US Pharm. 2012;37(8):HS2-HS6.
Parasitic infections, starvation, insufficient shelter, and lack of clean water sources are the greatest barriers to health in our world’s growing population. Several of the parasitic infections that are very common throughout the world (e.g., ascariasis) frequently occur with mild, obscure symptoms or none at all. It is common for a host (especially of an intestinal parasite) to be asymptomatic, which is technically more of a mutualistic coexistence than true parasitism. The parasitic infections thought to be the most prevalent worldwide include toxoplasmosis, ascariasis, hookworm disease, and trichomoniasis (sorted by worldwide prevalence in TABLE 1).1
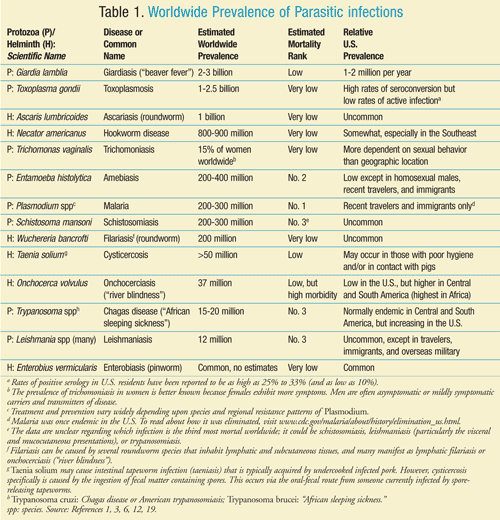
Some infections with low mortality rates may cause extensive morbidity, including fetal/neonatal damage, nutritional deficiencies, cutaneous nodules, skin eruptions or necrosis, and major end-organ damage of the eyes, central nervous system (CNS), lungs, heart, or liver.2-4 Although they are not the most prevalent infections, malaria, amebiasis, leishmaniasis, schistosomiasis, and trypanosomiasis are the leading causes of mortality worldwide due to parasitic infections.5 The scope of this topic could be immense, as there are hundreds of parasitic species that infect humans. This article will address drug therapy (and sometimes prevention) for the parasitic infections most relevant to pharmacy practice in the United States.
Parasitic Infections in the U.S.
It is postulated that U.S. residents suffer less from parasitic infections because of better provisions of food, shelter, matters of hygiene, and clean water. The U.S. is also somewhat geographically isolated from the rest of the world, and its climate, particularly in the northern part of the country, is also likely protective. When U.S. residents are affected, it is usually due to travel to tropical areas, recent immigration to the U.S., or direct contact with new immigrants. Occasionally, infections are caused by indigenous parasites, particularly through improperly prepared food or direct contact with household pets or farm animals.6
Antiparasitic Drugs
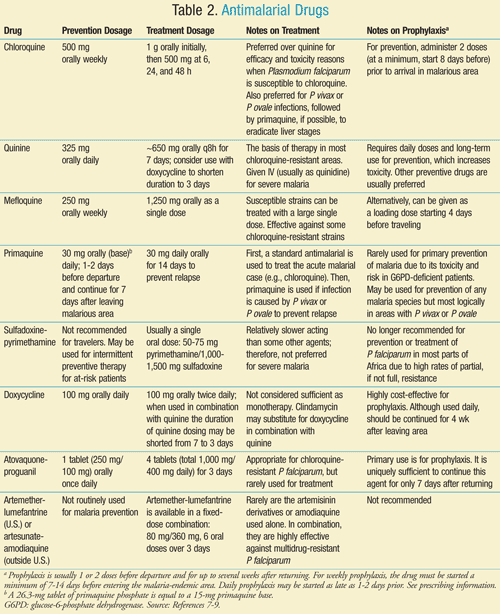
Antimalarial Agents: The most common use of agents against parasites by U.S. residents is for malaria prophylaxis. The same arsenal of drugs is used for both prevention and treatment of malaria. TABLE 2 highlights the most important antimalarial drugs for U.S. pharmacists.7-9 To achieve therapeutic levels, it is generally necessary for travelers to take 2 doses before entering a malaria-endemic area and to continue for 4 weeks after returning. Atovaquone-proguanil is an exception and only needs to be continued for 1 week after returning.10,11
Pharmacists should educate their patients that continuing the doses (for 1-4 weeks) after returning home is vital for successful antimalarial prophylaxis. Antimalarial drugs do not prevent the acquisition of malaria nor do they sufficiently prevent parasitic migration to or occupation of the liver. Instead, these drugs treat the later blood stages that cause clinical malaria. Therefore, when used for prophylaxis, these drugs must be continued for a time after the traveler returns from the malaria-endemic area in case of unknown acquisition of a Plasmodium species.
