US Pharm. 2016;41(11):17-19.
The common denominator between chronic pain, anxiety, trauma, and depression is the nervous system, especially a modified, or dysregulated, nervous system.1 With regard to the elderly, what is significantly predictive of onset of chronic pain later in life is a prior history of anxiety, physical and psychological trauma, and depression.2-5 It has been shown that a relationship exists between stress and the lowering of pain thresholds, based on prospective studies in humans, and substantiated by direct experimental evidence in animals and humans as well.6-10 Overall, what this evidence has suggested is a spectrum ranging from mood and anxiety disorders to painful functional somatic syndromes.11 Additionally, various types of pre-existing anxiety about pain is related to higher pain sensitivities.12,13
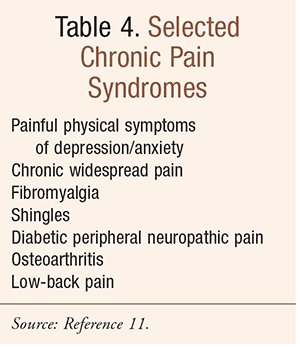
Affective spectrum disorders include mood (e.g., major depressive disorder) and anxiety disorders (TABLES 1, 2). The term functional somatic syndrome is used to describe disorders such as fibromyalgia (TABLE 3) and chronic widespread pain.11 It is important to note that pain is largely considered the same symptom, with the same treatments, whether occurring by itself or as part of any number of chronic pain syndromes, including neuropathic pain (TABLE 4).11
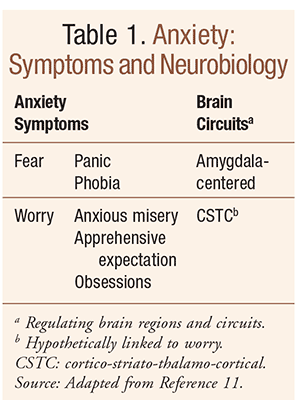
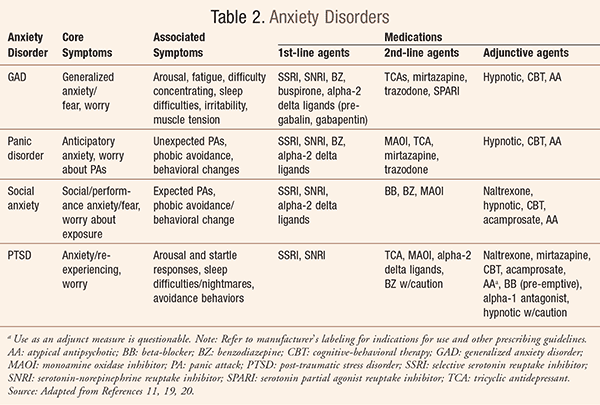
While the core symptoms of major depression (i.e., loss of interest and depressed mood) differ from the core symptoms of anxiety disorders (TABLE 2), there is considerable overlap among the rest of the associated symptoms of these disorders.11 For example, fatigue, sleep difficulties, and problems concentrating are common to both categories of conditions.
Central Sensitization
Many seniors tend to be stoic with regard to withstanding and tolerating pain. Additionally, a fear of becoming dependent on opioids, or meeting healthcare provider resistance to prescribing pain medications in light of the current opioid overdose epidemic in the United States, can often lead older adults to live with chronic pain unnecessarily.14 Data suggest that pain in the elderly is widely under-reognized and undertreated.15,16
Complications of untreated pain include not only an increased risk of depression and anxiety, but also exacerbation of cognitive impairment; functional loss and increased dependency; impaired immune function and healing; impaired mobility and increased risk of falls and/or fractures; increased healthcare utilization and costs; postoperative complications related to immobility (e.g., thrombosis, emboli, pneumonia); sleep disturbances; and withdrawal and decreased socialization.15,16
Despite the significant physical and social consequences, it is estimated that 50% of older adults residing at home and almost 80% of elderly individuals in long-term care facilities continue to suffer from persistent pain.15 Furthermore, pharmacists need to be aware of central sensitization, in which the nervous system is in a persistent state of high reactivity, and pain registers in the brain at a heightened level.1
Central sensitization has two main characteristics involving a heightened sensitivity to pain and the sensation of touch.1 Allodynia is the experience of pain from a trigger normally not painful, such as touch or massage, and hyperalgesia, in which an actual painful stimulus is perceived as more painful than it should be, such as a simple bump that sends an individual with chronic pain “through the roof.”1
Predisposing and Antecedent Factors
There are multiple factors that lead to the development of central sensitization in so-called peripheral chronic pain disorders.1 Predisposing factors include a prior history of depression, anxiety, physical trauma, or psychological trauma. Factors that are associated with the central nervous system following the onset of the original injury or pain condition, referred to as antecedent factors, include depression, anxiety, fear avoidance, or other stressors. These psychophysiologic factors suggest that the pre-existing state of the nervous system is a significant determining factor in the development of central sensitization subsequent to the onset of pain; that is, the already dysregulated nervous system, at the time of injury, for example, may interfere with the normal trajectory of healing and thereby prevent pain from subsiding once tissue damage heals.1
Gray Matter Volume Changes
There are reports that suggest chronic pain may even decrease gray matter in the dorsolateral prefrontal cortex (DLPFC) and thereby contribute to cognitive dysfunction in some pain states, such as fibromyalgia and low-back pain.11 A recent study provides evidence that psychotherapy, one of the least invasive treatments for chronic pain—specifically, cognitive behavioral therapy (CBT)—can produce measurable changes in cerebral gray matter volume and density that correlate with patients’ recovery.17
In CBT, patients are coached to identify and diminish pain catastrophizing, which is a hallmark of chronic pain in which patients anticipate the worst about incoming or ongoing pain; patients are taught relaxation techniques to improve stress and pain control.17,18 The research team suggested that the increase in gray matter reflects an improvement in the modulation of pain, or better mental or emotional assessment of their pain. The structural changes were consistent with the clinical outcomes of CBT; changes in sensory and motor areas potentially represent treatment-induced adaptation to continuous nociceptive signals, the corollary of which is the diversion of attention from pain that is emphasized in CBT.17,18
Conclusion
Evidence suggests pain in the elderly is widely underrecognized and undertreated despite significant mental, emotional, physical, and social consequences. Pharmacists have a significant role to play in advocating for older adults who have stressful conditions that cause pain, such as depression and anxiety, as well as pain—from conditions such as osteoarthritis, fibromyalgia, and shingles—that causes distress. Raising awareness of central sensitization and its characteristic symptoms should help pharmacists better guide patients in the early treatment of underlying conditions that may lead to chronic pain and central sensitization and avoid progression within the nervous system to a persistent state of high reactivity registering pain at a heightened level.
