use of these therapies.30,33 Study and application of these concepts may lead to novel approaches in the diagnosis and management of CD in the near future.
REFERENCES
2009;132:1-9.
1. DiPiro JT, Schade RR. Irritable bowel disease. In: DePiro JT, Talbert RL, Yee JC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 6th ed. New York, NY: McGraw-Hill Medical; 2005:649-664.
2. Braus NA, Elliott DE. Advances in the pathogenesis and treatment of IBD. Clin Immunol.
3. Steinhart AH. Crohn's and Colitis: Understanding and Managing IBD. Toronto, ON: Robert Rose Inc; 2006.
4. Cho J. Inflammatory bowel disease: genetic and epidemiologic considerations. World J Gastroenterology. 2008;14:338-347.
5. Knösel T, Schewe C, Petersen N, et al. Prevalence of infectious pathogens in Crohn's disease. Pathol Res Pract. 2009;205:223-230.
6. Chisholm-Burns MA, Wells BG, Schwinghammer TL, et al, eds. Pharmacotherapy Principles & Practice. New York, NY: McGraw-Hill Medical; 2008:282.
7. Evans PE, Pardi DS. Extraintestinal manifestations of inflammatory bowel disease: focus on the musculoskeletal, dermatologic, and ocular manifestations. MedGenMed. 2007;9:55.
8. Carter MJ, Lobo AJ, Travis SP. Guidelines for the management of inflammatory bowel disease in adults. Gut. 2004;53:v1-v16.
9. Shanahan F. Crohn's disease. Lancet. 2003;359:62-69.
10. Hanauer SB, Sandborn W. Management of Crohn's disease in adults. Am J Gastroenterol. 2001;96:635-643.
11. Podolsky DK. Inflammatory bowel disease. New Engl J Med. 2002;347:417-429.
12. Caprilli R, Gassull MA, Escher JC, et al. European evidence based consensus on the diagnosis and management of Crohn's disease: special situations. Gut. 2006;55(suppl 1):i36-i58.
13. Swain P. Wireless capsule endoscopy and Crohn's disease. Gut. 2005;54:323-326.
14. Sands BE, Blank MA, Diamond RH, et al. Maintenance infliximab does not result in increased abscess development in fistulizing Crohn's disease: results from the ACCENT II study. Aliment Pharmacol Ther. 2006;23:1127-1136.
15. Vermeire S, Van Assche G, Rutgeerts P. Laboratory markers in IBD: useful, magic, or unnecessary toys? Gut. 2006;55:426-431.
16. Thomsen O, Cortot A. A comparison of budesonide and mesalamine for active Crohn's disease. International Budesonide-Mesalamine Study Group. New Engl J Med. 1998;339:370-374.
17. Honeywell M, Touchstone K, Caspi A. Remicade (infliximab). P&T Product Profiler. 2007;32:1-43.
18. Rutgeerts P, Schreiber S, Feagan B, et al. Certolizumab pegol, a monthly subcutaneously administered Fc-free anti-TNFα, improves health-related quality of life in patients with moderate to severe Crohn's disease. Int J Colorectal Dis. 2008;23:289-296.
19. Van Deventer SJ. Tumour necrosis factor and Crohn's disease. Gut. 1997;40:443-448.
20. Stange EF, Travis SP, Vermeire S, et al. European evidence based consensus on the diagnosis and management of Crohn's disease: definitions and diagnosis. Gut. 2006;55(suppl 1):i1-i15.
21. Malchow H, Ewe K, Brandes JW, et. al. European Cooperative Crohn's Disease Study (ECCDS): results of drug treatment. Gastroenterology. 1984;86:246-266.
22. Colombel JF, Sandborn WJ, Rutgeerts P, et al. Adalimumab for maintenance of clinical response and remission in patients with Crohn's disease: the CHARM trial. Gastroenterology. 2007;132:52-65.
23. Hanauer SB, Sandborn WJ, Rutgeerts P, et al. Human anti-tumor necrosis factor monoclonal antibody (adalimumab) in Crohn's disease: the CLASSIC-I trial. Gastroenterology. 2006;130:323-333.
24. Sandborn WK, Hanauer SB, Rutgeerts PJ, et al. Adalimumab for maintenance treatment of Crohn's disease: results of the CLASSIC II trial. Gut. 2007;56:1232-1239.
25. Mannon P. GAIN for loss: adalimumab for infliximab-refractory Crohn disease. Ann Intern Med. 2007;146:888-890.
26. Sandborn WJ, Colombel JF, Enns R, et al. Natalizumab induction and maintenance therapy for Crohn's disease. New Engl J Med. 2005;353:1912-1925.
27. Targan SR, Feagan BG, Fedorak RN, et al. Natalizumab for the treatment of active Crohn's disease: results of the ENCORE Trial. Gastroenterology. 2007;132:1672-1683.
28. Sandborn WJ, Feagan BG, Stoinov S, et al. Certolizumab pegol for the treatment of Crohn's disease. New Engl J Med. 2007;357:228-238.
29. Schreiber S, Khaliq-Kareemi M, Lawrance IC, et al. Maintenance therapy with certolizumab pegol for Crohn's disease. New Engl J Med. 2007;357:239-250.
30. Sandborn WJ, Faubion WA. Biologics in inflammatory bowel disease: how much progress have we made? Gut. 2004;53:1366-1373.
31. Sutherland L, Singleton J, Sessions J, et al. Double blind, placebo controlled trial of metronidazole in Crohn's disease. Gut. 1991;32:1071-1075.
32. Schnitzler F, Fidder H, Ferrante M, et al. Long-term outcome of treatment with infliximab in 614 patients with Crohn's disease: results from a single-centre cohort. Gut. 2009;58:492-500.
33. Hommes D, Baert F, van Assche G, et al. Management of recent onset Crohn's disease: a controlled, randomized trial comparing step up to top down therapy [abstract]. Gastroenterology. 2005;129:371.
34. Baert F, Caprilli R, Angelucci E. Medical therapy for Crohn's disease: top-down or step-up? Dig Dis. 2007;25:260-266.
35. Domènech E, Mañosa M, Cabré E. Top-down therapy: is the evidence strong enough? Dig Dis. 2009;27:306-311.
36. Hanauer SB, Feagan BG, Lichtenstein GR, et al. Maintenance infliximab for Crohn's disease: the ACCENT I randomised trial. Lancet. 2002;359:1541-1549.
To comment on this article, contact rdavidson@uspharmacist.com.





