ABSTRACT: Inflammatory bowel disease (IBD) is an idiopathic inflammatory disorder with immunologic, genetic, and environmental influences of unknown etiology. IBD features two distinct diseases: Crohn’s disease (CD) and ulcerative colitis. The prevalence of CD has consistently increased over the past several decades. In March 2018, the American College of Gastroenterology (ACG) released an updated guideline on managing CD in adult patients. It includes preferable approaches on diagnosis, disease modifiers, and medical therapy for the various disease severities. ACG used the GRADE (Grading of Recommendations Assessment, Development and Evaluation) criteria to evaluate the level of evidence and rate recommendations.
Inflammatory bowel disease (IBD) is an idiopathic inflammatory disorder of unknown etiology with immunologic, genetic, and environmental influences. IBD encompasses two distinct diseases: Crohn’s disease (CD) and ulcerative colitis (UC).1,2 CD involves any area of the gastrointestinal tract (GIT) from the oral cavity to the anus, but it is limited primarily to the colon with or without small-intestine disease. Moreover, the inflammation in CD is often described as transmural, damaging each mucosal layer of the GIT, and noncontinuous.2 Therapy for CD includes medical therapy with pharmacologic agents consisting of 5-aminosalicylates (5-ASA), antibiotics, corticosteroids, immunomodulators, and biologics.3 Surgery is reserved for patients who are refractory to medical therapy.
Epidemiology and Etiology
The prevalence of CD is approximately 201 cases per 100,000 adults, whereas incidence is 3.1 to 14.6 cases per 100,000 person-years.1,4 CD is occurring more frequently in women and among smokers.1
The precise etiology of CD is unknown, but there are several known risk factors, including family history, smoking, use of oral contraceptives, diet, and ethnicity. A combination of these factors, along with aberrant mucosal immune responses, intestinal epithelial dysfunction, and defects of host interactions with intestinal microbes, likely contribute to CD.5,6
Environmental factors, especially cigarette smoking and diet, are also clearly involved in this disease. Tobacco smoking doubles the risk of both initial and recurrent CD. Decreased food contamination with improved food storage may also contribute to the increase in incidence. This so-called hygiene hypothesis suggests that the reduction in enteric infections in developed countries has resulted in inadequate development of the regulatory processes that limit mucosal immune responses. Pathogens that otherwise might cause only self-limited disease may instead trigger overwhelming inflammatory responses in susceptible individuals.7,8 Immunosuppression remains the mainstay of CD therapy, and it is likely that a combination of derangements that alter mucosal immunity and suppress immunoregulation is crucial in the development of this disease.
Clinical Features
The cardinal symptoms of CD include abdominal pain, diarrhea, and fatigue. Weight loss, fever, growth failure, anemia, recurrent fistulas, or extraintestinal manifestations (e.g., arthritis, iritis) can also occur.
The clinical presentation and symptoms of CD vary greatly depending on the segment of intestine involved. CD of the upper GIT usually presents with nausea, vomiting, dysphagia, or odynophagia. In CD of the small intestine, the principal symptom of small bowel CD is abdominal pain, which occurs in 90% of patients; other common symptoms are weight loss and anorexia. CD of the large intestine typically presents with diarrhea (may or may not be bloody) and abdominal pain and fever (often exacerbated by bowel movements). In large-intestine CD, stricturing of the colon can give rise to colonic obstruction, while fistulizing disease can give rise to abscess formation and fistulas. Finally, 60% and 80% of CD patients suffer from perianal fistulas (perianal CD).9,10
The Crohn’s Disease Activity Index (CDAI) is frequently used to assess disease severity.11 It gives a score ranging from 0 to over 600, based on a diary of symptoms kept by the patient for 7 days, general well-being, deviation of weight, features of extraintestinal disease, use of antidiarrheal medications, presence of abdominal mass, and hematocrit levels.