Pharmacists should also inform their patients that while pharmacologic prophylaxis is important, the best way to prevent malaria is to avoid the bite of the Anopheles mosquito, which is most active in the early evening. Preventive methods include wearing long-sleeved and long-legged clothing, applying strong insect repellants, and using mosquito nets.10
Not all drugs approved for malaria treatment or prophylaxis are included in TABLE 2. For example, amodiaquine and hydroxychloroquine are infrequently used for monotherapy against Plasmodium falciparum today due to resistance (although they are occasionally effective against chloroquine-resistant strains).7,9 Similarly to quinine, they are infrequently used for prevention because they require daily dosing and also have higher rates of toxicity (e.g., blood dyscrasias, liver toxicity).
Metronidazole and Tinidazole: Metronidazole, or alternatively tinidazole, is the basis of therapy for giardiasis, trichomoniasis, and amebiasis. Of these, amebiasis presents the greatest challenge because metronidazole alone is often not sufficient to eradicate this intestinal parasite.12 Drug therapy for amebiasis should generally consist of a “luminal agent” that is capable of eradicating the luminal (intestinal) infection. If the infection is only luminal and the patient is asymptomatic, then a luminal agent may be used alone.13 The luminal agents available for use in the U.S. are iodoquinol and paromomycin.12 However, if there are symptoms or the disease is extraintestinal, it is imperative to use a systemic agent as well, such as metronidazole or tinidazole. Metronidazole 750 mg is given orally three times daily or 500 mg intravenously every 6 hours for 10 days. Tetracycline (when available) is the most commonly used alternative systemic agent for mild-to-severe intestinal infection; another alternative, erythromycin, can be considered for mild or moderate infection.
For a hepatic abscess or other extraintestinal disease, metronidazole or tinidazole is still the preferred agent and may be combined with chloroquine for added efficacy. For cases of resistance or intolerance to metronidazole and tinidazole, rarely dehydroemetine or emetine plus chloroquine for 21 to 23 days may have to be used. Dehydroemetine and emetine must be given either subcutaneously or intramuscularly and are not commercially available in the U.S.12
Treatment of giardiasis is simpler and can generally be accomplished with a low-dose course of metronidazole (e.g., 250 mg three times daily with meals for 5 days) or a single 2-g dose of tinidazole with a meal. If metronidazole or tinidazole cannot be used or is ineffective, nitazoxanide or paromomycin is also an option.12,14
Trichomoniasis, whether it is vaginal or urethral (most common sites), is also easily treated, often with a single 2-g dose of metronidazole. However, this large single dose frequently causes dyspepsia, and alternatively metronidazole 500 mg twice daily over 7 days (or similar regimen) may be prescribed. Tinidazole may also be substituted for metronidazole and is at least as effective. It is important to note that intravaginal metronidazole is intended for bacterial vaginosis, not Trichomonas vaginalis.12,15,16
Adverse effects occur frequently with metronidazole and may include nausea, parageusia, dermatologic reactions (occasionally severe), and vaginal candida infection. Some neurologic reactions are common, like dizziness or headache, but severe neurologic reactions may also occur, including peripheral neuropathy, seizure, and disorders of special senses (optic or auditory nerve disorders). Other serious adverse effects may include leukopenia, aseptic meningitis, encephalopathy, and hemolytic uremic syndrome. Alcohol should be avoided due to the risk of a disulfiram-like reaction.17
Ivermectin: Ivermectin is one of the more frequently used antiparasitic drugs in the U.S., but not for the most impactful parasitic infections mentioned above. While it is FDA approved for systemic infections by Onchocerca volvulus (microfilariae, not adult) and intestinal strongyloidiasis, it is most frequently used in the U.S. off-label for scabies, lice, and enterobiasis (pinworms).18 Just south of the U.S. in southern Mexico and Guatemala, the availability of ivermectin is particularly important in the effort to eradicate onchocerciasis (“river blindness”).19
