US Pharm. 2008;33(11):Epub.
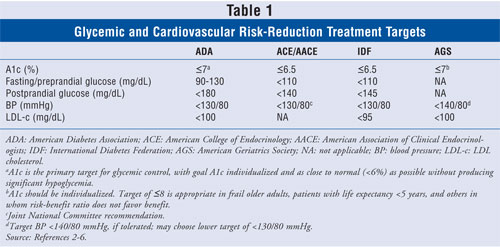
Type 2 diabetes mellitus (DM2) results from the combination of an insulin-secretory defect and peripheral insulin resistance. It is estimated to affect at least 7% of the U.S. population, with more than 20.9% of elderly adults having DM2.1 Despite the availability of numerous national and international clinical-practice guidelines recommending aggressive glycemic control and cardiovascular risk reduction (TABLE 1) to decrease morbidity and mortality, surveys suggest that only 34% to 42% of patients achieve a hemoglobin A1c <7%.2-6 Likewise, only 34% to 40% and 22.7%, respectively, of patients achieve an LDL-cholesterol (LDL-c) level <100 mg/dL or blood pressure (BP) <130/80 mmHg.7-9 Simultaneous control of A1c, LDL, and BP is uncommon.8,9
Case Presentation
J.K., a 72-year old male who has had DM2 for 10 years but is without DM complications, is scheduled to see his pharmacist today for medication-management assistance. His other medical diagnoses include hypertension and dyslipidemia, both of which have been controlled except for a recent elevation in triglycerides (TG). He does not smoke, and he drinks alcohol rarely. He has not suffered any falls. His Mini-Mental Status Examination score is 28 (normal: 24 or more). Clinic point-of-care A1c is 8.6%. Postclinic labs include glucose, 213 mg/dL; TG, 468 mg/dL; HDL cholesterol (HDL-c), 35 mg/dL; total cholesterol, 162 mg/dL; and serum creatinine, 1.2 mg/dL. On physical examination, his body-mass index (BMI), sitting BP, standing BP, and heart rate are, respectively, 30.5 kg/m2, 128/76 mmHg sitting, 126/72 mmHg standing, and 72 beats per minute. J.K.'s social history reveals that he is a retired salesman. He received diabetes education when he was initially diagnosed and believes that he is adherent to lifestyle measures. He declares, "I always skip the cake at family birthday parties." Yesterday J.K. consumed a whole-wheat bagel (regular size), 8 oz. orange juice, 4 oz. "lite" low-fat yogurt, two slices of pork sausage, and pasteurized egg product (equal to two eggs) for breakfast; a piece of vegetable lasagna, 8 oz. apple juice, and a slice of garlic bread for lunch; a salad, 8 oz. steak, and a potato for dinner; and apple slices with peanut butter for a bedtime snack. He cannot match foods according to composition; he remembers learning the Idaho Plate Method, but currently does not follow it. Consultation with the primary care provider (PCP) documents a medication-reconciliation list that includes pioglitazone 45 mg daily; metformin 1,000 mg twice daily; glyburide 10 mg twice daily; insulin glargine 50 units at bedtime; atorvastatin 80 mg at bedtime; aspirin 81 mg daily; OTC fish oil 1,000 mg three times daily; and lisinopril 20 mg daily. J.K. has been taking glyburide for nine years and insulin glargine for two years. He complains about his monthly pharmacy expenses. His self-monitoring blood glucose (SMBG) diary documents BG levels of 106 mg/dL to 125 mg/dL fasting; 150 mg/dL to 224 mg/dL and 264 mg/dL to 307 mg/dL premeal at lunch and supper, respectively; and occasional bedtime results >330 mg/dL. In general, he has no issues with hypoglycemia, but he develops morning hypoglycemia if he skips breakfast. His meal schedule tends to be erratic due to his daily activities. J.K. says that he is motivated to do what is necessary to achieve better glycemic control so that he is "there" for his great-grandchildren.
