US Pharm. 2017;42(1):HS21-HS24.
ABSTRACT: Epilepsy and the corresponding seizure disorders, such as focal (partial-onset) seizures, provide challenges for both patients and healthcare providers. Although many current treatment options show benefit in some patients, other patients have seizures that fail to respond to medications. Traditional medications, such as phenytoin, carbamazepine, and oxcarbazepine, are known for drug-drug interactions and intolerable adverse effects. Research has resulted in treatment advances for focal seizures. One new medication, eslicarbazepine, has been proven safe and effective for the treatment of focal seizure disorder. With convenient dosing and improved tolerability, eslicarbazepine and other new agents are considered suitable alternatives to traditional antiepileptics in selected patients.
Epilepsy is the fourth most common neurologic disorder, following migraine, stroke, and Alzheimer’s disease. It affects nearly 65 million people worldwide, and although many treatment options exist, almost one-third of patients have intractable forms of epilepsy. Unfortunately, patients who have seizures face many challenges, such as social stigma, discrimination based on the unknown, and stress caused by the notoriously unpredictable nature of the disease.1-3
The International League Against Epilepsy considers epilepsy to be present if any of the following conditions are met: two unprovoked seizures more than 24 hours apart; one unprovoked seizure with a probability of additional seizures that is similar to the general recurrence risk after two unprovoked seizures (at least 60%) occurring over the next 10 years; or a diagnosis of an epilepsy syndrome.4
Although there are many different types of epilepsy and seizure disorders, the discussion in this article will be limited to partial-onset seizures (more recently termed focal seizures). Focal seizures have a new classification system and have been the focus of many new drug therapies. These changes have enabled healthcare providers to improve the treatment approach for patients with focal seizure disorder.
Pathophysiology
A seizure occurs when there is excessive excitation or inhibition within a large group of neurons. It generally begins with an initial spike in activity that disturbs normal synapses. This spike is followed by abnormal hypersynchronization of the excitatory conductances across a large cortical area. In the case of focal seizures, this activity is localized within one cerebral hemisphere.5
Biochemical imbalances in neurotransmitters, receptor modification, changes in extracellular ion concentrations with an increase in potassium or buildup of presynaptic calcium, and alteration in the properties of ion channels are some of the mechanisms known to lead to seizure activity. The abnormal currents may be mediated by voltage-gated ion channels, including sodium or calcium channels, or by gamma-aminobutyric acid and glutamate through ligand-gated channels. These variations provide multiple targets for drug therapy.5
Classifying Focal Seizures
Traditional classification of partial-onset seizures depended on the extent to which a patient’s awareness was affected. If a patient was awake during the seizure, he or she was said to have had a simple partial seizure; if the patient was absent (confused and disoriented) after the seizure, he or she was said to have had a complex partial seizure. Although these terms made it easier to classify these common types of seizures, this nomenclature is no longer preferred and recently was eliminated. The presence or absence of cognitive impairment, as well as additional symptoms, is now considered in characterizing seizures, allowing for further specificity. It is important to note that a focal seizure can progress to a generalized seizure with tonic-clonic symptoms, but the latter is distinguishable from primary generalized seizure only with use of an electroencephalogram. For a brief overview of the different types of focal seizures, see TABLE 1.6
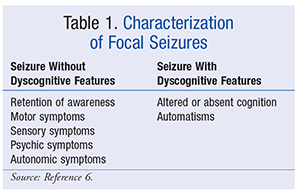
Focal Seizure Without Dyscognitive Features: Focal seizures may present with motor, sensory, psychic, or autonomic features without loss of cognition.6 The manifestations of unwanted movements or uncontrolled alterations in hearing or smell are common in patients experiencing this type of focal seizure. These patients may report déjà vu, a sense of impending doom, or anxiety. These intuitive changes, often referred to as aura, are common in most focal seizures.
Focal Seizure With Dyscognitive Features: When a patient is unable to respond appropriately during a seizure, appears to have lost the ability to recall the seizure, or loses awareness of his or her environment, the seizure is classified as a focal seizure with dyscognitive features. This type of seizure is often preceded by an aura, and the patient may appear to be in a state of absence, often described as a daydream. Patients occasionally display involuntary movements or behaviors—known as automatisms—such as swallowing, chewing, or other behaviors such as picking with the hands.6 The patient may seem disoriented for an extended period of time following a focal seizure with dyscognitive features.