Like other agents, ivermectin paralyzes certain helminths, but its molecular mechanism of action is different. Ivermectin intensifies GABA-mediated transmission, which causes postsynaptic neuron hyperpolarization in susceptible helminths.20 Enhancement of GABA activity may occur in humans as well, and ivermectin should not be coadministered with GABA-ergic drugs.21 Taking ivermectin with food (specifically, a high-fat meal) greatly increases bioavailability.17 Ivermectin is usually given as a single dose. However, when treating filariasis, a single dose is effective in reducing symptoms for many months but rarely induces remission. Repeat doses may be needed once or twice yearly for several years until the infection eventually subsides.21
Adverse effects of ivermectin are infrequent but may include fatigue, dizziness, abdominal pain, or rash. Reactions due to microfilariae death may occur within the first 2 days of treatment and include arthralgias, myalgias, hypotension, tachycardia, lymphadenitis, lymphangitis, and peripheral edema.21
Pyrantel Pamoate: Pyrantel is available OTC and is primarily used for the treatment of enterobiasis, commonly called pinworms (Enterobius vermicularis). Enterobius usually infects only the gastrointestinal (GI) tract, but can also occasionally infect the female genitourinary system and may increase the rates of bacterial urinary tract infections.22 Pyrantel pamoate has some efficacy against ascariasis and has occasionally been used as an alternative to mebendazole for hookworms.3,21 It is not absorbed systemically and is thus only effective against susceptible helminths within the intestinal tract. Therefore, adverse effects are usually GI in nature, although dizziness, headache, or somnolence may occur.
Albendazole: Albendazole is at least as efficacious as mebendazole for the treatment of many helminth infections.23,24 Albendazole is only FDA approved for the treatment of echinococcosis and neurocysticercosis.25 However, due to the recent unavailability of mebendazole, off-label use of albendazole may be required for the treatment of certain roundworms (i.e., ascariasis, hookworms, enterobiasis). Pyrantel pamoate, available OTC for the treatment of pinworms (enterobiasis), may also be a reasonable choice for ascariasis.26 Dosing recommendations are available in most drug references for off-label uses of albendazole.
There are some important pharmacokinetic properties to consider. Albendazole may be taken with or without food, and the ideal choice is based on the location of the parasitic infection. Because very little is systemically absorbed, the dosing for intestinal infections in children and adults is similar. It is important to understand that food increases albendazole absorption, particularly a fatty meal.21 For luminal infections (e.g., ascariasis), the dose can be given on an empty stomach to reduce absorption. For systemic or extraintestinal infections, albendazole can be given with food to enhance absorption.
Occasionally, serious adverse effects can occur with albendazole, especially during prolonged therapy or during certain infections (ocular and CNS infections are discussed below). In any extended treatment course (more than 1 or 2 single doses), and especially in hydatid disease, liver enzymes and blood cell counts should be monitored every 2 weeks and before each repeat treatment course. Ongoing physical examinations are necessary and must include monitoring for liver toxicity. Albendazole should be avoided or discontinued if liver transaminases are elevated or if the white blood cell count is depressed. The prescribing information includes a warning not to use in pregnancy and to avoid pregnancy for 1 month after discontinuation of albendazole. Fortunately, conclusive evidence indicating severe teratogenicity is lacking and is based on animal models; if it occurs, it is infrequent.27 Still, other agents are preferred over albendazole in pregnancy if appropriate for the clinical scenario (e.g., praziquantel for schistosomiasis).28
Adverse Effects of Antihelminthic Drugs
Memorizing a list of adverse effects for each antihelminthic drug is impractical in real clinical practice. Adverse effects that might be attributed to a drug are highly dependent on the infection being treated and the duration of therapy. An interesting example of the infection-specific adverse-effect profile of these medications is the prevalence of liver enzyme elevation in albendazole therapy for hydatid disease versus neurocysticercosis. In hydatid disease, abnormal liver function tests are seen 15.6% of the time but less than 1% of the time in patients with neurocysticercosis.25 Some adverse effects may be due to the effects of the dying helminths rather than the direct toxicity of the agents.
