US Pharm. 2010;35(8):HS2-HS4.
Methicillin-resistant Staphylococcus aureus (MRSA) is one of the leading causes of both health care– and community-acquired infections.1,2 Infections due to MRSA are associated with a high risk of morbidity and mortality and lead to high health care costs.3-5 The incidence of MRSA infections in the United States has risen steadily over the past few years from 22% in 1995 to 60% in 2003.6 Health care–associated MRSA (HA-MRSA) is related to hospitals and long-term care facilities and is common in patients with prolonged hospitalization, past antimicrobial usage, decubitus ulcers, dialysis, indwelling catheters, enteral feedings, or history of IV drug abuse.7,8 In recent years, however, there has been an increase in a new strain of MRSA called community-associated MRSA (CA-MRSA). This seems to be acquired in the outpatient setting, and transmission is through close physical contact such as in day care centers, Indian reservations, and correctional facilities and among athletes, military personnel, and men who have sex with men.7,9,10
Distinguishing MRSA Strains
Several reports have revealed differences between HA-MRSA and CA-MRSA strains, including the presence of different genotypes between the two strains. CA-MRSA is susceptible to several non-beta-lactam antibiotics, while HA-MRSA strains are not. CA-MRSA strains are more likely to encode certain virulent factors that have been associated with severe pneumonia (especially in children) and skin and soft tissue infections in adults.8,11,12 The CDC has established the criteria for differentiating CA-MRSA from HA-MRSA. These guidelines include isolating a positive MRSA culture within 48 hours of a patient’s admission to the hospital. To be considered CA-MRSA, the patient must not have any of the following: recent hospitalization; admission to a nursing home, hospice, or skilled nursing facility; dialysis; or recent surgery. The patient must also not have any indwelling catheters or other medical devices.13
Treating CA-MRSA
The CDC recommends that the management and prevention of outbreaks of CA-MRSA include covering infections that produce pus, washing hands frequently, not sharing personal items, washing soiled linens and clothing with hot water, and drying cloths in a hot dryer compared to air drying.13 For skin abscesses, treatment includes incision and drainage of the abscesses with or without systemic antibiotics, depending on the severity of the infection.14
Treatment of CA-MRSA usually includes the use of agents like clindamycin, tetracyclines, and trimethoprim-sulfamethoxazole (TMP-SMX). The antibiotic rifampin should never be used as monotherapy for the management of MRSA. Resistance develops quickly with rifampin monotherapy; however, when used in combination with another anti-MRSA agent, it can eradicate organisms from carrier sites more effectively.15 The aforementioned antibiotics are usually viable treatment options in patients treated on an outpatient basis. In severe infections that require hospitalization, IV antibiotics including vancomycin, linezolid, quinupristin-dalfopristin, daptomycin, tigecycline, and telavancin are appropriate therapies that can be used (TABLE 1).
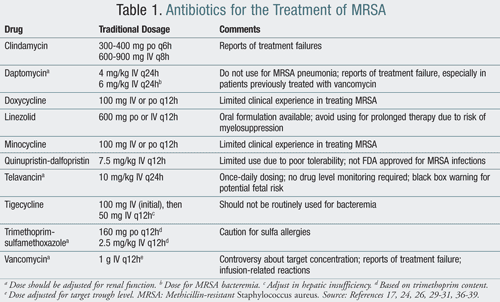
Although clindamycin (Cleocin) could be used for CA-MRSA, resistance has been reported to be up to 19% in patients treated with it. Thus, patients treated with clindamycin must be monitored more closely for the development of self-induced resistance.16 Clindamycin has also been associated with the development of Clostridium difficile infections, limiting its use over an extended period.17
TMP-SMX (Bactrim) may be considered an alternative to vancomycin therapy in some cases of MRSA infections. However, as with clindamycin, there have been reports of treatment failures. This therapy also has limited use due to the high incidence of sulfonamide allergies, which havebeen reported to occur in up to 10% of the population.18
Minocycline (Minocin) and doxycycline (Vibramycin) are tetracycline antibiotics that have been used for the treatment of MRSA. These agents have only limited data on use in MRSA, but clinical success has been reported.19
Treating HA-MRSA
Parenteral antibiotics are considered the gold standard for treatment of HA-MRSA. For several decades, the glycopeptide antibiotic vancomycin has been widely used.
