US Pharm. 2018;43(9):27-29.
Type 1 or type 2 diabetes mellitus is a condition that may result from genetic factors or a sedentary lifestyle, respectively. Signs and symptoms may include an increase in urination, often at night (polyuria); increased thirst (polydipsia); increased hunger (polyphagia); blurred vision; and undefined weight loss.1 When uncontrolled, diabetes may be associated with life-threatening outcomes, including damage to the blood vessels, nerves, eyes, kidneys, and heart.1 Additionally, uncontrolled diabetes may lead to diabetic ketoacidosis and, if left untreated, coma or death.2 Therefore, to mitigate sequelae, self-monitoring may serve as a useful tool for disease management. Further, ensuring that patients are properly educated is vital when attempting to adequately control their blood glucose or alter drug therapy.3
Epidemiology
According to the American Diabetes Association (ADA), diabetes remains the seventh leading cause of death in the United States. An estimated 30.3 million people (9.4% of the U.S. population) were diagnosed with diabetes in 2015. In fact, the prevalence of adults with diabetes increased among those aged >65 years.4
Diagnosis
Glycosylated hemoglobin (A1C), an oral glucose tolerance test, and random or fasting blood glucose are assays used to diagnose diabetes. Use of the aforementioned examinations may be explained as follows5:
A1C: A1C measures the percentage of blood glucose attached to hemoglobin and indicates the average blood glucose level over 2 to 3 months. ADA guidelines affirm that diabetes should be diagnosed if the patient’s A1C is greater than 6.5%.
Glucose Tolerance: Patients ingest 75 g of a sugar-containing drink, and blood glucose levels are measured after 2 hours. Levels greater than 200 mg/dl are consistent with the diagnosis of diabetes.
Random/Fasting: A random blood glucose level of 200 mg/dL suggests diabetes. Additionally, after 8 hours of fasting, diabetes is also confirmed if blood glucose is >126 mg/dL or higher on two separate occasions.
Although these tests have been employed to diagnose and monitor diabetes, a more recent device has been approved to assist in maintaining adequate control of the condition.
Device
Approved in March 2018, Guardian Connect is a continuous glucose-monitoring (CGM) system that measures glucose levels every 5 minutes and displays the values on the Guardian Connect app (Sugar.IQ Personal Assistant). The app can be installed on a compatible mobile device (i.e., an iPhone or iPad). As recommended by the manufacturer, Guardian Connect allows users aged >14 years to detect trends and track patterns in an effort to maintain normal glucose concentrations.6 An image of the Guardian Connect and patient instructions may be found in FIGURE 1 and TABLE 1, respectively.7,8

Efficacy
A clinical study was conducted to evaluate the accuracy and performance of the Guardian Connect in the abdomen and arm. Eighty-eight diabetic participants from age 14 to 75 years were included in the study. Participants wore two sensors on the abdomen that paired with either a MiniMed 640G insulin pump or an iPhone or iPod Touch linked to the Sugar.IQ Personal Assistant app. A third sensor was connected to a glucose recorder. Participants were randomized into two groups. Group A test values were recorded on Days 1, 3, and 7 at 50 and 146 hours after sensor insertion; Group B test values were recorded on Day 1, 14 hours after sensor insertion, and on Days 3 and 7 at 62 and 158 hours after sensor insertion, respectively.
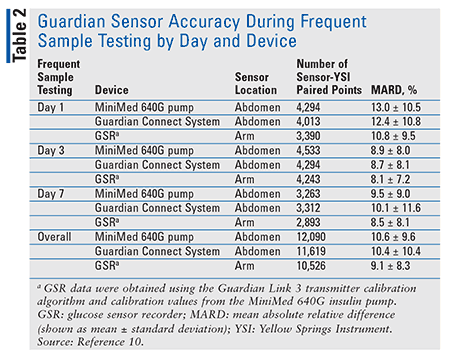
Participants with established insulin sensitivity and insulin-carbohydrate ratios experienced challenges of raising blood glucose levels to a target of 180 to 400 mg/dL for 2 hours and lowering blood glucose levels to a target of 50 to 75 mg/dL for 1 hour. Results demonstrated that the Guardian Connect, whether located in the abdomen or the arm, provided accurate glucose readings when compared with the Yellow Springs glucose monitor, which is commonly used to analyze the sensor’s accuracy (TABLE 2).9
Conclusion
The Guardian Connect CGM system was developed to help effectively manage patients with type 1 or type 2 diabetes. However, the device should not be used in diabetic patients who are unable to perform a minimum of two blood glucose tests per day, see or hear the alert sounds generated by the system, or stay in contact with their healthcare professional.10
REFERENCES
1. Centers for Disease Control and Prevention. Diabetes symptoms. www.cdc.gov/diabetes/basics/symptoms.html. Accessed July 9, 2018.
2. Mayo Clinic. Diabetic ketoacidosis. www.mayoclinic.org/diseases-conditions/diabetic-ketoacidosis/symptoms-causes/syc-20371551. Accessed July 9, 2018.
3. Kirk JK, Stegner J. Self-monitoring of blood glucose: practical aspects. J Diabetes Sci Technol. 2010;4(2):435-439.
4. American Diabetes Association. Statistics about diabetes. www.diabetes.org/diabetes-basics/statistics. Accessed July 10, 2018.
5. American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2009;32(suppl 1):S62-S67.
6. Guardian Connect System User Guide. Medtronic Diabetes. www.medtronicdiabetes.com/sites/default/files/library/download-library/user-guides/GuardianT%20Connect%20CGM%20System%20User%20Guide.PDF. Accessed August 2, 2018.
7. Integrated Diabetes Services. Choosing a CGM: 3 heads are better than one. http://integrateddiabetes.com/choosing-a-cgm-3-heads-are-better-than-one. Accessed August 15, 2018.
8. Medtronic. Meet your personal diabetes assistant. www.medtronicdiabetes.com/products/sugar.iq-diabetes-assistant. Accessed July 10, 2018.
9. Christiansen MP, Garg SK, Brazg R, et al. Accuracy of a fourth-generation subcutaneous continuous glucose sensor. Diabetes Technol Ther. 2017;19(8):446-456.
10. U.S. Food and Drug Administration. Guardian Connect System—P160007. www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/DeviceApprovalsandClearances/Recently-ApprovedDevices/ucm604253.htm. Accessed July 10, 2018.
To comment on this article, contact rdavidson@uspharmacist.com.