Special Considerations When Treating Ocular or CNS Infections
Patients with neurocysticercosis often present with seizures. Interestingly, this infection is an increasingly emerging infection within the U.S. In the U.S., seizures are the most common first-onset symptom (66% of patients), and antiseizure drug therapy is usually necessary, especially if antihelminthic pharmacotherapy is chosen. Neurocysticercosis is the most common cause of symptomatic seizures worldwide.29
Many drugs that would be effective in vitro for the treatment of a particular parasite may not reach the site of action when the infection is extraintestinal. GI absorption and distribution to the site of action must be considered. As previously mentioned, treatment of extraintestinal infections with albendazole may be enhanced by giving the drug with food. The use of a glucocorticoid (e.g., prednisone) can greatly reduce morbidity when treating certain CNS manifestations of parasitic infections, particularly certain forms of neurocysticercosis—cysticercal encephalitis, subarachnoid neurocysticercosis, and spinal intramedullary cysticercosis.26 Additionally, combining a glucocorticoid with albendazole may increase drug action and reduce damaging inflammation at the site of infection.21
Antiparasitic drug therapy is not always indicated for CNS or intraocular infections because dying helminths may cause more damage than live ones. Praziquantel in particular should be avoided when treating ocular infections, and rarely is it indicated when there are signs of CNS involvement. Use of praziquantel for infections in these sensitive tissues is further complicated by drug interactions with dexamethasone (and perhaps prednisone), as coadministration significantly reduces plasma praziquantel levels.21 If there is ocular or CNS involvement, an ophthalmologist or neurologist should be consulted prior to drug therapy.
Availability of Antiparasitic Agents
Many of the most toxic agents used for parasitic infections around the world are not available in the U.S. due to low occurrence of infections here. Because of this low occurrence, there has also been little financial incentive for the pharmaceutical industry in the U.S. and Western Europe to develop more effective, less toxic agents. If there is a need for agents not approved for use in the U.S., some may be procured directly from the CDC if there is an existing Investigational New Drug (IND) application on file with the FDA.30
Role of the Pharmacist
In conclusion, pharmacists should be aware of the following when encountering parasitic infections in their practice:
• Although mebendazole is now unavailable in the U.S., it is still the recommended therapy for many helminthic infections. There is no clear single alternative, but the pharmacist can help guide the prescriber and patient to the appropriate use and dosage of albendazole, pyrantel pamoate, or other antihelminthic agent.
• When prescribing under protocol, pharmacists can be an excellent clinical provider for travel consultations, a part of which is prescribing appropriate antimalarial prophylaxis that depends on a variety of patient factors/preferences and the travel destination(s).
• Pharmacists should recommend involvement of a specialist in the treatment of neurologic or ocular infections and provide information on use of anti-inflammatory drugs and their pharmacokinetic and pharmacodynamic interactions with antiparasitic agents.
• Pharmacists Pharmacists should recognize pinworm infection (enterobiasis) and recommend OTC therapy (pyrantel pamoate) as appropriate.
REFERENCES
1. Kok M, Pechere JC. Nature and pathogenicity of micro-organisms. In: Cohen J, Powderly WG, Opal SM, eds. Cohen & Powderly: Infectious Diseases. 3rd ed. New York, NY: Mosby Elsevier; 2010:5. www.mdconsult.com. Accessed July 6, 2012.
2. Montoya JG, Boothroyd JC, Kovacs JA. Toxoplasma gondii. In: Mandell GL, Bennett JE, Dolin R, eds. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases. 7th ed. Philadelphia, PA: Churchill Livingstone Elsevier; 2010:3495-3527.
3. Maguire JH. Intestinal nematodes (roundworms). In: Mandell GL, Bennett JE, Dolin R, eds. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases. 7th ed. Philadelphia, PA: Churchill Livingstone Elsevier; 2010:3577-3587.
4. Parasites—Cysticercosis. CDC. Updated November 2, 2010.
www.cdc.gov/parasites/cysticercosis/health_professionals/index.html.
Accessed February 16, 2012.
5. The Global Burden of Disease: 2004 Update. World Health Organization: Geneva, Switzerland; 2008:54.
6. Parasites. CDC. Updated February 1, 2012. www.cdc.gov/parasites. Accessed July 6, 2012.
7. Rosenthal PJ. Antiprotozoal drugs. In: Katzung BG, Masters SB, Trevor AJ, eds. Basic and Clinical Pharmacology. 12th ed. New York, NY: McGraw-Hill Lange; 2012:915-936.
8. Piola P, Fogg C, Bajunirwe F, et al. Supervised versus
unsupervised intake of six-dose artemether-lumefantrine for treatment of
acute, uncomplicated Plasmodium falciparum malaria in Mbarra, Uganda: a randomised trial. Lancet. 2005;365:1467-1473.