Treatment
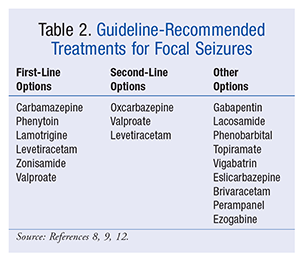
For all types of epilepsy, the goal of treatment is to attain seizure freedom.7 Many options are available to treat partial-onset seizures, either as monotherapy or as an adjunct to another treatment. Some of the best-studied medications for this indication include carbamazepine, phenytoin, levetiracetam, and lamotrigine, with carbamazepine and phenytoin having the largest body of evidence for their use.8,9 Other antiepileptic drugs (AEDs) with evidence-based support are valproate, gabapentin, lacosamide, oxcarbazepine, phenobarbital, topiramate, vigabatrin, and zonisamide. Clonazepam and primidone have limited evidence for use in focal seizures.9
The question of when to start an AED in the treatment of epilepsy is of considerable importance. Recurrence after the first unprovoked seizure is relatively high among generalized seizures.7 Recurrences are most likely during the first 2 years after the event, but this risk is lessened if the patient is taking an AED.10 Evidence supports the initiation of AEDs after the first unprovoked seizure, but the consensus is that treatment is indicated after at least two unprovoked seizures.7 This is based on the fact that many studies examining initial treatment of epilepsy included patients who had at least two untreated seizures.11 If initiation after the first seizure is deemed appropriate, it is recommended that the decision to start treatment after the first unprovoked seizure be weighed against the adverse effects (AEs) of AEDs and the risk of another occurrence.10
When an agent is being selected for treatment, guidelines recommend tailoring therapy to the individual and his or her seizure type,7 with a single agent being used first. Treatment options given in epilepsy guidelines are listed in TABLE 2.8,9,12 If the first agent fails because the patient cannot tolerate a certain dosage, experiences intolerable AEs from the drug, or has nonresolution of seizures, then a second monotherapy agent that is either first-line or second-line should be started. The new medication should be slowly titrated to goal or maximum tolerated dose, at which point the first medication may be slowly tapered down. Adjunctive therapy should be considered if the patient does not reach seizure freedom or if two well-tolerated medications have been tried and have failed to control seizures adequately.12 However, the majority of patients (~70%) will achieve seizure freedom, and most will respond to the initial medication.13
Eslicarbazepine: Eslicarbazepine acetate (ESL) is a newer AED that is structurally similar to carbamazepine (CBZ) and oxcarbazepine (OXC). ESL was FDA-approved in 2013 for the adjunctive treatment of partial-onset seizures, and in 2015 its approval was expanded to monotherapy in the treatment of partial-onset seizures. Similar to CBZ and OXC, ESL works by stabilizing inactivated voltage-gated sodium channels (VGSCs), thus inhibiting the communication of excitatory stimuli that may contribute to the formation or continuation and propagation of seizure activity. ESL differs from its structural counterparts in that it is more selective for VGSCs and has more affinity for the inactivated state compared with CBZ and OXC; however, the clinical significance of this is unknown. ESL acetate undergoes first-pass metabolism to ESL, the main active metabolite of OXC. The majority remains as ESL and is excreted as such.14
Compared with CBZ, ESL does not appear to induce its own metabolism; however, it still has effects on the CYP450 enzymes. Although its effect on metabolism is less significant than that of CBZ, ESL is considered a modest inhibitor of CYP2C19 and a modest inducer of CYP3A4. Therefore, ESL still has a clinically relevant interaction with oral contraceptives, warfarin, and any statins that are metabolized by CYP3A4,14,15 as well as a theoretical interaction with clopidogrel because of its CYP2C19 inhibition.16 ESL acetate should be adjusted for renal function, with a recommended dose decrease of 50% in moderate-to-severe renal impairment (creatinine clearance <50 mL/min). ESL has not been studied in patients with severe hepatic impairment and should not be used in this population; however, no adjustments are necessary if it is used in mild-to-moderate hepatic impairment.17
Perhaps one of the greatest advantages of ESL is its once-daily dosing. OXC has an extended-release formulation that is dosed once daily, but CBZ should be dosed at a minimum of twice daily.15 The most common AEs of ESL are dizziness, somnolence, nausea, headache, diplopia, vomiting, fatigue, vertigo, ataxia, blurred vision, and tremor.17 ESL is associated with clinically relevant hyponatremia, but it has the lowest incidence compared with CBZ and OXC. Drug-induced liver injury and abnormal thyroid-function tests have also been observed.14 Rash was reported in 1% to 3% of patients taking ESL, an incidence that is significantly higher than that for CBZ or OXC.15 However, incidence rates of Stevens-Johnson syndrome and toxic epidermal necrolysis have not been reported with ESL even though these disorders have been reported with its use.17 It is recommended that ESL not be used in a patient who experienced a serious dermatologic reaction to CBZ or OXC.18