Despite J.K.'s classification as an "older person" with diabetes, according to the American Geriatrics Society, he does not require less stringent treatment goals.5 He is ambulatory, he shows no cognitive decline, and his life expectancy likely exceeds five years (i.e., he lacks diabetic complications or other comorbidities and does not smoke). His TG are elevated secondary to poor glycemic control; urgent intervention is unnecessary, however, as the value is below the treatment threshold of 500 mg/dL. J.K.'s non-HDL-c level of 127 mg/dL is less than his non-HDL goal of <130 mg/dL. He does not require adjunctive antilipemic pharmacotherapy.
In terms of medication self-management, J.K.'s personal medication-reconciliation list reveals that he has self-decreased his glyburide to once daily. No other medication discrepancies exist between his medication list and that submitted by his PCP. J.K.'s patient diary notes the date of glyburide dose reduction. Review of his glucometer download indicates no worsening in overall glycemic control since glyburide was modified. J.K.'s endocrinologist concurs that his action is clinically appropriate, as the failure rate of glyburide after five years of treatment is 34%.10
Due to treatment failure, J.K. wonders whether he can try Januvia (sitagliptin), so he is educated as to sitagliptin's effectiveness, mechanism of action, and current role in therapy; one of the published insulin algorithms also is reviewed with him.11 He agrees to the next phase of insulin treatment and chooses intensive insulin, as he needs mealtime flexibility and has "always been good with numbers." If J.K. had scheduled meals, he also would have been a candidate for multidose insulin therapy with aspart and NPH insulin.
J.K. requires medical nutrition therapy to potentially include advanced carbohydrate counting to achieve preprandial and postprandial BG targets if he is to obtain a degree of success with basal-bolus correction insulin. Currently, he cannot identify which foods contain carbohydrates. He believes that his blood sugar will be significantly worsened by a slice of cake but is unconcerned about consuming a bagel, yogurt, and orange juice in the same meal. J.K.'s diet contains large quantities of carbohydrates, which contribute to his nonfasting hyperglycemia; likewise, his diet is high in calories from fats and proteins, thereby contributing to his obesity (BMI >30 kg/m2) and BG variability.
J.K.'s A1c exceeds 8.5%, and his SMBG documents a progressive rise during the day, with significant correction by morning. It is not appropriate to continue titration of basal insulin to target fasting BG values to <110 mg/dL due to the late-morning hypoglycemia J.K. experiences when he does not eat breakfast. This finding suggests overinsulinization and the need for basal insulin dose reduction.12 Additionally, as his daytime control is poor, the insulin secretagogue should be replaced with bolus insulin.13 When converting to basal-bolus correction insulin in the presence of overinsulinization, it may be appropriate to reduce the total daily dose (TDD) to 80% of the former basal insulin dose.12 The TDD should be physiologically divided between basal (50%) and bolus insulin (50%).14 Metformin should be maintained, as it may limit weight gain.15,16 Despite the limitations of a recent meta-analysis suggesting that rosiglitazone may be associated with myocardial infarction, the endocrinologist elects to withdraw pioglitazone.17
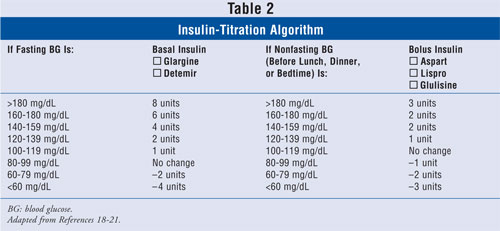
As it is unlikely that insulin glargine 20 units daily and aspart 6 units at each of the three major meals will achieve J.K.'s glycemic targets, insulin needs to be titrated over several weeks. The pharmacist instructs J.K. in the use of the clinic's insulin algorithm, which is designed for patient use (TABLE 2).18-21
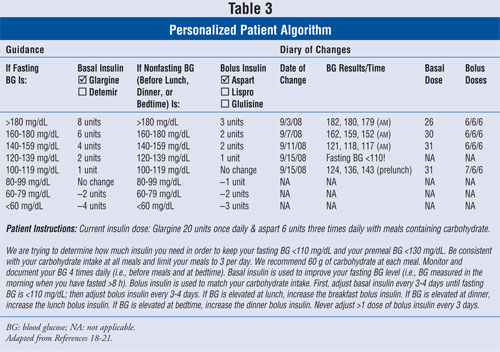
At home, J.K. focuses on maintaining an intake of 60 g of carbohydrate at each meal, documenting his SMBG, and titrating the individual insulins at three- to four-day intervals according to his personalized algorithm (TABLE 3). Once he achieves a fasting BG <110 mg/dL with basal insulin titration, he begins adjusting his bolus insulin doses. For example, between September 1, 2008 and September 3, 2008, J.K. has two fasting BGs 180 mg/dL or lower and one value >180 mg/dL; he therefore increases his glargine by 6 units, to 26 units daily. He continues basal insulin titration every four days until his fasting BG is <110 mg/dL. Then, on September 15, he increases his breakfast bolus insulin dose by 1 unit since two prelunch BGs are between 120 and 139 mg/dL.
