US Pharm. 2015;40(6):39-44.
ABSTRACT: Glaucoma is a disorder of the eyes that can lead to blindness. Its incidence increases with age. While there is no cure, numerous topical medications are used to reduce the elevated intraocular pressure seen in patients. Topical prostaglandin analogues and beta-blockers are the most commonly used medications. Pharmacists can be effective partners in the treatment of glaucoma by monitoring adherence, counseling patients on the proper use of eyedrops, and recommending preservative-free or combination products when problems arise.
Glaucoma refers to a group of chronic disorders that can lead to optic nerve damage, retinal ganglion cell death, and visual-field defects. If left untreated or not treated adequately, it can lead to blindness. Glaucoma is the second leading cause of blindness in the world.1 There is no cure, and patients will need treatment for the rest of their lives to prevent visual-field deterioration.2 Of the different types, the two most prevalent are primary open-angle glaucoma (POAG) and angle-closure glaucoma (ACG).2 A variety of oral and topical treatments are available.
EPIDEMIOLOGY
POAG affects 2.7 million Americans aged >40 years and is estimated to affect 2% of people internationally.3,4 By 2020, 58.6 million people worldwide and 3 million Americans will have POAG.4,5 ACG accounts for 26% of the cases worldwide, but only 10% in the United States.4,6 The prevalence of glaucoma increases with age and is highest in African Americans in the U.S.3
In 2010, 61% of glaucoma cases in the U.S. were in women.3 This discrepancy is greater in the 40- to 60-year-old age group as compared to later in life.3 POAG has caused about 4.5 million cases of blindness worldwide.1
ETIOLOGY
Primary Open-Angle Glaucoma
Intraocular Pressure (IOP): Although IOP is no longer considered a diagnostic criterion, POAG is more likely to occur at a higher IOP.7 Multiple randomized, controlled trials have shown that a reduction in IOP slows the progression of visual-field defects and prevents the onset of POAG.8,9 IOP is now thought to be one of many factors that cause optic neuropathy leading to glaucoma. Many people with glaucoma have normal IOP and, conversely, some with elevated IOP show no signs of optic neuropathy. IOP undergoes diurnal variations, and elevation in IOP is suspected to be worse after falling asleep.10 Since pharmacologic treatment of POAG focuses on lowering IOP, an understanding of the process would be beneficial to pharmacists.
Aqueous humor (AH) is produced by the epithelium of the ciliary body and is used to supply nourishment to the cornea and lens. AH is secreted into the posterior chamber, flows into the anterior chamber, and then drains from there.11 A decrease in the outflow of AH from the anterior chamber increases IOP. There are two mechanisms by which AH is drained from the anterior chamber, the conventional and unconventional pathways. The conventional pathway involves the outflow of AH through Schlemm’s canal. The trabecular meshwork controls the flow of AH into Schlemm’s canal and ultimately the bloodstream. The unconventional pathway is a collection of pathways and involves the seepage of AH through optic tissues. The most common of these pathways is the uveoscleral route (FIGURE 1).12
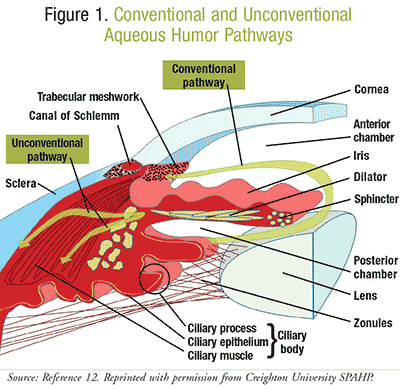
Optic Nerve Damage: Retinal ganglion cell axons converge in the optic nerve head and exit through the lamina cribrosa. The degeneration of retinal ganglion cells in the optic nerve head is the end result of multiple processes and ultimately leads to vision loss.13 Increased IOP can lead to stress on the retinal ganglion cell axons by reducing the flow of important neurotrophic factors for the axon’s function. This stress also leads to the release of degenerative substances such as tumor necrosis factor alpha (TNF-α). This causes damage to retinal ganglion cell axons.11 As retinal ganglion cells begin to die, the nerve fiber layer begins to thin and the cup at the top of the nerve head begins to increase in size.11 The lamina cribrosa begins to bow as well, and this increases the amount of cupping. The cup-to-disc ratio is used to assess optic neuropathy.14
Angle-Closure Glaucoma
ACG is characterized by blockage of AH outflow due to the closure of the angle between the iris and cornea. This closure results in the pinching-off of access to the trabecular meshwork.7 The decrease in outflow can cause a gradual increase in IOP or, more commonly, a rapid increase in IOP, leading to pain and permanent vision loss.
