US Pharm. 2013;38(12):38-42.
ABSTRACT: Proton pump
inhibitors (PPIs), available with or without a prescription, are
commonly used for the treatment of acid-related disorders. Despite their
ease of availability and common use, PPIs can have severe side effects.
The long-term consequences of chronic PPI use include the potential
increased risk of hypocalcemia, hypomagnesemia, Clostridium difficile
infections, and pneumonia. Community pharmacists are poised to provide
evidence-based recommendations and educate patients about the benefits
and risks associated with chronic PPI use.
Proton pump inhibitor (PPIs) have been on the market since the late 1980s and have replaced the histamine2 receptor-antagonists (H2RAs) as the most potent class of drugs for the treatment of acid-related diseases.1 Anti-ulcer medications (therapeutic areas are based on proprietary IMS Health definitions) were the ninth largest class based on prescription volume in the United States in 2012 and the 11th in sales.2,3
Medications in the PPI class are widely available with or without a prescription. Currently, the U.S. market contains six PPIs, two of which are also available as OTC products (TABLE 1).4-9 In the early 2000s, the FDA announced the availability of omeprazole (Prilosec OTC) as the first OTC PPI.10 It was soon followed by the approval of OTC lansoprazole (Prevacid 24HR).11
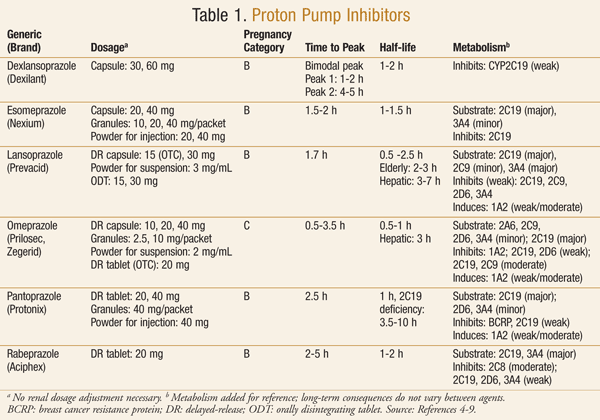
PPIs are used for the treatment of many gastric conditions including peptic ulcer disease, eradication of Helicobacter pylori infections, treatment and prevention of nonsteroidal anti-inflammatory drug (NSAID) gastroduodenal ulcer, Zollinger-Ellison syndrome, and gastroesophageal reflux disease (GERD).1 Generally, these medications are prescribed because of the low incidence of side effects and superior efficacy compared to other drugs used to treat the same conditions.1
Long-term use of any medication raises safety concerns, especially if that product is available OTC. The American Gastroenterological Association (AGA) released guidelines on the management of GERD in 2008 that recommended against routine monitoring for PPIs due to insufficient evidence.12 However, since then studies have continued to show long-term consequences from chronic PPI use including malabsorption consequences and infections. Subsequently, in March 2013, the American College of Gastroenterology (ACG) released guidelines for the diagnosis and treatment of GERD.13 These guidelines do provide some insight into monitoring for long-term consequences of chronic PPI use. This article is a review of the recent literature and guideline recommendations regarding the possible long-term consequences of chronic PPI pharmacotherapy and opportunities to prevent these complications.
In animal studies, PPIs raised concerns about a potential for hypergastrinemia, but human studies failed to show an association.1 Therefore, long-term consequences of chronic PPI use can be grouped into two main categories, malabsorption and infections.1 Malabsorption secondary to PPI use affects calcium and magnesium, and the literature specifies two infections most often associated with PPI use, Clostridium difficile and pneumonia. Unfortunately, a definition in the literature for “long-term” is lacking; neither the AGA guidelines nor the ACG guidelines define what is considered long-term. For the rest of this article, the authors use long-term to designate therapy greater than 14 days, the maximum therapy for the OTC products.10,11
Malabsorption
The first potential long-term consequence of chronic PPI use is malabsorption of key minerals in the body, namely calcium and magnesium. The loss of these minerals could lead to bone fractures or cardiac abnormalities.
