US Pharm. 2021;46(12):HS2-HS9.
ABSTRACT: Gastrointestinal (GI) diseases affect the alimentary tract, liver, biliary system, and pancreas. GI diseases are often encountered in the ICU setting, either as the major cause that prompted admission to the ICU or as a comorbid complication of another primary disease process. GI complications of diabetes have become increasingly prevalent as the rate of diabetes has risen. The most common and immediately life-threatening diabetes-related GI complications seen in emergency departments are gallstones and acute pancreatitis. Pharmacists in acute-care settings need to be aware of the prevalence, etiology, diagnosis, pathogenesis, and management of these two common complications of diabetes.
Gastrointestinal (GI) diseases affect the alimentary tract, liver, biliary system, and pancreas, and they are often encountered in the ICU setting, either as the major cause that prompted admission to the ICU or as a comorbid complication of another primary disease process.1 Diabetes is a common condition, affecting 34.5 million people of all ages, or 10.5% of the U.S. population. The most common and immediately life-threatening diabetes-related GI complications seen in emergency departments are gallstones and acute pancreatitis.
In 2015, annual healthcare expenditures for GI diseases in the U.S. totaled $135.9 billion. Yearly, there were more than 54.4 million ambulatory visits with a primary diagnosis for a GI disease, 3.0 million hospital admissions, and 540,500 all-cause 30-day readmissions.1 GI complications of diabetes have become increasingly prevalent as the rate of diabetes has risen. Up to 75% of patients with diabetes may experience GI symptoms, leading to a significant decrement in quality of life and an increase in healthcare costs. In fact, at some point in their lives, diabetic patients will develop a GI problem, such as ulcers, gallstones, irritable bowel syndrome, or another GI disorder. Both type 1 and type 2 diabetes can affect a patient’s entire GI tract, from the esophagus to the anus. No data are available as to how many of the 3 million patients who are hospitalized annually for GI disease have diabetes.2,3
GALLSTONES
Gallstones, formally known as cholelithiasis, are one of the most common and costly GI diseases. These hard, pebble-like deposits that form in the gallbladder are typically composed of cholesterol or bilirubin. Gallstones can be as small as a grain of salt or as large as a golf ball. The gallbladder can make one large gallstone, hundreds of tiny stones, or both small and large stones. Gallstones can also travel from the gallbladder to the common bile duct, which is the largest of the ducts in the liver. Blockage of bile ducts by gallstones may cause sudden, severe pain in upper right portion of the abdomen. Common bile duct stones are much less common than gallstones.4
Prevalence and Etiology
Gallstones are a significant health problem in developed societies, affecting 10% to 15% of the adult population; in the U.S., 20 million to 25 million people have or will develop gallstones. The resultant direct and indirect costs of gallbladder disease in the U.S. are calculated at roughly $6.2 billion annually, constituting a major health burden that has increased more than 20% over the past 3 decades. With an estimated 1.8 million ambulatory-care visits for this condition each year, gallstone disease is a leading cause of hospital admissions related to GI problems.5
Gallstones are encountered more frequently in patients with diabetes, which is not surprising given that risk factors for the development of stones, such as intestinal dysmotility, obesity, and hypertriglyceridemia, are more common in this population (particularly patients with type 2 diabetes). In addition, impairment of gallbladder motility and autonomic neuropathy, as well as elements such as cholesterol supersaturation and crystal nucleation–promoting factors, are considered important in the development of gallstones.6