CLINICAL PRESENTATION
Glaucoma is described as optic nerve damage that leads to visual dysfunction.15 Generally, POAG is bilateral, asymmetric, and asymptomatic until significant peripheral vision loss occurs. ACG could include prodromal symptoms. It could also be an acute situation with distinct symptoms. Some of the features that are commonly seen in both types of glaucoma are shown in TABLE 1.13

DIAGNOSIS
A thorough patient history should be conducted at the start of every comprehensive adult eye evaluation. Visual acuity is measured at both near and far distances. Both pupils are assessed for restricted constriction of the affected pupil(s).16 An anterior segment examination is conducted to assess the integrity of the cornea and anterior and posterior chambers. Tonometry measures IOP to determine the level of pressure elevation, if present.16 The time of day and instrument used are recorded due to diurnal variations.10 A goniolens (gonioscope) measures the angle between the iris and cornea in order to differentiate between POAG and ACG.16 The optic nerve head and retinal nerve fiber layer are examined for characteristic changes with glaucoma. The cup-to-disc ratio is used to assess the amount of glaucomatous atrophy, and anything >0.5 is suggestive of atrophy. Perimetry measures the visual field and is used to judge the extent of peripheral vision loss. Although glaucoma is a bilateral process, the level of severity is not necessarily congruent between both eyes.16,17 Therefore, tests should be conducted on each eye individually.
TREATMENT
Nonpharmacologic Treatment
Laser Trabeculoplasty: This procedure is considered in patients who fail to be adherent to medication regimens, are unable to administer eyedrops, or cannot tolerate topical medications. Trabeculoplasty uses a very focused beam of light to cause increased drainage through the trabecular meshwork. The effects of this may wear off over time and, therefore, long-term benefit is uncertain.16
Trabeculectomy: This is the most common surgical procedure used to lower IOP. It involves creating a new pathway for the drainage of AH via bypassing the trabecular meshwork. Trabeculectomy is generally considered after topical agents and trabeculoplasty have been deemed insufficient at controlling IOP.17 It may also be considered as an initial therapy when IOP is extremely elevated.16,17
Cyclodestructive Procedures: These are used when medical and surgical treatments have failed and glaucoma is highly advanced. During cyclodestructive procedures, the ciliary body is intentionally damaged so that AH production is permanently reduced.18
Pharmacologic Treatment
Medications for the treatment of glaucoma are aimed at lowering IOP through two mechanisms, decreasing AH production and increasing AH outflow. It is recommended that IOP be lowered to a target level. That level is generally 20% below the baseline as measured several times.17 Prostaglandin analogues and beta-blockers are currently the most frequently used agents. Due to their once-daily dosing and effectiveness, prostaglandin analogues are generally selected as first-line options in treatment.16 Information regarding medications used to treat POAG can be found in TABLE 2.13,16
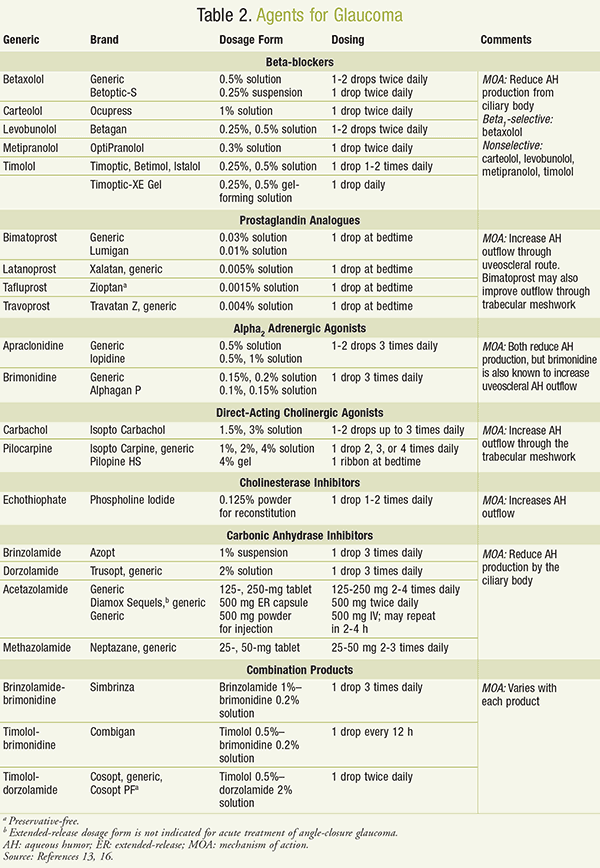
Beta-Blockers: Topical beta-blockers are one of the most commonly used classes of medications in the treatment of POAG.13,14,16 They produce an IOP-lowering effect by reducing the production of AH by the ciliary body. Local side effects that occur with the beta-blockers consist of stinging, burning, irritation, inflammation, and blurred vision. The local side effects are normally minor. Side effects are generally short-lived and will go away in time.