Diagnosis
The diagnosis of CD is based on a combination of clinical presentation and endoscopic, radiologic, histologic, and pathologic findings that demonstrate some degree of focal, asymmetric, and transmural granulomatous inflammation of the luminal GIT. Laboratory testing is complementary in assessing disease severity and complications of disease. There is no single laboratory test that can make an unequivocal diagnosis of CD. The sequence of testing is dependent on presenting clinical features.3
Management
Management of CD has advanced significantly since the advent of biomarkers and therapeutic drug monitoring (TDM) to optimize individual therapies. Treatment for active disease should be continued until symptomatic remission or failure to continue improvement. Clinical evidence of improvement should be evident within 2 to 4 weeks, and the maximal improvement should occur within 12 to 16 weeks.3
TDM has become common in the management of CD, especially among patients who initially responded to biologic therapy but developed loss of clinical response.12 Three different scenarios can explain drug failure: mechanistic failure, nonimmune-mediated drug failure, and immune-mediated drug failure.3
Mechanistic failure is considered when a patient with active mucosal ulceration has therapeutic drug levels and no antibodies. Nonimmune-mediated pharmacokinetic mechanisms occur when patients have subtherapeutic trough levels and absent antidrug antibodies. Immune-mediated drug failure is seen in patients who have low or undetectable trough levels and high titers of antidrug antibodies. Recent guidelines suggest minimal therapeutic target trough levels for use of infliximab at 7.5 µg/mL, adalimumab at 5 µg/mL, and certolizumab pegol at 20 µg/mL.13
The anatomic distribution of disease is important only for medications with targeted delivery systems, such as sulfasalazine, mesalamine, and enteric-coated budesonide. For all other agents, activity against CD is believed to occur throughout the entire GI tract.3
Goals of Therapy
The first goal of treatment for patients with CD is to induce a treatment response, measured as a decrease in CDAI by 100 points. A person is in symptomatic remission (usually corresponding to a CDAI score <150) when the patient is asymptomatic. The next goal is maintenance of remission, followed by reduction or elimination of steroid use. (Individuals who require the use of conventional corticosteroids to achieve clinical well-being are said to be steroid dependent and are not considered in remission.) The final goals are avoidance of hospitalization and surgery, followed by restoration and maintenance of nutrition.
Medical Management
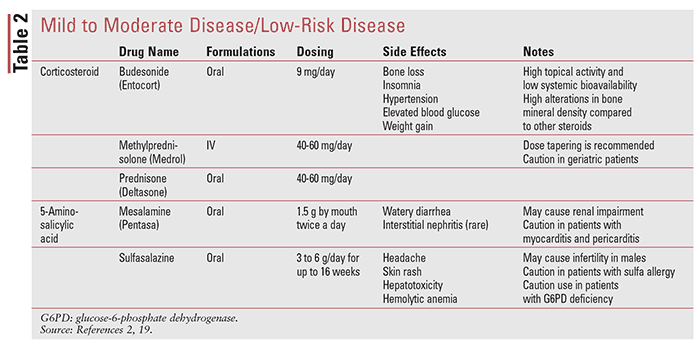
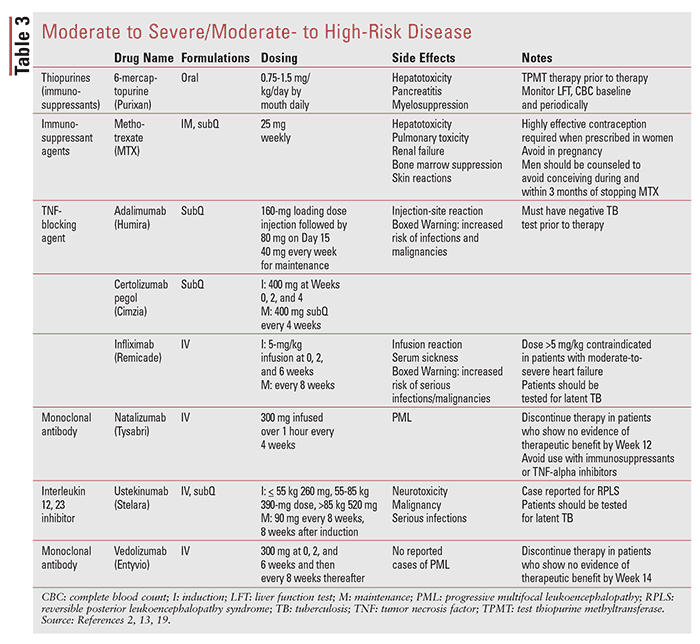
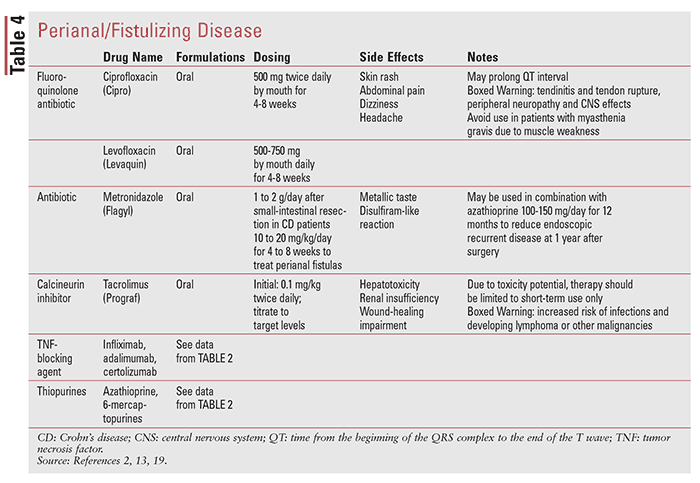