In the general U.S. population, both men and women with a diabetes diagnosis were more than 50% more likely to have gallstone disease compared with persons without a diabetes diagnosis after adjustment for multiple shared risk factors. Men and women with undiagnosed diabetes (fasting plasma glucose 126 mg/dL or greater) were approximately twice as likely to have gallstone disease as those with normal fasting glucose (<110 mg/dL [<6.11 mmol/L]). This relationship reached statistical significance only among women. Impaired fasting glucose (110-125 mg/dL) was unrelated to gallstone disease in women or men.6
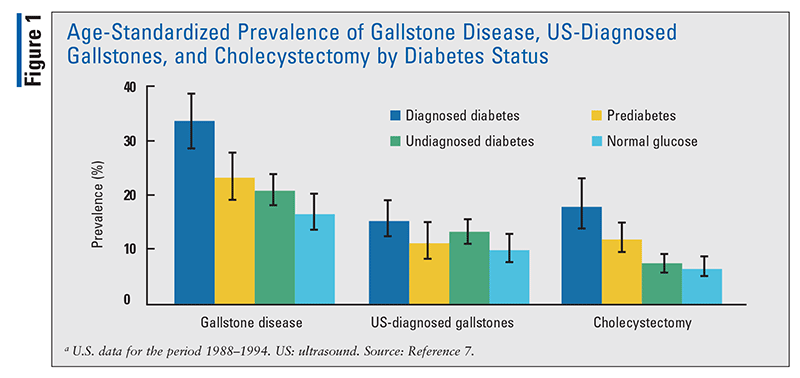
To further assess the relationship between diabetes and gallstone disease in the general U.S. population, new analyses for the publication Diabetes in America were conducted using gallbladder ultrasonography data on adults aged 20 to 74 years from the Third National Health and Nutrition Examination Study (NHANES III). In these analyses, gallstone-disease prevalence and odds ratios may not be identical to those in previously published NHANES III reports because of differences in definitions of diabetes and adjustment only for age (prevalence) or for age, ethnicity/race, or sex. NHANES III defines diabetes as diagnosed (i.e., self-reported healthcare provider diagnosis) and—in patients without a diagnosis—as undiagnosed (A1C 6.5% or greater, or fasting glucose 126 mg/dL or greater), prediabetes (A1C 5.7%-6.4% or fasting glucose 100-125 mg/dL), or normal glucose (A1C <5.7% and fasting glucose <100 mg/dL). The prevalence (± standard error) of gallstone disease was 33.3% ± 2.6% among patients with diagnosed diabetes, 23.3% ± 2.2% among those with undiagnosed diabetes, 20.8% ± 1.5% among those with prediabetes, and 16.7% ± 1.7% among those with normal glucose (FIGURE 1).7
Pathogenesis
Five primary defects play a critical role in the pathogenesis of cholesterol gallstones: Lith genes and genetic factors; hepatic hypersecretion of cholesterol, resulting in supersaturated gallbladder bile; rapid phase transitions of cholesterol in bile, with the precipitation of solid cholesterol crystals; impaired gallbladder motility with hypersecretion and accumulation of mucin gel in the gallbladder lumen and immune-mediated gallbladder inflammation; and intestinal factors involving absorption of cholesterol, slow intestinal motility, and altered gut microbiota. The principal gallbladder pathologic feature in diabetic patients is a functional deficit of uncertain etiologic factors, leading to a large, flaccid, poorly emptying organ. Bile acid and lipid composition are usually increased in diabetic patients.8,9
Diagnosis
The occurrence of right-upper-quadrant abdominal pain, nausea, or vomiting suggests a diagnosis of gallstones. The location, duration, and character of the pain (i.e., gnawing, cramping, or stabbing) help the clinician determine the likelihood of gallstones. The patient may exhibit abdominal tenderness and abnormally high liver-function tests. Abdominal ultrasound (US) is a sensitive, quick, and fairly inexpensive method of detecting gallstones in the gallbladder or common bile duct, and it is the test most often employed.10
CT should be considered in patients with negative or ambiguous US results or if complications of gallstones are suspected. Magnetic resonance cholangiopancreatography is a noninvasive screening method with high sensitivity and specificity for detecting gallstones. According to the 2010 American College of Radiology guidelines, MRI is recommended as a secondary imaging study if US does not result in a clear diagnosis of acute cholecystitis or gallstones.11
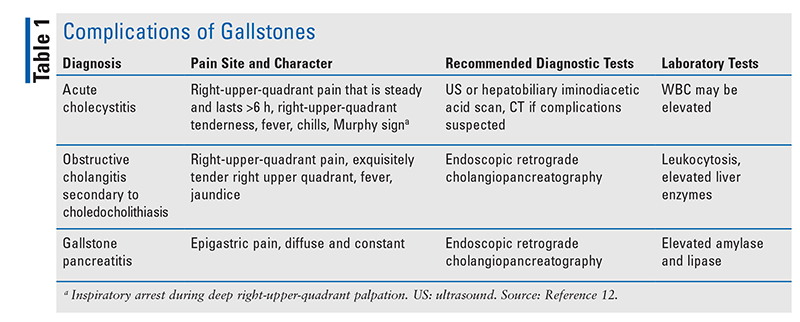
See TABLE 1 for more information on complications of gallstones.