Vancomycin (Vancocin) inhibits cell wall synthesis; however, it has slower bactericidal activity in vitro. With its overuse, there have been reports of reduced susceptibilities and the emergence of vancomycin heteroresistance in S aureus.20,21 The increase in a subpopulation of S aureus with minimum inhibitory concentration (MIC) of >2 mcg/mL to vancomycin (referred to as MIC creep) is associated with both microbiologic and clinical failure. The National Committee for Clinical Laboratory Standards (NCCLS) has proposed new breakpoints for vancomycin—changing the MIC for “susceptible” from ≤4 mcg/mL to ≤2 mcg/mL.22 The rationale for this proposal is partly based on anecdotal clinical observations of treatment failures during treatment with standard doses of vancomycin in patients with MIC of 4 mcg/mL. In addition, higher trough levels (i.e., trough levels of 15-20 mcg/mL) may be required for serious infections like pneumonia and osteomyelitis due to poor tissue penetration by vancomycin. There have been some anecdotal reports of possible nephrotoxicity associated with these high trough levels.
Jeffres et al presented an abstract at the 2006 Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC) that reviewed the risk of nephrotoxicity with higher vancomycin trough levels.23 In their study, they reported a 30% risk of nephrotoxicity (defined as an increase of 0.5 mg/dL or 50% increase of serum creatinine from baseline) in patients with vancomycin trough levels of ³15 mcg/mL compared to 13% in patients with trough levels <15 mcg/mL.23 Some patient populations are at a higher risk of development of vancomycin-induced nephro-toxicity. These include known risk factors such as older age, being critically ill, having lower renal function, and concomitant use of other nephrotoxic agents like aminoglycosides. All of these factors have led to an increase in the need and use of the newer agents.
Linezolid (Zyvox) is the first oxazolidinone approved for the treatment of skin and soft tissue infections, bacter-emia, and community and nosocomial pneumonia caused by MRSA.24 It inhibits the initiation of protein synthesis at the 50S ribosome; however, unlike most of the other drugs, it is a bacteriostatic agent. Advantages to its use include the availability of an oral formulation that has excellent bioavailability properties, with no dose adjustment warranted in patients with renal insufficiency, including those receiving renal replacement therapies. One limiting factor to its use, especially for extended duration of therapy, is myelosuppression among patients who received linezolid for more than 14 days.24,25 Although available as an oral formulation, it is not used as much in the outpatient setting due to its cost. It is also not covered under many insurance policies, so patients may need to use the manufacturer’s patient assistance programs.
Daptomycin (Cubicin), a lipopeptide antibiotic with rapid bactericidal activity, is approved for the treatment of complicated skin and soft tissue infections, as well as bacteremia caused by MRSA.26 Its mechanism of action involves depolarization of the bacterial cell wall. Side effects of daptomycin include elevated creatine kinase (CK) and rhabdomyolysis, especially in patients taking concurrent HMG-CoA reductase inhibitors (statins). Hence, weekly CK levels must be drawn while a patient is receiving daptomycin. It must also be noted that there have been reported cases of MRSA resistance to daptomycin, especially with prolonged use and in patients with previous exposure to vancomycin.26-28 Daptomycin should not be used for the treatment of MRSA pneumonia because its activity is inhibited by pulmonary surfactants.
Quinupristin-dalfopristin (Synercid) is a streptogramin that has activity against MRSA. Although not approved by the FDA for use in MRSA infections, it is used in skin and soft tissue infections in patients who are intolerant to or fail vancomycin therapy. Because this treatment has been poorly tolerated by patients, its use is limited in clinical practice.29 Some of the major adverse events include hyperbilirubinemia, myalgias, and arthralgias. Based on these factors, this agent is used only as a last option when patients have failed or cannot tolerate other agents.
Newer Therapies
Tigecycline (Tygacil) is the first bacteriostatic agent in the glycylcycline class of antibiotics (a synthetic derivative of tetracycline). It is indicated for the treatment of complicated skin and skin structure infections (cSSSI), complicated intra-abdominal infections, and community-acquired pneumonia caused by MRSA.30 Tigecycline should not routinely be used for bacteremia due to concerns about achieving inadequate serum levels. Because tigecycline is a broad-spectrum antibiotic, its use should be restricted to serious and life-threatening infections in hospitalized patients.
Telavancin (Vibativ) is the newest drug to be FDA approved for the treatment of MRSA infections.31 It is a lipoglycopeptide similar to vancomycin, although its mechanism of action differs. Telavancin disrupts cell membrane permeability by depolarization of the plasma membrane, increasing the permeability and leakage of cellular adeno-sine triphosphate and potassium (K+). The timing of these changes correlates with rapid, concentration-dependent loss of bacterial viability, suggesting that the early bactericidal activity of telavancin results from dissipation of cell membrane potential and an increase in membrane permeability.