The initial recommended dosage of ESL is 400 mg once daily, with increases of 400 to 600 mg once daily every week to a target of 800 to 1,600 mg daily. The only available formulation of ESL is a tablet, which may be crushed and taken with or without food. ESL may be taken with CBZ; however, because ESL is a metabolite of OXC, ESL and OXC should not be taken in combination. If ESL is taken with CBZ, the dose of ESL must be adjusted because of the induction effect of CBZ on the enzymes by which ESL is metabolized. When ESL is discontinued, it should be tapered slowly in order to avoid withdrawal symptoms such as increased seizure frequency or status epilepticus. ESL has no boxed warnings, but this does not rule out the possibility of AEs similar to those occurring with CBZ and OXC.17
Monitoring parameters for ESL include serum sodium levels (in patients at risk for hyponatremia), neurologic AEs, and suicidal behavior or ideation. The international normalized ratio should be monitored in patients taking warfarin concomitantly.14 Baseline liver-function tests should also be performed owing to the risk of liver injury.17
The role of ESL will continue to emerge as this drug is used more frequently for the treatment of focal seizures. ESL’s once-daily dosing may result in better adherence in patients in whom medication compliance is a concern. The lower incidence of some AEs could be advantageous, especially in patients who have failed therapy with more traditional AEDs because they could not tolerate them. ESL is not a controlled substance, so the risk of addiction is not a concern compared with certain other agents used to treat seizure disorders.
Other Recent Approvals for Focal Seizures: Besides ESL, several other drugs have been approved over the past few years for partial-onset seizures (TABLE 3). Brivaracetam (Briviact) was approved for partial-onset seizures in early 2016. Like levetiracetam, brivaracetam binds to synaptic vesicle protein 2A; however, it does so with higher affinity. It has a low potential for drug-drug interactions, but it may increase the concentration of CBZ epoxide, an active metabolite of CBZ, which will likely increase CBZ-related AEs and require attentive monitoring.19
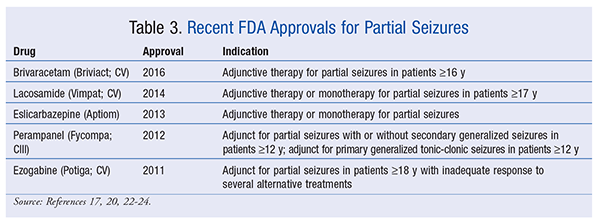
Perampanel is an AMPA (alpha-3-hydroxy-5-methyl-4-isoxazolepropionic acid) receptor antagonist that was originally approved for use in partial-onset seizures, and more recently approved for generalized tonic-clonic seizures.20 This medication might be best used in patients with focal seizures that progress to generalized seizures.
Lacosamide is an AED with a novel mechanism of enhancing slow inactivation of VGSCs, which stabilizes hyperexcitable neurons. Many of the other AEDs that act on VGSCs appear to stabilize the fast inactivation.21 The clinical significance of this alternative mechanism of action is unclear.
Ezogabine holds a narrow place in therapy since it is approved only for patients who have failed several other treatments, and in whom the benefit of treatment outweighs the risks of potential vision loss. Ezogabine has a black box warning for causing retinal abnormalities that can lead to vision loss, so it requires an extensive eye examination every 6 months.22
Role of the Pharmacist
The complexity of seizure disorders and FDA approval of a variety of new drug therapies render the role of the pharmacist increasingly important in the treatment of patients with epilepsy or seizure disorders. Additionally, many AEDs are used for multiple off-label indications, making these medications widely prescribed. As with any treatment, the pharmacist’s primary role is to counsel patients regarding AEs and safe usage of the medications. Whether in the hospital or the retail setting, the pharmacist should review medication lists and address medication-related problems. Finally, the pharmacist should make recommendations and communicate with providers when there are concerns or questions about a patient’s drug regimen.
Conclusion
Epilepsy is a complex disorder that can lead to further morbidity. Although there are many different treatment options with varying levels of evidence supporting their use, CBZ is one of the best-studied medications for treating partial-onset seizures. ESL acetate is a new, easier-to-manage, more convenient cousin of CBZ that has been approved for the treatment of partial-onset seizures. Although many of its pharmacokinetic and pharmacodynamic features are similar to those of CBZ and OXC, ESL has fewer AEs and the benefit of once-daily dosing. ESL is an advancement in the treatment of seizures, and with its advantages over traditional AEDs, it may play an emerging role in the treatment of epilepsy and, more specifically, focal seizure disorder.