REFERENCES
1. McAllister MJ. Central sensitization. Updated September 8, 2016. www.instituteforchronicpain.org/understanding-chronic-pain/what-is-chronic-pain/central-sensitization. Accessed October 14, 2016.
2. Nahit ES, Hunt IM, Lunt M, et al. Effects of psychosocial and individual psychological factors on the onset of musculoskeletal pain: common and site-specific effects. Ann Rheum Dis. 2003;62:755-760.
3. Talbot NL, Chapman B, Conwell Y, et al. Childhood sexual abuse is associated with physical illness burden and functioning in psychiatric patients 50 years of age or older. Psychosom Med. 2009;71:417-422.
4. McLean SA, Clauw DJ, Abelson JL, et al. The development of persistent pain and psychological morbidity after motor vehicle collision: integrating the potential role of stress response systems into a biopsychosocial model. Psychosom Med. 2005;67:783-790.
5. Hauser W, Galek A, Erbsloh-Moller B, et al. Posttraumatic stress disorder in fibromyalgia syndrome: prevalence, temporal relationship between posttraumatic stress and fibromyalgia symptoms and impact on clinical outcome. Pain. 2013;154:1216-1223.
6. Slade GD, Diatchenko L, Bhalang K, et al. Influence of psychological factors on risk of temporomandibular disorders. J Dent Res. 2007;86:1120-1125.
7. Alexander J, DeVries A, Kigerl K, et al. Stress exacerbates neuropathic pain via glucocorticoid and NMDA receptor activation. Brain Behav Immun. 2009;23:851-860.
8. Imbe H, Iwai-Liao Y, Senba E. Stress-induced hyperalgesia: animal models and putative mechanisms. Front Biosci. 2006;11:2179-2192.
9. Kuehl LK, Michaux GP, Richter S, et al. Increased basal mechanical sensitivity but decreased perceptual wind-up in a human model of relative hypocortisolism. Pain. 2010;194:539-546.
10. Rivat C, Becker C, Blugeot A, et al. Chronic stress induces transient spinal neuroinflammation, triggering sensory hypersensitivity and long-lasting anxiety-induced hyperalgesia. Pain. 2010;150:358-368.
11. Stahl SM. Stahl’s Essential Psychopharmacology: Neuroscientific Basis and Practical Applications. 4th ed. New York: Cambridge University Press; 2013:388-443.
12. Hirsh AT, George SZ, Bialosky JE, Robinson ME. Fear of pain, pain catastrophizing, and acute pain perception: relative prediction and timing of assessment. J Pain. 2008;9(9):806-812.
13. Sullivan MJ. Thorn B, Rodgers W, Ward LC. Path model of psychological antecedents to pain experience: experimental and clinical findings. Clin J Pain. 2004;20(3):164-173.
14. CDC. Injury prevention & control: opioid overdose. Updated: October 17, 2016. www.cdc.gov/drugoverdose/. Accessed October 17, 2016.
15. American Geriatric Society Panel on Persistent Pain in Older Persons. The management of persistent pain in older persons. J Am Geriatr Soc. 2002;50(6):S205-S224.
16. Weiner DK, Rudy TE, Gaur S. Are all older adults with persistent pain created equal? Preliminary evidence for a multiaxial taxonomy. Pain Res Manag. 2001;6(3):133-141.
17. Seminowicz DA, Shpaner M, Keaser ML, et al. Cognitive-behavioral therapy increases prefrontal cortex gray matter in patients with chronic pain. J Pain. 2013;14(12):1573-1584.
18. Abdul-Kareem Ahmed. Cognitive behavioral therapy changes gray matter morphology in chronic pain: Increase in gray matter correlates with decrease in catastrophizing. November 25, 2013. www.painresearchforum.org/news/34218-cognitive-behavioral-therapy-changes-gray-matter-morphology-chronic-pain. Accessed October 17, 2016.
19. Epocrates.com. Epocrates Plus Version 15.12.1. Updated June 23, 2016. Accessed September 20, 2016.
20. Semla TP, Beizer JL, Higbee MD. Geriatric Dosage Handbook. 20th ed. Hudson, OH: Lexicomp; 2015.
To comment on this article, contact rdavidson@uspharmacist.com.