Management
Surgery: Cholecystectomy (removal of the gallbladder), whether open or minimally invasive, remains the primary means of managing symptomatic gallstone disease; it is safe, has the lowest risk of recurrence, and provides 92% of patients with complete relief from biliary pain. Laparoscopic cholecystectomy remains the surgical choice for symptomatic and complicated gallstones, replacing open cholecystectomy as the standard of care. Laparoscopic cholecystectomy is associated with a shorter hospital stay and a shorter convalescence period than open cholecystectomy. Laparoscopic cholecystectomy continues to have numerous advantages over the open technique, and the laparoscopic approach to treating gallstone disease in various patient populations is gaining clinical acceptance based on its safety. Percutaneous cholecystostomy is an alternative for patients who are critically ill with gallbladder empyema and sepsis. Diabetic patients with symptomatic gallbladder disease usually require surgery. The cholecystectomy risk in patients with diabetes is similar to that in patients without diabetes. Formerly, prophylactic cholecystectomy was recommended for diabetic patients with silent gallstones based on an apparent high risk of cholecystitis.11
Nonsurgical Treatments: For asymptomatic pigmented or calcified gallstones, no medical therapy other than pain prophylaxis is recommended. For cholesterol-containing gallstones, litholysis with oral agents is a historical option not often used in current clinical practice. Symptomatic patients who are not candidates for surgery or those who have small gallstones (5 mm or smaller) in a functioning gallbladder with a patent cystic duct are candidates for dissolution therapy. Options include oral ursodeoxycholic acid (ursodiol [Actigall]) and chenodeoxycholic acid (chenodiol [Chenodal]). Both agents reduce hepatic secretion of biliary cholesterol, cause formation of unsaturated bile, and promote dissolution of cholesterol crystals and gallstones. After 6 to 12 months, dissolution therapy may eventually result in dissolution of small gallstones, but the recurrence rate exceeds 50%. Oral dissolution has several disadvantages, including the lengthy time frame of observation (up to 2 years). Gallstone recurrence is another disadvantage of this treatment, as approximately 25% of patients develop recurrent gallstones within 5 years. Fewer than 10% of patients with symptomatic gallstones are candidates for this therapy. Presently, bile acid therapy is indicated only for patients unfit for or unwilling to undergo surgery. When surgery is to be avoided, extracorporeal shock wave lithotripsy is a noninvasive therapeutic alternative for symptomatic patients. Although serious adverse effects (e.g., biliary pancreatitis, liver hematoma) are rare, stone recurrence is a limitation of the procedure. Complications of gallstones include acute cholecystitis, obstructive cholangitis secondary to choledocholithiasis, and gallstone pancreatitis.11,12
ACUTE PANCREATITIS
Chronic pancreatitis is a continuing inflammatory disease of the pancreas that is characterized by irreversible morphological changes. These changes typically cause pain and loss of exocrine and endocrine pancreatic function. Acute pancreatitis is a condition that typically presents with abdominal pain and is usually associated with raised pancreatic enzymes in blood or urine due to inflammatory disease of the pancreas. Acute pancreatitis is an unpredictable and potentially lethal disease, and its prognosis depends mainly on the development of organ failure and secondary infection of pancreatic or peripancreatic necrosis. Despite improvements in treatment and critical care, severe acute pancreatitis is still associated with high mortality rates.13,14
Prevalence and Etiology
Pancreatitis is common in the U.S., with a yearly incidence of 40 per 100,000 persons; it leads to more than 300,000 inpatient admissions and 20,000 deaths annually, with costs exceeding $2.2 billion per year. From 35% to 55% of pancreatitis cases are related to gallstones.15 Acute pancreatitis is the more common form of pancreatitis, accounting for about 275,000 hospital stays each year in the U.S.1 The reason for the increase in acute pancreatitis is unknown. However, a concurrent trend has been the rapid worldwide increase in type 2 diabetes and obesity. Several clinical factors associated with type 2 diabetes and obesity (in particular, abdominal obesity) are known risk factors for acute pancreatitis (e.g., gallstone disease). Therefore, it can be hypothesized that in type 2 diabetic patients the risk of acute pancreatitis may be higher than that occurring in the general population.