Decreased Calcium Absorption (Hypocalcemia): Long-term PPI use has been associated with an increased risk of osteoporosis and decreased bone mineral density (BMD), with a 35% increased risk of fractures.14 Calcium serves an important role in bone health and formation, as it is a key component of hydroxyapatite (the main structural element of bone). Bone material is a major reservoir for calcium and may contain greater than 99% of a body’s calcium.15 The hypothesis for the mechanism of PPI-induced bone fractures is that dietary calcium absorption is dependent upon an acidic environment in the gastrointestinal (GI) tract. Due to the decrease in acidity from the pharmacologic effect of PPIs, a potential loss of calcium absorption occurs. This reduction in calcium absorption leads to decreased osteoclastic activity and thus decreases in BMD, thereby increasing fracture risk.1
The 2013 ACG guidelines on GERD state that existing osteoporosis is not a contraindication to PPI therapy.13 Patients with osteoporosis may remain on PPI therapy unless another risk factor for hip fracture exists.13 Furthermore, in March 2011, the FDA modified its osteoporosis and fracture warning. It was concluded that OTC products do not warrant label changes to include warnings of fracture risk.16
However, several studies have demonstrated an association between long-term PPI use and risk of fractures, but they contain numerous confounders. Common risk factors for fractures such as a sedentary lifestyle and concomitant use of certain medications (e.g., thiazide diuretics, hormone replacement therapy, corticosteroids) are often observed in patients who routinely take PPIs.14 Additionally, patients who take high doses of PPIs are at higher fracture risk versus patients who take lower OTC doses.17 Finally, patients who take PPIs for extended periods of time (>1 year) are more likely to experience a fracture.18
An analysis of the data obtained from the Canadian Multicentre Osteoporosis Study revealed that the use of PPIs was associated with lower BMD, particularly at the hip and femoral neck, when compared to non-PPI use.19 However, long-term PPI use was not associated with an accelerated decline in BMD. Targownik et al reported that patients using PPIs did have lower BMD; however, these patients were significantly older (66.3 vs. 60.9 years; P <.001) and had a higher mean body mass index (BMI) (28.3 vs. 26.9; P <.001).19
Data remain relatively inconclusive and conflicting regarding the magnitude of the PPI and fracture association in the absence of additional risk factors. According to the 2013 ACG guidelines, there is insufficient evidence to warrant routine BMD tests, calcium supplementation, or other routine precautions because of PPI use.13 In contrast, Health Canada issued an alert in April 2013 stating that patients with existing risk factors for osteoporosis should be monitored closely and should also receive short-term PPI therapy at the lowest effective dose.20 This is parallel to current recommendations from the FDA despite the lack of recommendations from the ACG.16 If calcium supplementation is indicated, use of calcium citrate is the preferred calcium supplement in patients taking PPIs, as it can be absorbed in the absence of an acidic environment.1
Decreased Magnesium Absorption (Hypomagnesemia): In March 2011, the FDA released a warning regarding low serum magnesium levels associated with long-term use of PPIs.21 An analysis of reports from the FDA’s Adverse Event Reporting System (AERS) states that approximately 1% of patients who experienced an adverse effect while on a PPI experienced hypomagnesemia.21 The mechanism behind the changes in absorption is unknown. Symptoms of hypomagnesemia include seizures, arrhythmias, hypotension, and tetany. Hypomagnesemia is also potentially fatal.22 Hypomagnesemia related to chronic PPI use was not addressed in the 2013 ACG guidelines.13
All PPIs are associated with decreased magnesium absorption.21 Hypomagnesemia was more common in older patients taking a PPI (mean age 64.4 years).21 Mean time to onset of hypomagnesemia was 5.5 years after initiation of therapy.21 Similarly, a systematic review of case reports found that patients who presented with hypomagnesemia in association with PPI use also presented with other electrolyte disturbances, specifically hypokalemia and hypocalcemia.23 Hypomagnesemia generally resolved with the discontinuation of the PPI and recurred soon after the PPI was rechallenged.24
Concurrent use of medications that also decrease magnesium increases the risk of significant hypomagnesemia. Danziger et al reported that patients who take a PPI with a diuretic have nearly a 55% greater risk of hypomagnesemia than patients who take only a PPI.22