Systemic side effects with beta-blockers are rare, but it is important to be aware of them. These include bradycardia, hypotension, bronchospasm, serum lipid changes, and masking of hypoglycemia. The nonselective beta-blockers are contraindicated in patients with asthma or chronic obstructive pulmonary disease (COPD) due to their ability to induce bronchospasm. All topical beta-blockers are contraindicated in patients with sinus bradycardia, second- or third-degree heart block, congestive heart failure, atherosclerosis, and diabetes. Topical beta-blockers should also be avoided in patients taking oral beta-blockers.13,17
Prostaglandin Analogues: Topical prostaglandin analogues are usually the first choice by prescribers for POAG.17 In a large meta-analysis, it was concluded that prostaglandin analogues showed a greater 24-hour IOP reduction than timolol and other POAG medications.19 In a separate systematic review that included tafluprost, all of the prostaglandin analogues except tafluprost were shown to have significantly greater IOP reduction than timolol.20 It was also shown that bimatoprost was most effective at achieving a 30% reduction in IOP, which was the goal of the study.20 Prostaglandin analogues lower IOP by increasing the uveoscleral outflow of AH. It is suspected that bimatoprost also increases AH outflow through the trabecular meshwork. The prostaglandins are dosed once daily at night, where they have shown to be most effective.19
Systemic side effects are rare with these medications. Local adverse reactions are more common and include conjunctival hyperemia, lengthening and darkening of eyelashes, irreversible altered iris pigmentation, cornea inflammation, and macular edema.13,14,16
Adrenergic Agonists: Dipivefrin is a nonspecific adrenergic agonist. It is a prodrug of epinephrine and decreases IOP by increasing AH outflow through the uveoscleral route and trabecular meshwork. Dipivefrin is less effective at lowering IOP than other treatment options and is poorly tolerated. Therefore, it is rarely used.13,18 There are currently no commercial dipivefrin products available in the U.S.
Brimonidine and apraclonidine are alpha2 adrenergic agonists that lower IOP by decreasing AH production. Brimonidine also increases uveoscleral outflow. Brimonidine lowers IOP at a similar level to timolol and can be considered as an initial therapy.21 Brimonidine can be used as a monotherapy or as adjunctive to beta-blockers or prosta-glandin analogues, while apraclonidine is considered to be a second-line agent. Both are indicated for the treatment of glaucoma.
An ocular allergic-like reaction is the most common side effect with these agents. It occurs at a lower rate with brimonidine, but it is still significant and generally causes these medications to be discontinued.13 Other local events include burning, stinging, and irritation upon administration. Systemic side effects are rare, but dizziness, drowsiness, and dry mouth may occur. Caution should be used with coadministration of central nervous system (CNS) depressants, as these glaucoma agents could exacerbate CNS depression.13
Cholinergics: Cholinergic agents lower IOP by increasing AH outflow through the trabecular meshwork. These agents are rarely used because of multiple daily dosing and adverse effects.13 The local adverse events include retinal detachment, myopia, miosis, and pupillary block. Systemic side effects would be typical cholinergic effects such as sweating, salivation, nausea, vomiting, diarrhea, and bradycardia.13,16
Carbonic Anhydrase Inhibitors: These agents lower IOP by blocking the secretion of sodium and bicarbonate ions into the AH, thereby inhibiting the production of AH.13 Topical formulations (brinzolamide and dorzolamide) are considered for monotherapy or adjunctive therapy in those who do not achieve effective control with other medications. These are well tolerated, with adverse events including transient burning and stinging, blurred vision, tearing, and corneal edema.13,16 Dorzolamide is reported to produce more stinging than brinzolamide, while brinzolamide can cause more blurred vision than dorzolamide.13 Systemic effects are rare and are generally caused by oral formulations, although topical formulations can cause altered taste.
