US Pharm. 2016;41(4):39-42.
ABSTRACT: Legionnaires’ disease (LD) is a severe respiratory infection caused by the Legionella pneumophila bacterium, which was first identified by the CDC in 1976. LD is classified as an atypical type of community-acquired pneumonia, since exposure is through inhalation or aspiration of contaminated aerosolized water. The CDC reports that between 5% and 30% of patients with LD die each year. Infected individuals usually require hospitalization and may be treated with a number of antibiotics including azithromycin, levofloxacin, or doxycycline. LD may also manifest as a more benign, acute febrile illness known as Pontiac fever, which usually resolves on its own without hospitalization or pharmacotherapy.
Legionnaires’ disease (LD) is the term that collectively describes respiratory infections caused by members of the Legionellaceae family.1,2 The first documented outbreak of LD occurred at the annual American Legion convention in Philadelphia during the summer of 1976.1 Several hundred people were stricken with the disease and 29 died. The causative pathogen identified was Legionella pneumophila.
Epidemiology
The CDC reports that between 5% and 30% of patients with LD die each year.1 The U.S. Occupational Safety and Health Administration indicates that about 25,000 cases of LD occur annually, with >4,000 deaths.3 About 10% of nonimmunocompromised patients treated will die, and about 80% of untreated immunocompromised patients die due to bacteremia or abscess formations in the lungs or at extrapulmonary sites.2,3 In the first 7 months of 2015, 2,472 cases of legionellosis were reported to the CDC.1 Recently, there have also been reported outbreaks of LD in New York City and Flint, Michigan.4
Legionellosis refers to two distinct clinical syndromes: LD, which most often manifests as severe pneumonia accompanied by multiorgan system disease, and Pontiac fever, which is an acute, febrile, self-limiting, viral-like illness (TABLE 1).3 When infection is established, Legionella organisms cause an acute fibrinopurulent pneu-monia with alveolitis and bronchitis. In addition to lung involvement, Legionella organisms may infect the lymph nodes, brain, kidneys, liver, spleen, bone marrow, and myocardium.5,6
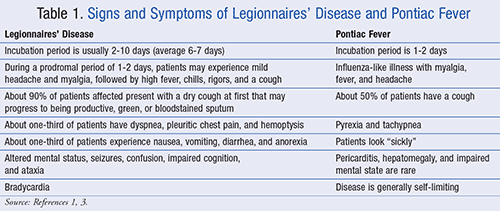
LD can be life-threatening, but with appropriate treatment most people recover, although recovery time varies greatly. Depending on their health status, some patients may experience rapid improvement, while others may have a more protracted course despite treatment. The mortality rate approaches 50% in patients with nosocomial infections.2 Progressive respiratory failure is believed to be the most common cause of death. However, the mortality rate depends on the comorbid conditions of patients, as well as on the choice of antibiotic and timeliness of admin-istration. The mode of acquisition (i.e., nosocomial or community-acquired) may also affect the outcome. In addition to typical signs and symptoms of pneumonia in a patient, a clinical parameter that may be especially useful at increasing the index of suspicion for LD is hyponatremia.
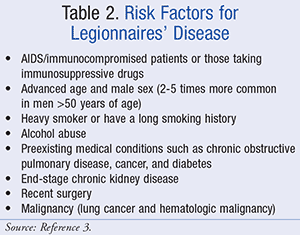
Risk factors for LD (TABLE 2) include age >50 years, although infants and neonates may also be affected.1,3 Premature neonates and immunocompromised or ventilated neonates in hospitals may be at high risk. About 40% to 50% of all cases are associated with travel, usually abroad, where air conditioning systems and stored water may be less well regulated.1 Person-to-person transmission has not been documented. Since some LD symptoms are similar to those of other types of pneumonia, LD may go undiagnosed for a period of time.