An FDA Drug Safety Communication warns of the risks of hypomagnesemia and recommends that providers monitor serum magnesium levels in patients taking PPIs.21 The FDA suggests that providers obtain serum magnesium levels prior to initiation of therapy and periodically thereafter for patients who will continue prolonged treatment and for patients who take medications that also cause hypo-magnesemia. Patients who present with clinically significant hypomagnesemia may require discontinuation of PPI therapy, magnesium replacement via oral or IV methods, and treatment with an alternative class of drugs for GI conditions such as an H2RA.1
Infections
In addition to decreased magnesium and calcium absorption, patients on long-term PPIs may be at an increased risk of infection. The hypothesis for the mechanism of action is that the gastric acid secretions act as a defense mechanism against enteric bacteria, and the increased gastric pH during PPI use allows for colonization of opportunistic microbes.1 The 2013 ACG guidelines warned about the risk of increased infections of C difficile and community-acquired pneumonia (CAP).13
Clostridium difficile: In a 2005 retrospective study, researchers found that patients who were taking PPIs had a hazard ratio (HR) of 2.9 (95% CI, 2.4-3.4); i.e., patients had a 2.9-fold increase in the risk of acquiring C difficile than patients who were not on a PPI.25 Seventy-five percent of the patients with reported cases were over the age of 65 years. Not only does long-term use of PPIs cause an increased incidence of C difficile, but patients who received a PPI during treatment of C difficile were also 42% (95% CI, 1.11-1.82) more likely to have a recurrent infection after finishing therapy.25
A 2010 study by Linsky et al looked at the association of PPI use and recurrent C difficile.26 The authors determined whether or not the patient had an infection with recurrent C difficile, 15 to 90 days after initial C difficile infection, if the patient received a PPI within 14 days of initial C difficle infection. The HR for patients exposed to PPIs during treatment was 1.42 (95% CI, 1.11-1.82). For patients over the age of 80 years, the HR increases from 1.42 to 1.86 (95% CI, 1.15-3.01).26
In 2012, the FDA issued a statement detailing the relationship between C difficile–associated diarrhea (CDAD) with the use of a PPI.27 The FDA safety alert warns patients and healthcare professionals to consider CDAD if a patient takes a PPI and experiences persistent diarrhea.27 The FDA also recommends that patients be on the lowest dose for the shortest period of time to treat their current condition.27 The 2013 ACG guidelines recommend use of PPIs with caution in patients with a risk of C difficile infections.13
Community-Acquired Pneumonia: Patients taking PPIs may potentially be at an increased risk for CAP. However, the degree of association is unclear due to conflicting data.28-30 The 2013 ACG guidelines state that short-term PPI use may increase the risk of CAP, but the risk does not seem to be elevated in long-term use.13
A 2012 cohort study by de Jagar et al showed that patients on PPIs were 2.23 times (95% CI, 1.28-3.75) more likely to develop a CAP infection compared to patients not on PPIs.31 Unfortunately, the duration of time that patients were prescribed was not included in the study design.31 In a meta-analysis completed in 2004, researchers discovered that patients who were on an acid-suppressing agent, either a PPI or an H2RA, were 4.5 (95% CI, 3.8-5.1) times more likely to develop pneumonia.30 Mean duration of use for H2RAs was 2.8 months; for PPIs the mean duration was 5 months.30
Conversely, a 2008 study conducted by Sarkar et al showed that current PPI use was not associated with an increased risk of CAP (odds ratio [OR] 1.02, 95% CI 0.97-1.08).29 However, the study did observe an increased risk of acquiring an infection in patients initiated on a PPI within the past 14 days (adjusted OR 3.21, 95% CI 2.46-4.18).29
The data support a short-term increase risk of pneumonia infections, but they are conflicting regarding long-term consequences. Despite the conflicting data, this risk is important to consider, especially because of the new Centers for Medicare & Medicaid (CMS) regulations on hospital readmissions.32 Laheij et al determined that the incidence rate of pneumonia was 2.5 per 100 patient-years for patients on PPIs.30 With 65.7 million prescriptions of omeprazole alone and an increased $15,682 cost to Medicare beneficiaries due to pneumonia hospitalizations, the risk of PPI use-associated infections warrants vigilance and evidence-based medicine on the part of the pharmacist.33,34
Conclusion
PPIs are an efficacious and safe drug class. They offer relief to patients in a patient-centered healthcare system. Unfortunately, these agents do potentially have some long-term consequences from continued use, including malabsorption issues and increased risk of infections. The pharmacist can be an advocate for the patient in the hospital system or in the community by understanding these risks and fostering patient-centered care by empowering a well-informed patient in healthcare decisions.