Oral formulations (acetazolamide and methazolamide) are used for those who do not effectively respond to maximum topical therapy. Systemic side effects are common and include malaise, depression, metallic taste, anorexia, diarrhea, kidney stones, metabolic acidosis, and decreased libido.13,16
Cholinesterase Inhibitor: The cholinesterase inhibitor echothiophate widens the trabecular meshwork by inhibiting the destruction of acetylcholine and lowers IOP by increasing AH outflow. Side effects are a major reason for limited use. These include fibrinous iritis, iris cysts, conjunctival thickening, and nasolacrimal occlusion.13 In addition, this drug has limited commercial availability and should only be used in nonresponsive patients.
Combination Products: Products that combine timolol with brimonidine or dorzolamide are available. A product that combines brimonidine and brinzolamide is also available. Combination therapy should be considered when initial therapy produces only a partial response.13,16 Advantages of using one product instead of two include less total preservative exposure, no washout effect, a single copay, and a possible improvement in adherence to treatment.
Preservative-Free Products: Recent research has shown that preservatives like benzalkonium chloride (BAK) in eyedrops could be causing a significant increase in local adverse events, such as dry eyes.20,22 These studies also showed that preservative-free eyedrops lead to a significant decrease in local adverse events. Studies have also found equal therapeutic efficacy between preservative-free and preserved products.23,24 Products with multiple daily dosing are more likely to cause side effects because of an increase in the amount of BAK.24 These studies looked at products available primarily in Canada and Europe. The only currently available preservative-free product in the U.S. is Cosopt PF, a combination of timolol and dorzolamide.
PHARMACIST INTERVENTION
It is vital that pharmacists understand the pathophysiology of glaucoma and the goals of therapy. Pharmacists should counsel patients that the goal of treatment is to prevent progression of glaucoma by lowering IOP. Pharmacists also need to remind patients that many of the adverse effects of the medications are brief and will go away in time. This may help to improve adherence to the regimen.
Absorption of certain medications, especially beta-blockers and drugs that can produce anticholinergic effects, may produce serious systemic side effects. Therefore, pharmacists should counsel patients on the proper instillation of eyedrops, including the use of punctal occlusion (pressing on the bridge of the nose to prevent the drops from entering the nasolacrimal duct). Simply closing the eye (but not blinking) is an equally effective alternative. Either of these procedures should be done for about 2 minutes.14
Adherence is a problem with glaucoma patients. In one study, 45% of patients were shown to take <75% of their doses.25 Pharmacists can play an important role in helping to improve adherence by thoroughly counseling patients about the disease and the medications used to treat it. In the case of poor adherence to therapy, pharmacists can recommend that the prescriber consider a combination product. Pharmacists should also consider recommending a preservative-free product for patients experiencing ocular side effects as previously discussed. A combination product may also be a good recommendation if a patient is experiencing side effects due to preservatives and a preservative-free product is not commercially available.
CONCLUSION
Glaucoma is an irreversible optic neuropathy that leads to visual-field defects and potentially blindness. IOP is a major factor in the development of optic nerve damage and is the target of medical treatments for POAG. Prostaglandin analogues are the most effective at lowering IOP and are considered first-line therapy. Other options for initial monotherapy include timolol and brimonidine. Pharmacists must be aware of potential local and systemic effects to help promote adherence and enhance treatment of glaucoma.
ACKNOWLEDGEMENT: The authors would like to thank Drs. Catherine Opere and Karen O’Brien for their assistance and suggestions in the preparation of this manuscript.
REFERENCES
1. World Health Organization. Prevention of blindness and visual impairment. www.who.int/blindness/causes/priority/en/index6.html. Accessed October 1, 2014.
2. American Optometric Association. Glaucoma. www.aoa.org/patients-and-public/eye-and-vision-problems/glossary-of-eye-and-vision-conditions/glaucoma?sso=y. Accessed October 1, 2014.
3. National Eye Institute. Glaucoma, open-angle. www.nei.nih.gov/eyedata/glaucoma.asp. Accessed October 1, 2014.
4. Quigley HA, Broman AT. The number of people with glaucoma worldwide in 2010 and 2020. Br J Ophthalmol. 2006;90:262-267.
5. Friedman DS, Wolfs RC, O’Colmain BJ, et al. Prevalence of open-angle glaucoma among adults in the United States. Arch Ophthalmol. 2004;122: 532-538.
6. American Academy of Ophthalmology Glaucoma Panel. Preferred Practice Pattern Guideline: Primary Angle Closure. San Francisco, CA: American Academy of Ophthalmology; 2010. www.aao.org/preferred-practice-pattern/primary-angle-closure-ppp--october-2010. Accessed October 15, 2014.