Fortunately, most people exposed to the bacteria do not become sick because LD is not contagious. Exposure must be through inhalation or aspiration of contaminated aerosolized water in aircraft or hotel facilities (TABLE 3).1,3 The highest incidence of LD occurs from late spring through early fall, when air conditioning systems are used more frequently. Nosocomial acquisition may also occur via aspiration with respiratory therapy equipment or consumption of contaminated water. In addition, transmission has been linked to the use of humidifiers, nebulizers, and items rinsed with contaminated tap water. Colonization and amplification of Legionella bacteria are facilitated by artificial water environments with water temperatures of 25°C to 42°C; stagnant water; scale and sediment in the water lines; and the presence of certain free-living aquatic amoebae that are capable of supporting intracellular growth of Legionella organisms.1

Pathophysiology
The Legionella bacterium is a small, aerobic, waterborne, gram-negative, rod-shaped, unencapsulated bacillus that is nonmotile, catalase-positive, and weakly oxidase-positive.2,7 It is a fastidious organism that does not grow anaerobically or on standard media. The Legionellaceae family consists of more than 50 species, constituting 64 serogroups. L pneumophila is the most common species, causing up to 90% of the cases of legionellosis, followed by L micdadei (otherwise known as the Pittsburgh pneumonia agent), L bozemanii, L dumoffii, and L longbeachae. Eighteen serogroups of L pneumophila have been identified, with serogroups 1, 4, and 6 being the primary causes of LD in humans. Serogroup 1 is thought to be responsible for about 80% of the reported cases of legionellosis caused by L pneumophila.7 Although most cases of LD are caused by L pneumophila, there are other species of the organism (most of which are also found in soil ) and mild illnesses including Pontiac fever that may be caused by these organisms.7 According to the CDC, Legionella bacteria are found naturally in water supply systems where they breed rapidly in favorable conditions; optimal conditions being a temperature of 35°C (range 25°C-45°C), stagnant water, and water containing debris, all acting as foci for bacterial breeding.1,3 The bacteria are distributed widely in the environment and can breed in natural and artificial water sources. However, they only become a risk to health when conditions allow their rapid multiplication.
Legionella species are obligate or facultative intracellular parasites.1,7 Water is the major environmental reservoir for Legionella; the bacteria can infect and replicate within protozoa such as Acanthamoeba and Hartmannella, which are free-living amoebae found in natural and manufactured water systems. Within the amebic cells, Legionella species can avoid the endosomal-lysosomal pathway and replicate within the phagosome. Surviving and growing in amebic cells allows Legionella to persist in nature. Some types of plastic and organic substances found in plumbing can provide nutrients and promote growth of the bacteria.1,2 These factors make modern plumbing systems a good habitat for the organism. Other factors that enable the organism to grow include temperature above 20°C, iron, L-cysteine, and biofilm (particularly protozoans).1,2 The bacteria enter plumbing systems via these water sources. The biofilm, scale, and corrosion that may build up over time allow the organism to proliferate.
Treatment for Legionnaires’ Disease
Treatment recommendations for LD are generally based on clinical experience with the first recognized breakout in Philadelphia in 1976.8 In a retrospective review, patients treated with erythromycin or tetracycline had a 50% lower mortality rate compared with patients treated with beta-lactam antibiotics.9 The choice of antibiotic is often dependent on the patient’s clinical state, the risk of adverse drug interactions and tolerance to the medication, and the degree of certainty as to the diagnosis. There are three major classes of antibiotics that are highly active in vitro against Legionella bacteria: fluoroquinolones (e.g., levofloxacin, moxifloxacin); macrolides (e.g., erythromycin, azithromycin); and tetracyclines (e.g., doxycycline) (TABLE 4).10,11

Macrolides except azithromycin can interact with drugs metabolized by the CYP3A4 isoenzyme (e.g., cyclosporine). Unlike the 14-member macrolides previously mentioned, the 15-member lactone ring of azithromycin does not interact with the CYP3A4 isoenzyme. Macrolides also have the potential to induce QT-interval prolongation syndrome; the rank (from highest to lowest) for this interaction is erythromycin, clarithromycin, roxithromycin, and azithromycin.12,13
The quinolones mentioned above do not inhibit CYP3A4 and are suitable drugs to treat LD in patients taking cyclosporine or other CYP3A4 substrates. An older fluoroquinolone, ciprofloxacin, inhibits CYP3A4 and may have limited use because the FDA has not approved fluoroquinolones for persons younger than 18 years (because of concerns about arthropathy in studies of juvenile animals), but they have been successfully used to treat children with LD.14-16 Doxycycline may cause some relatively minor gastrointestinal (GI) issues, but it may make exposed skin more sensitive to the sun. Patients should avoid prolonged sun exposure, tanning booths, and sunlamps, and use a sunscreen when exposed to longer periods of direct sunlight.