9. Arguin PM, Mali S. Chapter 3—Malaria. In: The Yellow Book.
CDC. Updated November 8, 2011.
wwwnc.cdc.gov/travel/yellowbook/2012/chapter-3-infectious-diseases-related-to-travel/malaria.htm.
Accessed February 20, 2012.
10. Atovaquone; Proguanil. Drugs. MD Consult [subscription required]. www.mdconsult.com. Accessed March 15, 2012.
11. Malaria. CDC. Updated October 26, 2011. www.cdc.gov/malaria. Accessed February 16, 2012.
12. Rosenthal PJ. Antiprotozoal drugs. In: Katzung BG, Masters SB, Trevor AJ, eds. Basic and Clinical Pharmacology. 11th ed. New York, NY: Lange Medical Publications; 2009:899-921.
13. Petri WA, Haque R. Entamoeba species, including amebiasis. In: Mandell GL, Bennett JE, Dolin R, eds. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases. 7th ed. Philadelphia, PA: Churchill Livingstone Elsevier; 2010:3411-3426.
14. Parasites—Giardia. CDC. Updated November 2, 2010. www.cdc.gov/parasites/giardia/treatment.html. Accessed February 20, 2012.
15. Schwebke JR. Trichomonas vaginalis. In: Mandell GL, Bennett JE, Dolin R, eds. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases. 7th ed. Philadelphia, PA: Churchill Livingstone Elsevier; 2010:3535-3559.
16. MetroGel vaginal (metronidazole) package insert. Northridge, CA: 3M Pharmaceuticals; 1997.
17. Metronidazole. Micromedex 2.0 [subscription required]. www.thomsonhc.com. Accessed July 10, 2012.
18. Ivermectin. Lexi-Comp [online database]. Hudson, OH: Lexi-Comp, Inc; 2012. www.lexi.com. Accessed March 15, 2012.
19. Neglected tropical diseases. The path to the
elimination of onchocerciasis in the Americas. CDC. Updated June 6,
2011. www.cdc.gov/globalhealth/ntd/features/oncho_launch.html. Accessed
March 1, 2012.
20. Stromectol (ivermectin) package insert. Whitehouse Station, NJ: Merck & Co., Inc; May 2010.
21. Rosenthal PJ. Clinical pharmacology of the antihelminthic drugs. In: Katzung BG, Masters SB, Trevor AJ, eds. Basic and Clinical Pharmacology. 12th ed. New York, NY: McGraw-Hill Lange; 2012:937-947.
22. Ok UZ, Ertan P, Limoncu E, et al. Relationship between pinworm and urinary tract infections in young girls. APMIS. 1999;107:474-476.
23. Steinmann P, Utzinger J, Du ZW, et al. Efficacy of
single-dose and triple-dose albendazole and mebendazole against
soil-transmitted helminths and Taenia spp.: a randomized controlled trial. PLoS One. 2011;6:e25003.
24. Namwanje H, Kabatereine NB, Olsen A. Efficacy of
single and double doses of albendazole and mebendazole alone and in
combination in the treatment of Trichuris trichiura in school-age children in Uganda. Trans R Soc Trop Med Hyg. 2011;105:586-590.
25. Albenza (albendazole) package insert. Research Triangle Park, NC: GlaxoSmithKline; 2009.
26. Wilson CM, Freedman DO. Antiparasitic agents. In: Long SS, Pickering LK, Prober CG, eds. Principles and Practice of Pediatric Infectious Disease. 3rd ed. Philadelphia, PA: Churchill Livingstone Elsevier; 2008:1488-1506.
27. Albendazole. Micromedex 2.0 [subscription required]. www.thomsonhc.com. Accessed July 10, 2012.
28. Praziquantel. Micromedex 2.0 [subscription required]. www.thomsonhc.com. Accessed July 10, 2012.
29. Wallin MT, Kurtzke JF. Neuroepidemiology. In: Daroff RB, Fenichel GM, Jankovic J, Mazziotta J, eds. Bradley’s Neurology in Clinical Practice. 6th ed. Philadelphia, PA: Elsevier Saunders; 2012:702-703.
30. CDC Drug Service. Updated November 3, 2010. www.cdc.gov/laboratory/drugservice/. Accessed March 3, 2012.
To comment on this article, contact rdavidson@uspharmacist.com.