7. Quigley HA. Glaucoma. Lancet. 2011;377:1367-1377.
8. Kass MA, Heuer DK, Higginbotham EJ, et al. The ocular hypertension treatment study: a randomized trial determines that topical ocular hypotensive medication delays or prevents the onset of primary open-angle glaucoma. Arch Ophthalmol. 2002;120(6):701-713.
9. The AGIS Investigators. The Advanced Glaucoma Intervention Study (AGIS), 7: the relationship between control of intraocular pressure and visual field deterioration. Am J Ophthalmol. 2000;130(4):429-440.
10. Wax MB, Camras CB, Fiscella RG, et al. Emerging perspectives in glaucoma: optimizing 24-hour control of intraocular pressure. Am J Ophthalmol. 2002;133(suppl):S1-S10.
11. Kwon YH, Fingert JH, Kuehn MH, Alward WLM. Primary open-angle glaucoma. N Engl J Med. 2009;360:1113-1124.
12. O’Brien K, Chock AW, Opere CA. An overview of glaucoma management for pharmacists. US Pharm. 2010;35(4):52-61. www.uspharmacist.com/continuing_education/ceviewtest/lessonid/106698. Accessed May 12, 2015.
13. Fiscella RG, Lesar TS, Edward DP. Chapter 75. Glaucoma. In: DiPiro JT, Talbert RL, Yee GC, et al, eds. Pharmacotherapy: A Pathophysiologic Approach. 9th ed. New York, NY: McGraw-Hill; 2014.
14. Weinreb RN, Aung T, Medeiros FA. The pathophysiology and treatment of glaucoma: a review. JAMA. 2014;311(18):1901-1911.
15. Foster PJ, Buhrmann R, Quigley HA, Johnson GJ. The definition and classification of glaucoma in prevalence surveys. Br J Ophthalmol. 2002;86(2): 238-242.
16. American Optometric Association. Care of the Patient with Open-Angle Glaucoma. Saint Louis, MO: American Optometric Association; 2011.
17. American Academy of Ophthalmology Glaucoma Panel. Preferred Practice Pattern Guideline: Primary Open-Angle Glaucoma Suspect. San Francisco, CA: American Academy of Ophthalmology; 2010. www.aao.org/preferred-practice-pattern/primary-openangle-glaucoma-suspect-ppp--october-20. Accessed October 1, 2014.
18. Salmon JF. Chapter 11. Glaucoma. In: Riordan-Eva P, Cunningham ET Jr, eds. Vaughan & Asbury’s General Ophthalmology. 18th ed. New York, NY: McGraw-Hill; 2011.
19. Stewart W, Konstas A, Nelson L, Kruft B. Meta-analysis of 24-hour intraocular pressure studies evaluating the efficacy of glaucoma medicines. Ophthalmology. 2008;115:1117-1122.
20. Lin L, Zhao YJ, Chew PT, et al. Comparative efficacy and tolerability of topical prostaglandin analogues for primary open-angle glaucoma and ocular hypertension. Ann Pharmacother. 2014;48(12):1585-1593.
21. Krupin T, Liebmann JM, Greenfield DS, et al. A randomized trial of brimonidine versus timolol in preserving visual function: results from the Low-pressure Glaucoma Treatment Study. Am J Ophthalmol 2011;151: 671-681.
22. Pisella PJ, Pouliquen P, Baudouin C. Prevalence of ocular symptoms and signs with preserved and preservative-free glaucoma medications. Br J Ophthalmol. 2002;86:418-423.
23. Rouland JF, Traverso CE, Stalmans I, et al. Efficacy and safety of preservative-free latanoprost eyedrops, compared with BAK-preserved latanoprost in patients with ocular hypertension or glaucoma. Br J Ophthalmol. 2013;97(2):196-200.
24. Day DG, Walters TR, Schwartz GF, et al. Bimatoprost 0.03% preservative-free ophthalmic solution versus bimatoprost 0.03% ophthalmic solution (Lumigan) for glaucoma or ocular hypertension: a 12-week, randomised, double-masked trial. Br J Ophthalmol. 2013;97(8):989-993.
25. Okeke CO, Quigley HA, Jampel HD, et al. Adherence with topical glaucoma medication monitored electronically: the Travatan Dosing Aid Study. Ophthalmology. 2009;116:191-199.
To comment on this article, contact rdavidson@uspharmacist.com.