Beta-lactam antibiotics are not included because they do not penetrate Legionella’s intracellular compartment; therefore, they are not considered effective agents.8 Generally, azithromycin has been shown to be more active and is considered the drug of choice because it has high intracellular compartment penetration to kill the bacteria and may be used in adults and children.17-20 Azithromycin is particularly effective because of decreased GI irritation, higher potency, better intracellular penetration into tissue, and once-daily dosing.18,21 Severe cases of LD may be resistant to a single antibiotic. In these cases, a second drug such as rifampin may be added because rifampin is very active against extracellular and intracellular Legionella species.8,21-27
Although LD may present as a mild illness called Pontiac fever, this condition usually requires no specific antibiotic treatment. However, many patients with LD require hospitalization to receive parenteral antibiotics and fluids to treat the infection and prevent and/or treat dehydration or septic shock.3 Most healthy patients show clinical improvement within 3 to 5 days after antibiotic therapy begins. A delay in getting appropriate treatment significantly increases the risk of mortality in LD. Therefore, empirical anti-Legionella therapy is initiated promptly to treat severe community-acquired and nosocomial pneumonias. A study by van Loenhout et al, which included 190 patients with LD, found that a year after the disease’s onset, many patients were still suffering from one or more adverse health effects, particularly fatigue and reduced general quality of life.28 Most nosocomial infections and hospital outbreaks have been linked to a contaminated hot and/or cold water supply.29 Nosocomial LD associated with water birth is reported in a few neonates, but the risk appears to be low.30,31 Person-to-person transmission has not been demonstrated.
Role of Pharmacists
Pharmacists can assure patients that LD cannot be spread person-to-person and advise them that their water systems, including hot tubs and decorative pools, are possible sources of Legionella bacteria. The water must have the proper concentration of disinfectant chemicals, such as chlorine or bromine, and the desired pH (7.2-7.8) should be maintained in water systems. Establishing a proper cleaning and scrubbing schedule for the water system will help reduce the risk of Legionella bacterial growth.
If someone contracts LD, pharmacists can assure patients that this infection can usually be treated with common oral antibiotics. Prompt recognition of pneumonia signs and symptoms such as cough, fever, headache, and sometimes GI symptoms will help assure that patients are being seen faster by a physician, so that the illness can be treated with appropriate antibiotics as soon as possible.
If antibiotic therapy is necessary (i.e., with macrolides fluoroquinolones, or tetracyclines), pharmacists can monitor patients for any drug allergies, adverse drug interactions, and contraindications. Since new adverse drug reactions and drug-drug interactions are identified daily from postmarketing studies and clinical research, pharmacists need to review current research literature to determine the safety and appropriateness of drugs patients receive. All of these agents have the potential for a number of adverse drug-drug and food-drug interactions; ensuring that the patient is counseled on how to optimally administer prescribed antibiotics with regard to other drugs and foods is very important as well.
REFERENCES
1. Legionnaires’ disease. MedlinePlus. www.nlm.nih.gov/medlineplus/legionnairesdisease.html. Accessed November 19, 2015.
2. Legionnaires disease. Medscape. http://emedicine.medscape.com/article/220163-overview. Accessed December 28, 2015.
3. Legionella and Legionnaires’ disease. Patient. http://patient.info/doctor/legionella-and-legionnaires-disease. Accessed December 3, 2015.
4. McCoy W. How New York City can learn from Flint. NY Daily News. February 20, 2016. www.nydailynews.com/opinion/new-york-city-learn-flint-article-1.2537823. Accessed March 16, 2016.
5. American Academy of Pediatrics. Guideline. Legionella pneumophila infections. In: Pickering LK, Baker CJ, Kimberlin DW, Long SS, eds. Red Book: 2012 Report of the Committee on Infectious Diseases. 29th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2012:461-462.
6. Monforte R, Marco F, Estruch R, Campo E. Multiple organ involvement by Legionella pneumophila in a fatal case of Legionnaires’ disease. J Infect Dis. 1989;159(4):809.
7. Kozak-Muiznieks NA, Lucas CE, Brown E, et al. Prevalence of sequence types among clinical and environ-mental isolates of Legionella pneumophila serogroup 1 in the United States from 1982 to 2012. J Clin Microbiol. 2014;52(1):201-211.
8. Dedicoat, M, Venkatesan P. The treatment of Legionnaires’ disease. J Antimicrob Chemother. 1999;43(6):747-752.
9. Fraser DW, Tsal TR, Orenstein W, et al. 1977 Legionnaires’ disease: description of an epidemic of pneumonia. N Engl J Med. 1977;297:189-197.
10. Blázquez Garrido RM, Espinosa Parra FJ, Alemany Francés L, et al. Antimicrobial chemotherapy for Legionnaires’ disease: levofloxacin versus macrolides. Clin Infect Dis. 2005;40(6):800-806.