Medical treatment of CD is usually divided into induction and maintenance therapy. These phases of treatment involve achieving control of inflammation relatively quickly (3 months) and then sustaining that control for prolonged periods (>3 months). Treatment is generally chosen based on the patient’s risk profile and disease severity, with a goal of controlling inflammation and symptoms arising from active inflammation.3 TABLE 1 shows clinical presentation and corresponding CDAI score. TABLES 2 through 4 outline medications used in CD.

As a pharmacist, it is important, through clear communication, to determine whether a patient has symptoms from their CD or other conditions, such as bile salt diarrhea, intestinal infection such as Clostridium difficile, lactose intolerance, irritable bowel syndrome, food intolerance, accentuated gastrocolic reflex, a medication-related adverse event (such as diarrhea from an aminosalicylate [ASA] agent or antibiotics prescribed), or other conditions.
Therapeutic recommendations for patients with CD are established based upon disease location, disease severity, disease-associated complications, and future disease prognosis.
Mild to Moderately Severe/Low-Risk Disease
5-ASA Agents: Per guidelines, oral mesalamine has not consistently been demonstrated to be effective compared with placebo for induction of remission and achieving mucosal healing (MH) in patients with active CD, and it should not be used to treat patients with active CD.14 Sulfasalazine is formulated as combination of a sulfapyridine with 5-ASA; 5-ASA is responsible for the anti-inflammatory property of this drug, whereas sulfapyridine is the carrier that allows 5-ASA to be delivered into the colon.3 Oral sulfasalazine is an effective therapy for treatment of symptoms of patients with mild to moderately active colonic CD and/or ileo-colonic CD, but not isolated small bowel disease. There is lack of evidence on the ability of 5-ASAs to achieve MH in CD.15 For rectal and sigmoid disease, topical mesalamine is of limited benefit.3
Corticosteroids: Corticosteroids are a well-established treatment for the flares of CD. Oral formulations may be used for mild to moderate disease. Although not as effective as conventional oral corticosteroids such as prednisone, controlled ileal–release budesonide may be effective for short-term relief of symptoms of mild to moderate CD in patients whose disease is confined to the terminal ileum and right colon.3
Historically, corticosteroids have been used as a “bridge” to permit symptom control until immunomodulators and/or biologic agents become effective and enable mucosal healing.3 However, they should be used judiciously due to their adverse drug reactions, steroid dependency, and inability to achieve consistent mucosal healing. Corticosteroid doses are typically maintained for 1 to 2 weeks (prednisone: 40-60 mg/day) and tapered at 5 mg weekly until 20 mg, and then 2.5 to 5.0 mg weekly. Corticosteroid tapers should generally not exceed 3 months.