Commentary
Individualization of Treatment Goals: The goal of treatment is euglycemia. Generally, the goal of ≤6.5% should be targeted by achieving a fasting BG <110 mg/dL and a postprandial BG <140 to 180 mg/dL.2-4 In routine care, the patient monitors fasting and preprandial BG. If the A1c remains above target, BG measured 90 to 120 minutes after a meal may be evaluated for results exceeding the postprandial treatment goal.
Management of Hypertriglyceridemia: When diabetes becomes uncontrolled, TG may increase. When TG are elevated >400 mg/dL, an LDL-c calculation is not possible. Instead, the clinician should complete a non-HDL calculation to assess for atherogenic remnant lipoproteins and the need for additional LDL-c-reducing pharmacotherapy. The non-HDL-c is calculated by subtracting the HDL-c value from the total cholesterol value. The target non-HDL-c is 30 mg/dL higher than the usual LDL-c target.22
Baseline Treatment Concordance: The patient is integral to the success or failure of treatment. Concordance is a therapeutic alliance or partnership between the patient and the physician with regard to whether, when, and how the patient takes medications (and performs SMBG). Concordance requires open communication between the provider(s) and the patient. The patient must be well educated about the diagnosis, treatment, and risk-benefit ratio of treatment options and about self-management skills, and the patient's opinion must be respected. Concordance and compliance are two different things; concordance denotes patient empowerment, whereas compliance implies patient obedience.23
When to Use a Treatment Algorithm: The average visit duration for diabetes-related office-based services is 19.3 +/- 8.2 minutes.24 Despite sufficient visit length, failure to intensify medication management during appointments--termed clinical inertia--often occurs.9,25 A survey of patient records from U.S. academic medical centers found that fewer than 50% of eligible patients had therapy intensified.9 Survey data further suggested that a moderate A1c elevation is necessary before action is taken, whereas any A1c result exceeding treatment goal should prompt immediate action.9 Intensification for patients on monotherapy or less-than-maximum doses of dual oral agents is uncomplicated. First- and second-line oral drug selection is based on effectiveness in lowering BG, perceived extraglycemic effects, safety profiles, tolerability, and expense.13 Drug selection after dual therapy is less clear; treatment algorithms, which provide guidance according to ambient BG control, may facilitate intensification, enable rapid interval follow-up, and limit inappropriate polypharmacy.2,11,13,26,27
Treatment Burden: Surveys have documented that patients perceive insulin regimens as highly burdensome and that the burden is associated with emotional distress.28,29 Treatment burden may be attenuated by experience with insulin administration, but this finding may be limited to patients who receive less intensive insulin regimens or have less rigorous SMBG requirements.28 When an intensive insulin regimen is necessary, management in which insulin is matched to carbohydrate intake on a meal-by-meal basis may improve quality of life.30
Individualized Medical Nutrition Therapy: Many patients identify nutrition as their greatest challenge. Counting carbohydrates may be useful for patients who are frustrated with structured meal planning, eat variable amounts of carbohydrates from meal to meal, overeat carbohydrates, or are transitioning to basal-bolus correction insulin. The quantity of carbohydrate, rather than whether it is a simple or a complex sugar--i.e., a carbohydrate is a carbohydrate--may be the most important factor in determining postprandial glucose excursion and bolus insulin requirements.31 Preferential consumption of low-glycemic-index (GI) rather than high-GI carbohydrates on a gram-per-gram basis may afford an additional modest reduction in A1c for the motivated patient.32