Clinical studies have shown that telavancin is non-inferior to vancomycin in the treatment of cSSSI.32,33 One advantage telavancin has over vancomycin is once-daily dosing and no need for the monitoring of drug levels. However, like vancomycin, the dose needs to be adjusted in patients with renal insufficiency.31 Renal function must be monitored during and after cessation of therapy in all patients. Telavancin is associated with QTc prolongation and must be used with caution in patients on medications that could also prolong QTc. In clinical trials comparing telavancin with vancomycin, adverse reactions reported in more than 10% of patients treated with telavancin included taste disturbance, nausea, vomiting, and foamy urine.31 Serious adverse events were reported in 7% of patients treated with telavancin and most commonly included renal, respiratory, or cardiac events. There is a black box warning concerning fetal risk associated with telavancin use; thus, women of childbearing age must have a serum pregnancy test prior to beginning telavancin.31
Ceftobiprole (Zeftera) is the first cephalosporin with bactericidal activity against MRSA. Its antibacterial activity includes inhibiting the penicillin-binding proteins involved in cell wall synthesis. In clinical studies, it was as effective as the combination of vancomycin and ceftazidime in the treatment of cSSSI.34 It is generally well tolerated, with main adverse drug events including nausea and taste disturbances. Ceftobiprole is not currently approved for use in the U.S., and although it was previously approved for use in Canada, the manufacturer discontinued the sale of the drug as of April 16, 2010.35 This was following recommendations from regulatory bodies in Europe (European Committee for Medicinal Products for Human Use) and the U.S. (FDA) not to approve the drug. Although phase III results suggested benefit of ceftobiprole, there were concerns as to the integrity of these study results due to tampering with clinical data from some of the study sites. With this recent development, the future of ceftobiprole use in the U.S. market remains questionable.
Conclusion
With the emergence of CA-MRSA as well as hetero-resistant MRSA, it is important that pharmacists be familiar with all agents available for the management of MRSA infections. It is also essential to choose the appropriate antimicrobial agent for the infection being treated. In addition, agents with a broader spectrum of activity (e.g., tigecycline) should be reserved for very severe infections and in the setting of multipathogen infections. It is also of utmost importance to reserve the newer agents for more severe infections in order to minimize the development of resistance.
REFERENCES
1. Boucher HW, Corey GR. Epidemiology of methicillin-resistant Staphylococcus aureus. Clin Infect Dis.
2. Styers D, Sheehan DJ, Hogan P, et al. Laboratory-based surveillance of current antimicrobial resistance patterns and trends among Staphylococcus aureus: 2005 status in the United States. Ann Clin Microbiol Antimicrob. 2006;5:2.
3. Chien JW, Kucia ML, Salata RA. Use of linezolid, an oxazolidinone, in the treatment of multidrug-resistant gram-positive bacterial infections. Clin Infect Dis. 2000;30:146-151.
4. Soriano A, Martinez JA, Mensa J, et al. Pathogenic significance of methicillin resistance for patients with Staphylococcus aureus bacteremia. Clin Infect Dis. 2000;30:368-673.
5. Graffunder EM, Venezia RA. Risk factors associated with nosocomial methicillin-resistant Staphylococcus aureus (MRSA) infection including previous use of antimicrobials. J Antimicrob Chemother. 2002;49:999-1005.
6. Klevens RM, Edwards JR, Tenover FC, et al. Changes in the epidemiology of methicillin-resistant Staphylococcus aureus in intensive care units in US hospitals, 1992–2003. Clin Infect Dis.
7. Levine DP, Cushing RD, Jui J, Brown WJ. Community-acquired methicillin-resistant Staphylococcus aureus endocarditis in the Detroit Medical Center. Ann Intern Med. 1982;97:330-338.
8. Lodise TP Jr, McKinnon PS, Rybak M. Prediction model to identify patients with Staphylococcus aureus bacteremia at risk for methicillin resistance. Infect Control Hosp Epidemiol. 2003;24:655-661.
9. King MD, Humphrey BJ, Wang YF, et al. Emergence of community-acquired methicillin-resistant Staphylococcus aureus USA 300 clone as the predominant cause of skin and soft-tissue infections. Ann Intern Med. 2006;144:309-317.
10. Centers for Disease Control and Prevention. Methicillin-resistant Staphylococcus aureus infections among competitive sports participants—Colorado, Indiana, Pennsylvania, and Los Angeles County, 2000-2003. MMWR. 2003;52:793-795.
11. Lowy FD. Staphylococcus aureus infections. N Engl J Med. 1998;339:520-532.
12. Chambers HF. The changing epidemiology of Staphylococcus aureus? Emerg Infect Dis.
13. Community-associated MRSA information for clinicians. CDC. www.cdc.gov/ncidod/dhqp/ar_ 2008;46(suppl 5): S344-S349. 2006;42:389-391. 2001;7:178-182.