Antimicrobial Therapy: While antibiotics have a definite role in treating specific, episodic, or persisting bacterial infections that may complicate CD, their role in symptomatic management of CD is not fully established and has proved to be controversial. Antimicrobial therapy (metronidazole and ciprofloxacin) has not been shown to be effective for induction or for maintenance of remission or MH in patients with CD and should not be used as primary therapy.16 However, metronidazole and ciprofloxacin can be beneficial to improve symptoms related to simple superficial perianal fistulas in CD. Metronidazole is also helpful to prevent postoperative recurrence in CD. Its efficacy is increased over placebo when used with azathioprine to prevent postoperative recurrences.3
Moderate to Severe/Moderate- to High-Risk Disease
Corticosteroids: Oral corticosteroids are effective and can be used for short-term use in alleviating signs and symptoms of moderately to severely active CD, but they are ineffective for maintenance therapy in CD.3,17
Immounomodulators: Due to their slower onset of action, between 8 and 12 weeks, 6-mercaptopurine, azathioprine, and methotrexate (MTX) are not effective agents for short-term induction in active, symptomatic disease but are recommended for maintenance of remission.3 Thiopurines and MTX are considered steroid sparing and, therefore, can be used in patients with moderate to severe CD who remain symptomatic despite current or prior corticosteroid therapy. Thiopurine methyltransferase (TPMT) testing should be considered before initial use of azathioprine or 6-mercaptopurine to treat patients with CD.18
Thiopurines and MTX at reduced doses can also be used as adjunctive therapy for reducing immunogenicity against biologic therapy.
Anti–tumor Necrosis Factor Agents: The anti–tumor necrosis factor (anti-TNF) agents (infliximab, adalimumab, and certolizumab pegol) are effective for treatment of patients with CD who respond inadequately to treatment with corticosteroids, thiopurines, and MTX. Anti-TNF agents have rapid onset of effect, with benefit often noted within 2 weeks of initiating therapy. Treatment with anti-TNF agents appears to be more effective when given earlier during disease, and rates of response and remission are higher if given within 2 years of disease onset.3
Anti-TNF monotherapy is effective at maintaining anti-TNF–induced remission, but because of the potential for immunogenicity and loss of response, combination with azathioprine/6-mercaptopurine or methotrexate is recommended.
Inhibitors of Leukocyte Trafficking: Anti-integrin therapy with vedolizumab with or without an immunomodulator should be considered for induction of symptomatic remission in patients with CD. Natalizumab (anti-integrin antibody) can be used for induction and maintenance of remission in CD. Natalizumab should be used for maintenance of natalizumab-induced remission of CD only if serum antibody to John Cunningham virus, a risk factor for progressive multifocal leukoencephalopathy, is negative.3
Interleukin (IL 12/23) Inhibitors (Anti-p40 Antibody): Ustekinumab should be given for moderate to severe CD patients who have failed previous treatment with corticosteroids, immunomodulators, or anti-TNF inhibitors, or who have had no prior exposure to anti-TNF inhibitors.3
Severe/Fulminant Disease: Intravenous corticosteroids should be used to treat severe or fulminant CD.3 Anti-TNF agents can be considered in severe CD; however, only infliximab should be used in fulminant CD.3
Fistulizing CD: Infliximab should be considered in treating perianal, enterocutaneous, and rectovaginal fistulas in CD.3 Imidazole antibiotics in addition to infliximab is more effective than infliximab alone in perianal fistulas. Adalimumab, certolizumab pegol, and immunomodulators should be considered in treating perianal fistulas in CD. Tacrolimus can be administered for short-term treatment of perianal and cutaneous fistulas in CD.3 Drainage of abscesses is recommended prior to treatment of fistulizing CD with anti-TNF agents.3
The Pharmacist’s Role
Pharmacists can play many roles in improving outcomes for patients with CD. Among patients who are initial anti–TNF therapy responders, approximately one-third eventually lose response or become intolerant to therapy. Pharmacists should evaluate these patients to either escalate their current medication dose, switch to another anti-TNF agent, or switch to another medication class.