Patients with diabetes do not have to avoid simple sugars. The emphasis on quantity of carbohydrate does not mean that patients should indulge in simple sugars such as desserts. Healthy carbohydrates occasionally may be replaced with dessert in meal plans using the principle of carbohydrate counting. Carbohydrates are measured in grams; one carbohydrate serving is 15 g.33 Since carbohydrate intake should not be less than 40% of total calories, total carbohydrate intake should be 130 g or more per day. For obese patients with diabetes, recommended total daily carbohydrate intake ranges from 130 g to 200 g for 1,200- to 2,000-calorie diets.34 Total grams are divided between three daily meals , with the usual intake ranging from 45 g to 60 g of carbohydrate per meal. The initial target for carbohydrate intake is determined after a review of the patient's usual meal planning and through negotiation.31 Since fiber is an unabsorbed carbohydrate, it should not be included in grams of carbohydrate calculation when the meal contains 5 g or more.33
Comprehensive carbohydrate-counting instruction may require up to nine office visits with a registered dietician over a period of five to 24 months. All instructional levels stress the importance of portion control, and patients must achieve the requisite skill before moving to the next level of instruction. Throughout the instructional and follow-up phases, patients are encouraged to keep nutrition logs that include weighed or measured amounts of carbohydrate.23 Basic and intermediate instruction teaches the patient about which foods contain carbohydrates; carbohydrate choices from the food guide pyramid; how to count grams using the Nutrition Facts panel (i.e., reading labels); the effect of carbohydrates, medications, and exercise on BG values; interpretation of BG results; and appropriate action (e.g., medication dose adjustment). Advanced instruction requires the patient to have good math and problem-solving skills in order to match planned carbohydrate intake to bolus insulin doses using carbohydrate-to-insulin ratios. A carbohydrate-to-insulin ratio in g per unit is calculated by dividing grams of carbohydrate by the number of units of bolus insulin necessary to meet postprandial BG goals.31 For example, a patient who uses 10 units of aspart insulin to achieve a postprandial BG <180 mg/dL when consuming 60 g of carbohydrate has a ratio of 1 unit of insulin to 6 g of carbohydrate, or 1:6. If the patient eats more or fewer carbohydrates in a meal, the units of rapid-acting insulin can be adjusted up or down to prevent hyperglycemia or hypoglycemia.
Analyses of the effectiveness of carbohydrate counting in association with intensive insulin management have been conducted in patients with type 1 DM (DM1).30,35 The Dose Adjustment for Normal Eating trial demonstrated that a five-day outpatient-skills course that provided advanced carbohydrate counting and intensive insulin instruction led to improvements in A1c, dietary freedom, and quality of life.30 These results were significant despite an increased treatment burden (mean number of daily insulin injections increased from 3.6 at baseline to 5.3 at endpoint).30 The Dusseldorf Diabetes Treatment and Teaching Programme documented an improvement in glycemic control in patients with DM1 without an increase in risk of hypoglycemia when a flexible diet and intensive insulin were used.35 The benefit of medical nutrition therapy likely can be extrapolated to patients with DM2 who are on similar treatment regimens.