REFERENCES
1. Wolfe M. Overview and comparison of proton pump inhibitors for the treatment of acid-related disorders. In: Basow DW, ed. UpToDate. Waltham, MA: UpToDate; 2013.
2. Top therapeutic classes by dispensed prescriptions
(US). IMS Health. Updated March 22, 2013.
www.imshealth.com/deployedfiles/imshealth/Global/Content/Corporate/Press%20Room/2012_U.S/Top_Therapeutic_Classes_Dispensed_Prescriptions_2012.pdf.
Accessed November 4, 2013.
3. Top therapeutic classes by non-discounted spending
(US). IMS Health. Updated February 17, 2013.
www.imshealth.com/deployedfiles/imshealth/Global/Content/Corporate/Press%20Room/2012_U.S/Top_Therapeutic_Classes_by_Non-Discounted_Spending.U.S.pdf.
Accessed November 4 2013.
4. Dexlansoprazole. Lexi-Comp Online. Hudson, OH: Lexi-Comp, Inc. http://online.lexi.com. Accessed August 15, 2013.
5. Esomeprazole. Lexi-Comp Online. Hudson, OH: Lexi-Comp, Inc. http://online.lexi.com. Accessed August 15, 2013.
6. Lansoprazole. Lexi-Comp Online. Hudson, OH: Lexi-Comp, Inc. http://online.lexi.com. Accessed August 15, 2013.
7. Omeprazole. Lexi-Comp Online. Hudson, OH: Lexi-Comp, Inc. http://online.lexi.com. Accessed August 15, 2013.
8. Pantoprazole. Lexi-Comp Online. Hudson, OH: Lexi-Comp, Inc. http://online.lexi.com. Accessed August 15, 2013.
9. Rabeprazole. Lexi-Comp Online. Hudson, OH: Lexi-Comp, Inc. http://online.lexi.com. Accessed August 15, 2013.
10. Prilosec OTC (omeprazole) information. FDA.
September 21, 2010.
www.fda.gov/AboutFDA/CentersOffices/OfficeofMedicalProductsandTobacco/CDER/ucm220944.htm.
Accessed June 5, 2013.
11. Novartis launches Prevacid 24HR over-the-counter for
true 24-hour frequent heartburn relief. Novartis press releases.
November 12, 2009.
https://www.prevacid24hr.com/news-press-releases.html. Accessed June 5,
2013.
12. Kahrilas PJ, Shaheen NJ, Vaezi MF, et al. American
Gastroenterological Association Institute technical review on the
management of gastroesophageal reflux disease. Gastroenterology. 2008;135:1392-1413.
13. Katz P, Gerson L, Vela, M. Guidelines for the diagnosis and management of gastroesophageal reflux disease. Am J Gastroenterol. 2013;108:308-328.
14. Khalili H, Huang ES, Jacobson BC, et al. Use of proton
pump inhibitors and risk of hip fracture in relation to dietary and
lifestyle factors: a prospective cohort study. BMJ. 2012;344:e372.
15. Flynn A. The role of dietary calcium in bone health. Proc Nutr Soc. 2003;62:851-858.
16. FDA Drug Safety Communication: Possible increased risk
of fractures of the hip, wrist, and spine with the use of proton pump
inhibitors. FDA. March 28, 2011.
www.fda.gov/drugs/drugsafety/postmarketdrugsafetyinformationforpatientsandproviders/ucm213206.htm.
Accessed June 11, 2013.