A case-control study concluded that sulfonylurea glyburide increased the risk of acute pancreatitis, but neither insulin nor metformin seemed to lower the risk. In fact, there are reports of cases of acute pancreatitis in patients using metformin after an episode of acute renal failure. Although acute pancreatitis has numerous causes, the two most common factors are alcohol abuse and biliary tract obstruction related to cholelithiasis. These two conditions account for 60% to 80% of all cases of acute pancreatitis. The incidence of gallstone pancreatitis is higher in white women older than 60 years, and it is highest in patients with small gallstones (<5 mm in diameter) or microlithiasis.16
In general, patients with gallstone pancreatitis tend to have a higher mortality rate than patients with alcoholic pancreatitis. In addition, the presence of type 2 diabetes significantly increases the risk of complications and death. In patients with multiorgan involvement, the mortality rate is as high as 20%. Most deaths are due to multiorgan failure and hypotensive shock.17
Pathogenesis
The pathogenesis of acute pancreatitis relates to inappropriate activation of trypsinogen to trypsin (the key enzyme in the activation of pancreatic zymogens) and a lack of prompt elimination of active trypsin inside the pancreas. Activation of digestive enzymes causes pancreatic injury and results in an inflammatory response that is out of proportion to the response of other organs to a similar insult. The acute inflammatory response itself causes substantial tissue damage and may progress beyond the pancreas to a systemic inflammatory response syndrome, multiorgan failure, or death.18
Diagnosis
The diagnosis of acute pancreatitis is established by the presence of at least two of the following: stereotypical abdominal pain, serum amylase and/or lipase more than three times the upper limit of normal (ULN), and/or characteristic findings on abdominal imaging. Patients with acute pancreatitis typically present with midepigastric or right-upper-quadrant pain that is constant, is stabbing in character, and radiates to the back or flank.19
Serum pancreatic enzymes (amylase and lipase) remain the gold standard for diagnosing acute pancreatitis. If a diagnosis of acute pancreatitis is established by the presence of abdominal pain and increases in serum pancreatic enzyme activity, CT is not usually required for diagnosis in the emergency department or upon hospital admission. The onset of pancreatitis is considered to coincide with the first day of pain, not the day on which the patient presents for care or the day of hospital admission.20
Serum amylase and lipase are the two blood tests most commonly used to confirm a diagnosis of acute pancreatitis. Serum amylase increases in 6 to 12 hours and remains elevated for 3 to 5 days in uncomplicated acute pancreatitis. Serum lipase is elevated on the initial day and remains elevated slightly longer than amylase. If the cutoff of three times the ULN is used, the specificity of both enzymes is high. It is recommended to measure blood lipase because it is reported to be superior to all other pancreatic enzymes in terms of sensitivity and specificity.20
Contrast-enhanced CT and MRI of the pancreas should be reserved for patients in whom diagnosis is uncertain from clinical and laboratory assessment alone or for patients who fail to improve clinically within the first 72 hours of hospitalization. US may be useful for evaluating gallstones or biliary ductal dilation. MRI with gadolinium provides the same information about the pancreas as CT scanning; however, it provides better information regarding the biliary tree.20
Management
Fluid Therapy: The American College of Gastroenterology clinical guideline for initial management of acute pancreatitis states that aggressive hydration (defined as 250-500 mL/h of isotonic crystalloid solution) should be provided to all patients, unless cardiovascular or renal comorbidities exist. Early aggressive IV hydration is most beneficial in the first 12 to 24 hours but may have little benefit beyond that (strong recommendation, moderate quality of evidence). In a patient with severe volume depletion manifesting as hypotension and tachycardia, more rapid repletion (i.e., bolus) may be needed (conditional recommendation, moderate quality of evidence). Fluid requirements should be reassessed at frequent intervals within 6 hours of admission and for the next 24 to 48 hours. The goal of aggressive hydration should be to reduce blood urea nitrogen (strong recommendation, moderate quality of evidence).21 Surgical intervention is indicated in patients with infected necrosis.
Symptom Control: The role of antibiotics in the management of early acute pancreatitis is controversial. Although some studies have shown reduced mortality with the use of antibiotics, the severity of complications makes it difficult to perform unblinded studies. Control patients who decompensate receive more surgical procedures, which may contribute to infection and bias the studies. However, the use of IV or gut-sterilizing oral antibiotics is recommended in patients with acute necrotizing pancreatitis. Antibiotics that show promising results include imipenem-cilastatin (Primaxin), cefuroxime (Zinacef), and ceftazidime (Ceptaz).22
CONCLUSION
GI disorders in patients with diabetes are quite common but are not typically diagnosed in the community medical practice. In the U.S., gallstones and acute pancreatitis are two leading causes of GI-related hospitalization in patients with diabetes, and their frequency continues to rise worldwide. Pharmacists in acute-care settings need to be aware of the prevalence, etiology, diagnosis, pathogenesis, and management of these two common complications of diabetes.