14. Rybak MJ, LaPlante KL. Community-associated methicillin-resistant Staphylococcus aureus; a review. Pharmacotherapy. 2005;25:74-85.
15. Gorwitz RJ, Jernigan DB, Powers JH, et al. Strategies for clinical management of MRSA in the community: summary of an experts’ meeting convened by the Centers for Disease Control and Prevention. March 2006. www.cdc.gov/ncidod/dhqp/pdf/
16. Sabol KE, Echevarria KL, Lewis JS II. Community-associated methicillin-resistant Staphylococcus aureus: new bug, old drugs. Ann Pharmacother. 2006;40:1125-1133.
17. Cleocin (clindamycin) package insert. New York, NY: Pfizer Inc; November 2005.
18. Strom BL, Schinnar R, Apter AJ, et al. Absence of cross-sensitivity between sulfonamide antibiotics and sulfonamide nonantibiotics. N Engl J Med. 2003;349:1628-1635.
19. Nicolau DP, Freeman CD, Nightingale CH, et al. Minocycline versus vancomycin for the treatment of experimental endocarditis caused by oxacillin-resistant Staphylococcus aureus. Antimicrob Agents Chemother. 1994;38:1515-1518.
20. Sakoulas G, Moellering RC Jr. Increasing antibiotic resistance among methicillin-resistant Staphylococcus aureus strains. Clin Infect Dis. 2008;46(suppl 5):S360-S367.
21. Rybak MJ, Akins RL. Emergence of methicillin-resistant Staphylococcus aureus with intermediate glycopeptides resistance: clinical significance and treatment options. Drugs. 201;61:1-7.
22. Tenover FC, Moellering RC Jr. The rationale for revising the Clinical and Laboratory Standards Institute vancomycin minimal inhibitory concentration interpretive criteria for Staphylococcus aureus. Clin Infect Dis. 2007;44:1208-1215.
23. Jeffres MN, Micek ST, Isakow W, et al. Increased incidence of nephrotoxicity with higher vancomycin serum trough concentrations. Program and abstracts of the 46th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC); September 27-30, 2006; San Francisco, CA. Abstract K789.
24. Zyvox (linezolid) package insert. New York, NY: Pfizer Inc; December 2009.
25. Kuter DJ, Tillotson GS. Hematological effects of antimicrobials, a focus on the oxazolidinone, linezolid. Pharmacotherapy. 201;21:1010-1013.
26. Cubicin (daptomycin) package insert. Lexington, MA: Cubist Pharmaceuticals; January 2010.
27. Murthy MH, Olson ME, Wickert RW, et al. Daptomycin non-susceptible methicillin-resistant Staphylococcus aureus USA 300 isolate. J Med Microbiol. 2008;57:1036-1038.
28. Mangili A, Bica I, Snydman R, Hamer DH. Daptomycin-resistant methicillin-resistant Staphylococcus aureus bacteremia. Clin Infect Dis. 2005;40:1058-1060.
29. Synercid (quinupristin-dalfopristin) package insert. Greenville, NC: Monarch Pharmaceuticals; June 2008.
30. Tygacil (tigecycline) package insert. Philadelphia, PA: Wyeth Pharmaceuticals; September 2009.
31. Vibativ (telavancin) package insert. Deerfield, IL: Astellas Pharma US, Inc; September 2009.
32. Stryjewski ME, Chu VH, O’Riordan WD, et al. Telavancin versus standard therapy for the treatment of complicated skin and skin structure infections caused by gram positive bacteria: FAST 2 study. Antimicrob Agents Chemother. 2006;50:862-867.
33. Corey GR. Studies of telavancin in cSSSI. Paper presented at: Third International Symposium on Resistant Gram-Positive Infections; October 10, 2006; Niagara-on-the-Lake, Canada.
34. Vidaillac C, Rybak MJ. Ceftobiprole: first cephalosporin with activity against methicillin-resistant Staphylococcus aureus. Pharmacotherapy. 2009;29:511-525.
35. Discontinuation of the sale of ceftobiprole in Canada. Basilea Pharmaceutica. April 9, 2010. http://hugin.info/134390/R/
36. Vibramycin (doxycycline) package insert. New York, NY: Pfizer Inc; April 2007.
37. Minocin (minocycline) package insert. Philadelphia, PA: Triax Pharmaceuticals; March 2006.
38. Bactrim (trimethoprim-
39. Vancocin (vancomycin) package insert. Exton, PA: ViroPharma Inc; October 2005.
To comment on this article, contact rdavidson@uspharmacist.com.