17. Yang YX, Lewis JD, Epstein S, Metz DC. Long-term proton pump inhibitor therapy and risk of hip fracture. JAMA. 2006;296:2947-2953.
18. Targownik LE, Lix LM, Metge CJ, et al. Use of proton pump inhibitors and risk of osteoporosis-related fractures. CMAJ. 2008;179:319-326.
19. Targownik LE, Leslie WE, Davison KS, et al. The
relationship between proton pump inhibitor use and longitudinal change
in bone mineral density: a population-based study from the Canadian
Multicentre Osteoporosis Study (CaMos). Am J Gastroenterol. 2012;107:1361-1369.
20. Proton pump inhibitors: risk of bone fractures. Health Canada. April 4, 2013. http://healthycanadians.gc.ca/recall-alert-rappel-avis/hc-sc/2013/26523a-eng.php. Accessed August 27, 2013.
21. FDA Drug Safety Communication: Low magnesium levels
can be associated with long-term use of proton pump inhibitor drugs
(PPIs). FDA. March 14, 2011. www.fda.gov/drugs/drugsafety/ucm245011.htm.
Accessed June 11, 2013.
22. Danziger J, William JH, Scott DJ, et al. Proton-pump inhibitor use is associated with low serum magnesium concentrations. Kidney Int. 2013;83:692-699.
23. Luk CP, Parsons R, Lee YP, Hughes JD. Proton pump inhibitor-associated hypomagnesemia: what do FDA data tell us? Ann Pharmacother. 2013;47:773-780.
24. Hess MW, Hoenderop JG, Bindels RJ, Drenth JP. Systematic review: hypomagnesaemia induced by proton pump inhibition. Aliment Pharmacol Ther. 2012;36:405-413.
25. Dial S, Delaney JA, Barkun AN, Suissa S. Use of gastric acid-suppressive agents and the risk of community-acquired Clostridium difficile-associated disease. JAMA. 2005;294:2989-2995.
26. Linsky A, Gupta K, Lawler EV, et al. Proton pump inhibitors and risk for recurrent Clostridium difficile infection. Arch Intern Med. 2010;170:772-778.
27. Proton pump inhibitors (PPIs)—drug safety communication: Clostridium difficile-associated
diarrhea (CDAD) can be associated with stomach acid drugs. FDA.
www.fda.gov/safety/medwatch/safetyinformation/safetyalertsforhumanmedicalproducts/ucm290838.htm.
Accessed June 5, 2013.
28. Gulmez SE, Holm A, Frederiksen H, et al. Use of proton
pump inhibitors and the risk of community-acquired pneumonia: a
population-based case-control study. Arch Intern Med. 2007;167:950-955.
29. Sarkar M, Hennessy S, Yang YX. Proton-pump inhibitor use and the risk for community-acquired pneumonia. Ann Intern Med. 2008;149:391-398.
30. Laheij RJ, Sturkenboom MC, Hassing RJ, et al. Risk of
community-acquired pneumonia and use of gastric acid-suppressive drugs. JAMA. 2004;292:1955-1960.
31. de Jager CP, Wever PC, Gemen EF, et al. Proton pump inhibitor therapy predisposes to community-acquired Streptococcus pneumoniae pneumonia. Aliment Pharmacol Ther. 2012;36:941-949.
32. Rau J. Medicare to penalize 2,217 hospitals for excess readmissions. Kaiser Health News.
August 13, 2012.
www.kaiserhealthnews.org/Stories/2012/August/13/medicare-hospitals-readmissions-penalties.aspx.
Accessed September 1, 2012.
33. Top 25 medicines by dispensed prescriptions (US). IMS
Health. Updated March 22, 2013.
www.imshealth.com/deployedfiles/imshealth/Global/Content/Corporate/Press%20Room/2012_U.S/Top_25_Medicines_Dispensed_Prescriptions_U.S.pdf.
Accessed November 4, 2013.
34. Thomas CP, Ryan M, Chapman JD, et al. Incidence and cost of pneumonia in Medicare beneficiaries. Chest. 2012;142:973-981.
To comment on this article, contact rdavidson@uspharmacist.com.