11. Stout JE, Sens K, Mietzner S, et al. Comparative activity of quinolones, macrolides and ketolides against Legionella species using in vitro broth dilution and intracellular susceptibility testing. Int J Antimicrob Agents. 2005;25(4):302-307.
12. Luttichau HR, Vinther C, Uldum SA, et al. An outbreak of Pontiac fever among children following use of a whirlpool. Clin Infect Dis. 1998;26(6):1374-1378.
13. Campins M, Ferrer A, Callis L, et al. Nosocomial Legionnaire’s disease in a children’s hospital. Pediatr Infect Dis J. 2000;19(3):228-234.
14. Gonzalez IA, Martin JM. Legionella pneumophila serogroup 1 pneumonia recurrence postbone marrow transplantation. Pediatr Infect Dis J. 2007;26:961-963.
15. Watson AM, Boyce TG, Wylam ME. Legionella pneumonia: infection during immunosuppressive therapy for idiopathic pulmonary hemosiderosis. Pediatr Infect Dis J. 2004;23(1):82-84.
16. Heine S, Fuchs A, von Muller L, et al. Legionellosis must be kept in mind in case of pneumonia with lung abscesses in children receiving therapeutic steroids. Infection. 2011;39(5):481-484.
17. Johnson DM, Erwin ME, Barrett MS, et al. Antimicro-bial activity of ten macrolide, lincosamine and streptogramin drugs tested against Legionella species. Eur J Clin Microbiol Infect Dis. 1992;11(8):751-755.
18. Edelstein PH, Edelstein MA. In vitro activity of azithromycin against clinical isolates of Legionella species. Antimicrob Agents Chemother. 1991;35(1):180-181.
19. Onody C, Matsiota-Bernard P, Nauciel C. Lack of resistance to erythromycin, rifampicin and ciprofloxacin in 98 clinical isolates of Legionella pneumophila. J Antimicrob Chemother. 1997;39:815-816.
20. File T Jr, Garau J, Blasi F, et al. Guidelines for empiric antimicrobial prescribing in community-acquired pneumonia. Chest. 2004;125(5):1888-1901.
21. Nimmo GR, Bull JZ. Comparative susceptibility of Legionella pneumophila and Legionella longbeachae to 12 antimicrobial agents. J Antimicrob Chemother. 1995;36(1):219-223.
22. Reda C, Quaresima T, Pastoris MC. In-vitro activity of six intracellular antibiotics against Legionella pneumophila strains of human and environmental origin. J Antimicrob Chemother. 1994;33:757-764.
23. Baltch AL, Smith RP, Ritz W. Inhibitory and bactericidal activities of levofloxacin, ofloxacin, erythromycin, and rifampin used singly and in combination against Legionella pneumophila. Antimicrob Agents Chemother. 1995;39:1661-1666.
24. Ridgway GL, Salman H, Robbins MJ, et al. The in-vitro activity of grepafloxacin against Chlamydia spp, Mycoplasma spp, Ureaplasma urealyticum and Legionella spp. J Antimicrob Chemother. 1997;40(suppl A):31-34.
25. Schulin T, Wennersten CB, Ferraro MJ, et al. Susceptibilities of Legionella spp to newer antimicrobials in vitro. Antimicrob Agents Chemother. 1998;42:1520-1523.
26. Cunha BA, ed. Pneumonia Essentials. 3rd ed. Sudbury, MA: Jones and Bartlett; 2010.
27. Cunha BA. Community-acquired pneumonia: empiric antimicrobial therapy. Consultant. 2013;53(6):410-417.
28. van Loenhout JA, van Tiel HH, van den Heuvel J, et al. Serious long-term health consequences of Q-fever and Legionnaires’ disease. J Infect. 2014;68(6):527-533.
29. Johansson PJ, Andersson K, Wiebe T, et al. Nosocomial transmission of Legionella pneumophila to a child from a hospital’s cold-water supply. Scand J Infect Dis. 2006;38:1023-1027.
30. Franzin L, Scolfaro C, Cabodi D, et al. Legionella pneumophila pneumonia in a newborn after water birth: a new mode of transmission [case report]. Clin Infect Dis. 2001;33(9):e103-e104.
31. Thoni A, Zech N, Moroder L, Ploner F. [Water contamination and infection rate after water births]. Gynakol Geburtshilfliche Rundsch. 2007;47(1):33-38.
To comment on this article, contact rdavidson@uspharmacist.com.