REFERENCES
1. Peery AF, Crockett SD, Murphy CC, et al. Burden and cost of gastrointestinal, liver, and pancreatic diseases in the United States: update 2018. Gastroenterology. 2019;156(1):254-272.e11.
2. Maisey A. A practical approach to gastrointestinal complications of diabetes. Diabetes Ther. 2016;7(3):379-386.
3. Montgomery B. Diabetes and gastrointestinal issues. www.thediabetescouncil.com/diabetes-and-gastrointestinal-issues/. Accessed November 19, 2021.
4. National Institute of Diabetes and Digestive and Kidney Diseases. Definition & facts for gallstones. www.niddk.nih.gov/health-information/digestive-diseases/gallstones/definition-facts. Accessed November 19, 2021.
5. Stinton LM, Shaffer EA. Epidemiology of gallbladder disease: cholelithiasis and cancer. Gut Liver. 2012;6(2):172-187.
6. Marathe CS, Rayner CK, Wu T, et al. Gastrointestinal disorders in diabetes. In: Finegold KR, Anawalt B, Boyce A, et al, eds. Endotext [Internet]. South Dartmouth, MA: MDText.com, Inc; 2000.
7. Ruhl CE, Clark JM, Everhart JE. Liver and gallbladder disease in diabetes. In: Cowie CC, Casagrande SS, Menke A, et al, eds. Diabetes in America. 3rd ed. Bethesda, MD: National Institute of Diabetes and Digestive and Kidney Diseases; 2018.
8. Di Ciaula A, Wang DQ, Portincasa P. An update on the pathogenesis of cholesterol gallstone disease. Curr Opin Gastroenterol. 2018;34(2):71-80.
9. Ikard RW. Gallstones, cholecystitis and diabetes. Surg Gynecol Obstet. 1990;171(6):528-532.
10. Choi Y, Silverman WB. Gallstones and gallstone disease. American College of Gastroenterology. https://gi.org/topics/biliary-tract-disorders-gallbladder-disorders-and-gallstone-pancreatitis/#tabs2. Accessed November 19, 2021.
11. Abraham S, Rivero HG, Erlikh IV, et al. Surgical and nonsurgical management of gallstones. Am Fam Physician. 2014;89(10):795-802.
12. Bellows CF, Berger DH, Crass RA. Management of gallstones. Am Fam Physician. 2005;72(4):637-642.
13. Goulden MR. The pain of chronic pancreatitis: a persistent clinical challenge. Br J Pain. 2013;7(1):8-22.
14. Boxhoorn L, Voermans RP, Bouwense SA, et al. Acute pancreatitis. Lancet. 2020;396(10252):726-734.
15. Mueck KM, Wei S, Pedroza C, et al. Gallstone pancreatitis: admission versus normal cholecystectomy—a randomized trial (Gallstone PANC Trial). Ann Surg. 2019;270(3):519-527.
16. Gonzalez-Perez A, Schlienger RG, García Rodríguez LA. Acute pancreatitis in association with type 2 diabetes and antidiabetic drugs: a population-based cohort study. Diabetes Care. 2010;33(12):2580-2585.
17. Gapp J, Chandra S. Acute pancreatitis. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; 2021.
18. Whitcomb DC. Acute pancreatitis. N Engl J Med. 2006;354(20):2142-2150.
19. James TW, Crockett SD. Management of acute pancreatitis in the first 72 hours. Curr Opin Gastroenterol. 2018;34(5):330-335.
20. Brizi MG, Perillo F, Cannone F, et al. The role of imaging in acute pancreatitis. Radiol Med. 2021;126(8):1017-1029.
21. Tenner S, Baillie J, DeWitt J, et al. American College of Gastroenterology guideline: management of acute pancreatitis. Am J Gastroenterol. 2013;108(9):1400-1415.
22. Munoz A, Katerndahl DA. Diagnosis and management of acute pancreatitis. Am Fam Physician. 2000;62(1):164-174.
The content contained in this article is for informational purposes only. The content is not intended to be a substitute for professional advice. Reliance on any information provided in this article is solely at your own risk.
To comment on this article, contact rdavidson@uspharmacist.com.