Management of Intensive Insulin: Physiologic insulin-replacement regimens are designed to mimic normal beta-cell function and consist of basal and bolus insulin. Basal insulin, also called background or augmentation insulin, is provided most effectively by peakless insulin analogs such as glargine and detemir. Peakless basal insulin provides a constant insulin level to allow flexibility in scheduling or skipping meals. Bolus insulin, also termed prandial, mealtime, or nutrient-related insulin, matches the physiologic short-term insulin response to carbohydrate intake; rapid-acting insulin analogs such as lispro, aspart, and glulisine are most effective. Due to their respective durations of action, NPH and regular insulin function independently as basal and bolus insulin, necessitating strict scheduling of meals to limit hypoglycemia. Correction-dose insulin, also known as supplemental insulin, is an adjunct dose applied to scheduled bolus insulin. Appropriate use requires the patient to perform SMBG to determine whether preprandial hyperglycemia is present. Initial correction doses may range from 1 unit for every 50 mg/dL to 1 unit for every 30 mg/dL for BG exceeding 130 mg/dL. If correction doses are frequently required, scheduled insulin needs to be adjusted.36
There is no maximum dose of insulin beyond which effect will not occur.13 All insulin modification should be considered as a trial dose requiring the patient to conduct SMBG to establish effect.4 For patients already receiving basal insulin and failing therapy, bolus insulin may be added. When converting from basal to physiologic insulin (basal-bolus correction), the initial TDD should remain constant and follow-up SMBG should be utilized to determine appropriate insulin titration.14
Literature supports the use of physiologic insulin replacement with insulin analogs in DM2.37-39 A 26-week, multinational, open-label, parallel-design clinical trial evaluated 505 patients receiving either detemir or NPH insulin in combination with aspart insulin.37 Background therapy with detemir was noninferior to NPH with regard to glycemic control and was associated with less weight gain.37 A 12-month, randomized, controlled clinical trial of 107 patients compared continuous subcutaneous insulin infusion of lispro with multiple daily injections (MDI) of glargine and lispro.38 Glycemic control was comparable for the two strategies, and 84% of MDI patients achieved an A1c <7%.38 An 18-month, nonrandomized, single-center study of 119 patients enrolled in an inpatient teaching program conducted by the German Diabetes Association assessed two MDI strategies of rapid-acting insulin analog (lispro or aspart) in combination with either NPH or glargine in patients with fair glycemic control at baseline (mean A1c 7.4%).39 This study demonstrated that glargine was associated with significantly less weight gain, lower rates of hypoglycemia, and a higher proportion of patients achieving an A1c <7%.39
Use of an Insulin Algorithm: Insulin titration may be performed by the provider or by the patient. In a study evaluating physician and patient algorithmic titration of insulin, it was determined that the risk of severe hypoglycemia associated with patient titration occurs at a rate of 2.36 events per 100 patient-years and does not differ significantly from rates associated with physician titration.18 Despite an increase in mild hypoglycemia, glycemic control improves with patient titration.18 Using an insulin algorithm, patients are able to titrate individual insulin doses at intervals of three to seven days. If two of three readings at a specified time on three consecutive days are not within the target, the insulin dose should be adjusted according to the lowest of the BG results.18-21 Traditionally, basal insulin is titrated until fasting BG is <110 mg/day, and then the focus shifts to bolus insulin.34 Alternatively, titration can rotate between basal and bolus insulin adjustment (i.e., four insulin-titration opportunities over a 12-day period). Balancing of basal and bolus insulin may prevent basal or bolus overinsulinization and more effectively improve glycemic control.14 The Opposing Step-By-Step Insulin Reinforcement to Intensified Strategy Study is currently enrolling patients to investigate the best stepwise approach to introducing bolus insulin.40
Conclusions
The increasing prevalence of diabetes, coupled with the complexity of its management, renders it critical for pharmacists to increase their knowledge and skills to better care for their patients. DM2 is a progressive disease that is associated with oral treatment failure over time. Available algorithms are useful for determining appropriate pharmacotherapy and intensification of insulin. Therapy must be patient-centered and therefore take into account cultural traditions, personal preferences, social situations, and lifestyle. Treatment goals should be individualized. Patients must be empowered to perform appropriate self-care, as they are living with a disease that requires constant self-management. Patients must receive initial and interval education, including management of diet and medication, so that they are equipped with the appropriate tools to be successful with physiologic insulin replacement.
References
1. National Institute of Diabetes and Digestive and Kidney Diseases. National Diabetes Statistics Fact Sheet: General Information and National Estimates on Diabetes in the United States, 2005. NIH Publication No. 06-3892. Bethesda, MD: US Department of Health and Human Services, National Institute of Health, 2005.