REFERENCES
1. Epilepsy Foundation. Information for professionals. www.epilepsy.com/information/information-professionals. Accessed September 6, 2016.
2. Thurman DJ, Beghi E, Begley CE, et al; ILAE Commission on Epidemiology. Standards for epidemiologic studies and surveillance of epilepsy. Epilepsia. 2011;52(suppl 7):2-26.
3. Taylor RS, Sander JW, Taylor RJ, Baker GA. Predictors of health-related quality of life and costs in adults with epilepsy: a systematic review. Epilepsia. 2011;52:2168-2180.
4. Trinka E, Cock H, Hesdorffer D, et al. A definition and classification of status epilepticus—Report of the ILAE Task Force on Classification of Status Epilepticus. Epilepsia. 2015;56:1515-1523.
5. Najm IM, Moddel G, Janigro D. Mechanisms of epileptogenesis and experimental models of seizures. In: Wyllie E, ed. The Treatment of Epilepsy. 4th ed. Philadelphia, PA: Lippincott Williams & Wilkins; 2006:91-102.
6. Lowenstein DH. Seizures and epilepsy. In: Kasper DL, Fauci AS, Hauser S, et al, eds. Harrison’s Principles of Internal Medicine. 19th ed. New York, NY: McGraw-Hill Medical; 2015.
7. Perucca E, Tomson T. The pharmacological treatment of epilepsy in adults. Lancet Neurol. 2011;10:446-456.
8. Glauser T, Ben-Menachem E, Bourgeois B, et al. ILAE treatment guidelines: evidence-based analysis of antiepileptic drug efficacy and effectiveness as initial monotherapy for epileptic seizures and syndromes. Epilepsia. 2006;47:1094-1120.
9. Glauser T, Ben-Menachem E, Bourgeois B, et al. Updated ILAE evidence review of antiepileptic drug efficacy and effectiveness as initial monotherapy for epileptic seizures and syndromes. Epilepsia. 2013;54:551-563.10. Krumholz A, Wiebe S, Gronseth GS, et al. Evidence-based guideline: management of an unprovoked first seizure in adults. Report of the Guideline Development Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Neurology. 2015;84:1705-1713.
11. French JA, Kanner AM, Bautista J, et al. Efficacy and tolerability of the new antiepileptic drugs I: treatment of new onset epilepsy: report of the Therapeutics and Technology Assessment Subcommittee and Quality Standards Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Neurology. 2004;62:1252-1260.
12. National Institute for Health and Care Excellence. Epilepsies: diagnosis and management. www.nice.org.uk/guidance/cg137. Accessed September 20, 2016.
13. Moshé S, Perucca E, Ryvlin P, Tomson T. Epilepsy: new advances. Lancet. 2015;385:884-898.
14. Shirley M, Dhillon S. Eslicarbazepine acetate monotherapy: a review in partial-onset seizures. Drugs. 2016;76:707-717.
15. Gierbolini J, Giarratano M, Benbadis SR. Carbamazepine-related antiepileptic drugs for the treatment of epilepsy—a comparative review. Expert Opin Pharmacother. 2016;17:885-888.
16. Lexicomp Online: Lexi-Drug Interactions. Hudson, OH: Lexi-Comp, Inc; 2016. http://online.lexi.com. Accessed September 26, 2016.
17. Aptiom (eslicarbazepine) package insert. Marlborough, MA: Sunovion Pharmaceuticals Inc; September 2016.
18. Eslicarbazepine acetate (Aptiom) for epilepsy. Med Lett Drugs Ther. 2014;56:42-43.
19. Hoy SM. Brivaracetam: a review in partial-onset (focal) seizures in patients with epilepsy. CNS Drugs. 2016;30:761-772.
20. Fycompa (perampanel) package insert. Woodcliff Lake, NJ: Eisai, Inc; April 2016.
21. Hebeisen S, Pires N, Loureiro AI, et al. Eslicarbazepine and the enhancement of slow inactivation of voltage-gated sodium channels: a comparison with carbamazepine, oxcarbazepine and lacosamide. Neuropharmacology. 2015;89:122-135.
22. Potiga (ezogabine) package insert. Research Triangle Park, NC: GlaxoSmithKline; May 2016.
23. Briviact (brivaracetam) package insert. Smyrna, GA: UCB, Inc; March 2016.
24. Vimpat (lacosamide) package insert. Smyrna, GA: UCB, Inc; August 2014.
To comment on this article, contact rdavidson@uspharmacist.com.