REFERENCES
1. Loftus EV Jr. Clinical epidemiology of inflammatory bowel disease: incidence, prevalence, and environmental influences. Gastroenterology. 2004;126:1504-1517.
2. Hanauer SB, Sandborn W. Practice Parameters Committee of the American College of Gastroenterology. Management of Crohn’s disease in adults. Am J Gastroenterol. 2001;96:635-643.
3. Lichtenstein GR, Loftus EV, Isaacs KL, et al. ACG Clinical Guideline: Management of Crohn’s Disease in Adults. Am J Gastroenterol. 2018; 113:481–517.
4. Kappelman MD, Rifas-Shiman SL, Kleinman K, et al. The prevalence and geographic distribution of Crohn’s disease and ulcerative colitis in the United States. Clin Gastroenterol Hepatol. 2007; 5:1424-1429.
5. Fiocchi C. Inflammatory bowel disease: etiology and pathogenesis. Gastroenterology. 1998 Jul. 115(1):182-205.
6. Kim DH, Cheon JH. Pathogenesis of inflammatory bowel disease and recent advances in biologic therapies. Immune Netw. 2017;17(1):25-40.
7. Farmer RG, Michener WM, Mortimer EA. Studies of family history among patients with inflammatory bowel disease. Clin Gastroenterol. 1980;9(2):271-277.
8. Weinstock JV, Elliott DE. Helminths and the IBD hygiene hypothesis. Inflamm Bowel Dis. 2009;15(1):128-133.
9. Day DW, Jass JR, Price AB, et al. Morson and Dawson’s Gastrointestinal Pathology. 4th ed. Malden, Massachusetts: Blackwell Science Ltd; 2003.
10. Korzenik JR, Dieckgraefe BK. Is Crohn’s disease an immunodeficiency? A hypothesis suggesting possible early events in the pathogenesis of Crohn’s disease. Dig Dis Sci. 2000;45(6):1121-1129.
11. Fielding JF. Perianal lesions in Crohn’s disease. J R Coll Surg Edinb. 1972;17(1):32-37.
12. Heron V, Afif W. Update on therapeutic drug monitoring in Crohn’s disease. Gastroenterol Clin North Am. 2017;46:645-659.13. Feuerstein JD, Nguyen GC, Kupfer SS, et al. American Gastroenterological Association Institute Guideline on therapeutic drug monitoring in inflammatory bowel disease. Gastroenterology. 2017;153:827-834.
14. Singleton JW, Hanauer SB, Gitnick GL, et al. Mesalamine capsules for the treatment of active Crohn’s disease: results of a 16-week trial. Pentasa Crohn’s Disease Study Group. Gastroenterology. 1993;104: 1293-1301.
15. Malchow H, Ewe K, Brandes JW, et al. European Cooperative Crohn’s Disease Study (ECCDS): results of drug treatment. Gastroenterology. 1984;86:249-266.
16. Borgaonkar MR, MacIntosh DG, Fardy JM. A meta-analysis of antimycobacterial therapy for Crohn’s disease. Am J Gastroenterol. 2000;95:725-729.
17. Benchimol EI, Seow CH, Steinhart AH, et al. Traditional corticosteroids for induction of remission in Crohn’s disease. Cochrane Database Syst Rev. 2008;(2):CD006792.
18. Winter JW, Gaffney D, Shapiro D, et al. Assessment of thiopurine methyltransferase enzyme activity is superior to genotype in predicting myelosuppression following azathioprine therapy in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2007;25:1069-1077.
19. Lexicomp. Accessed September 20, 2018.
To comment on this article, contact rdavidson@uspharmacist.com.