2. American Diabetes Association. Standards of medical care in diabetes--2007. Diabetes Care. 2007;30:S4-S41.
3. The American Association of Clinical Endocrinologists medical guidelines for the management of diabetes mellitus: the AACE system of intensive diabetes self-management--2002 update. Endocr Pract. 2002;8(suppl 1):40-82.
4. International Diabetes Federation. Global guideline for type 2 diabetes. 2005. www.idf.org/webdata/docs/IDF%
5. California Healthcare Foundation/American Geriatrics Society Panel on Improving Care for Elders with Diabetes. Guidelines for improving care of the older person with diabetes mellitus. J Am Geriatr Soc. 2003;51(suppl 5):S265-S280.
6. The sixth report of the Joint National Committee on prevention, detection, evaluation, and treatment of high blood pressure. Arch Intern Med. 1997;157:2413-2446.
7. Saaddine JB, Cadwell B, Gregg EW, et al. Improvements in diabetes processes of care and intermediate outcomes: United States, 1988-2002. Ann Intern Med. 2006;144:465-474.
8. Jackson GL, Edelman D, Weinberger M. Simultaneous control of intermediate diabetes outcomes among Veterans Affairs primary care patients. J Gen Intern Med. 2006;21:1050-1056.
9. Grant RW, Buse JB, Meigs JB, for the University HealthSystem Consortium (UHC) Diabetes Benchmarking Project Team. Quality of diabetes care in U.S. academic medical centers: low rates of medical regimen change. Diabetes Care. 2005;28:337-442.
10. Kahn SE, Haffner SM, Heise MA, et al, for the ADOPT Study Group. Glycemic durability of rosiglitazone, metformin, or glyburide monotherapy. New Engl J Med. 2006;355:2427-2443.
11. Texas Department of State Health Services. Texas Diabetes Council. Insulin algorithm for type 2 diabetes in children and adults. Publication #45-11647. www.dshs.state.tx.us/diabetes/
12. Braithwaite SS. Case study: five steps to freedom: dose titration for type 2 diabetes using basal-prandial-correction insulin therapy. Clin Diabetes. 2005;23:39-43.
13. Nathan DM, Buse JB, Davidson MB, et al. Management of hyperglycemia in type 2 diabetes: a consensus algorithm for the initiation and adjustment of therapy: a consensus statement from the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care. 2006;29:1963-1972.
14. Yokoyama H, Tada J, Kamikawa F, et al. Efficacy of conversion from bedtime NPH insulin to morning insulin glargine in type 2 diabetic patients on basal-prandial insulin therapy. Diabetes Res Clin Pract. 2006;73:35-40.
15. Yki-Järvinen H, Ryysy L, Nkilä K, et al. Comparison of bedtime insulin regimens in patients with diabetes mellitus. A randomized, controlled trial. Ann Intern Med. 1999;130:389-396.
16. Wulffelé MG, Kooy A, Lehert P, et. al. Combination of insulin and metformin in the treatment of type 2 diabetes. Diabetes Care. 2002;25:2133-2140.
17. Nissen SE, Wolski K. Effect of rosiglitazone on the risk of myocardial infarction and death from cardiovascular causes. N Engl J Med. 2007;356:2457-2471.
18. Davies M, Storms F, Shutler S, et al, for the ATLANTUS Study Group. Improvement of glycemic control in subjects with poorly controlled type 2 diabetes: comparison of two treatment algorithms using insulin glargine. Diabetes Care. 2005;28:1282-1288.
19. Raskin P, Allen E, Hollander P, et al, for the INITIATE Study Group. Initiating insulin therapy in type 2 diabetes: a comparison of biphasic and basal insulin analogs. Diabetes Care. 2005;28:260-265.
20. Janka HU, Plewe G, Riddle MC, et al. Comparison of basal insulin added to oral agents versus twice-daily premixed insulin as initial insulin therapy for type 2 diabetes. Diabetes Care. 2005;28:254-259.
21. Kennedy L, Herman WH, Strange P, et al, for the GOAL A1C Team. Impact of active versus usual algorithmic titration of basal insulin and point-of-care versus laboratory measurement of the HbA1c on glycemic control in patients with type 2 diabetes: the Glycemic Optimization with Algorithms and Labs at Point of Care (GOAL A1c) trial. Diabetes Care. 2006;29:1-8.
22. Third Report of the National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, and Treatment of High Blood Cholesterol in Adults (Adult Treatment Panel III) final report. Circulation. 2002;106:3143.
23. Dickinson D, Wilkie P, Harris M et al. Taking medicines: concordance is not compliance [letter]. BMJ. 1999;319:787.
24. Grant RW, Pirraglia PA, Meigs JB, Singer DE. Trends in complexity of diabetes care in the United States from 1991 to 2000. Arch Intern Med. 2004;164:1134-1139.
25. Shah BR, Hux JE, Laupacis A, et al. Clinical inertia in response to inadequate glycemic control: do specialists differ from primary care physicians? Diabetes Care. 2005;28:600-606.
26. Campbell RK, White JR. Insulin therapy in type 2 diabetes. J Am Pharm Assoc (Wash). 2002; 42:602-611.
27. Texas Department of State Health Services. Glycemic control algorithm for type 2 diabetes in children and adults. Publication #45-11265. www.dshs.state.tx.us/diabetes/
28. Vijan S, Hayward RA, Ronis DL, Hofer TP. Brief report: the burden of diabetes therapy: implications for the design of effective patient-centered treatment regimens. J Gen Intern Med. 2005;20:479-482.
29. Delahanty LM, Grant RW, Wittenberg E, et al. Association of diabetes-related emotional distress with diabetes treatment in primary care patients with Type 2 diabetes. Diabet Med. 2007;24:48-54.
30. DAFNE Study Group. Training in flexible, intensive insulin management to enable dietary freedom in people with type 1 diabetes: dose adjustment for normal eating (DAFNE) randomised controlled trial. BMJ. 2002;325:746-751.
31. Gillespie S, Kulkarni K, Daly A. Using carbohydrate counting in diabetes clinical practice. J Am Diet Assoc. 1998;98:897-905.
32. Brand-Miller J, Hayne S, Petocz P, Colagiuri S, et al. Low-glycemic index diets in the management of diabetes: a meta-analysis of randomized controlled trials. Diabetes Care. 2003;26:2261-2267.
33. Kulkarni KD. Carbohydrate counting: a practical meal-planning option for people with diabetes. Clin Diabetes. 2005;23:120-122.
34. Joslin Diabetes Center & Joslin Clinic. Clinical nutrition guideline for overweight and obese adults with type 2 diabetes, prediabetes, or those at high risk for developing type 2 diabetes. March 29, 2007. http://professionaled.joslin.
35. Sämann A, Mühlhauser I, Bender R, et al. Glycaemic control and severe hypoglycaemia following training in flexible, intensive insulin therapy to enable dietary freedom in people with type 1 diabetes: a prospective implementation study. Diabetologia. 2005;48:1965-1970.
36. DeWitt DE, Hirsch IB. Outpatient insulin therapy in type 1 and type 2 diabetes: scientific review. JAMA. 2003:289:2254-2264.
37. Haak T, Tiengo A, Draeger E, et al. Lower within-subject variability of fasting blood glucose and reduced weight gain with insulin detemir compared to NPH insulin in patients with type 2 diabetes. Diabetes Obes Metab. 2005;7:56-64.
38. Herman WH, Ilag LL, Johnson SL, et al. A clinical trial of continuous subcutaneous insulin infusion versus multiple daily injections in older adults with type 2 diabetes. Diabetes Care. 2005;28:1568-1573.
39. Siegmund T, Weber S, Blankenfeld H, et al. Comparison of insulin glargine versus NPH insulin in people with type 2 diabetes mellitus under outpatient-clinic conditions for 18 months using a basal-bolus regimen with a rapid-acting analogue as meal-time insulin. Exp Clin Endocrinol Diabetes. 2007;115:349-353.
40. ClinicalTrials.gov. OSIRIS: Opposing Step-by-Step Insulin Reinforcement to Intensified Strategy. www.clinicaltrials.gov/ct/
To comment on this article, contact rdavidson@jobson.com.





