US Pharm. 2007;32(8):28-37.
Osteoporosis, known as the silent disease, can occur in all patients once their bones become thin and weak. While the risk of osteoporosis is more frequent in postmenopausal women, Caucasian and Asian males are still at risk. In 2004, 30% of hip fractures were sustained by males, with greater morbidity and mortality than their female counterparts. 1 Approximately two million men are currently affected and three million men are at risk. By the year 2020, 20.5 million males older than 50 years will have osteoporosis and low bone mass.2
Bone mass peaks in most people during their third decade of life. At this time, males will have accumulated more bone mass than females.3 The rate of bone loss will equalize by age 65 or 70. Males have several genetic advantages conferring less risk; these include higher peak bone mass (10%-15% higher bone mass than females at skeletal maturity) with less bone loss, absence of menopause, and shorter life expectancy.4,5 Males primarily present with fractures 10 years later than females due to their greater bone mass.6 Although the majority of hip fractures occur in women, men have higher morbidity and disability. Six percent of males will suffer from a hip fracture and 5% from vertebral fractures.3
Etiology
Osteoporosis is categorized by two main types: primary (type I and II) and
secondary (type III). Primary osteoporosis is further divided into two
segments, age-related bone loss due to menopause (type I) or advancing age
(type II/senile osteoporosis) and bone loss of unknown origin (idiopathic).
Idiopathic osteoporosis is suspected in males diagnosed before age 70.3
Males primarily experience trabecular thinning of bone, while females lose
trabecular connectivity over time.1 Secondary osteoporosis (type
III) is often linked to more than one etiology, with the more common causes
being hypogonadism, tobacco abuse, gastrointestinal disease, immobilization,
chronic glucocorticoid use (more than 5 mg/day for longer than three months),
alcohol abuse, and hypercalcemia.3 Approximately 70% of
osteoporotic cases are secondary to these conditions, occurring before age 50.
Therefore, secondary osteoporosis should be suspected first and primary second
(Table 1).5
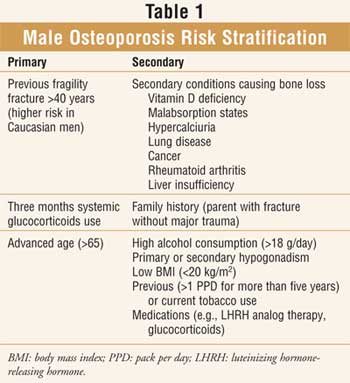
Evidence also suggests a link between secondary osteoporosis and estrogen deficiency. This condition exists in men afflicted with hypogonadism and other rare disorders leading to estrogen deficiencies. Alpha and beta estrogen receptors have been identified in bone marrow stromal cells and in osteoblasts.1 This hypothesis is still under investigation regarding skeletal maturation, sex-steroid synthesis, and bone mass in men.
Screening and Testing
Consensus guidelines are lacking for screening in men, but clinical experts
have made recommendations for these parameters. A history and physical exam
should be conducted initially for the diagnosis of osteoporosis. Additionally,
urine and hematologic tests, medication histories, and x-rays (evidence seen
with 30%-50% loss in bone mass) should also be conducted (Table 2).
Bone mineral density (BMD) testing can determine risk of development, identify
incidence, and assess efficacy of pharmacotherapy. The most recognized form of
BMD testing is the dual-energy x-ray absorptiometry test of the spine and hip.
3 Males commonly will present with vertebral fractures or hip fractures
prior to routine BMD screening.6

Interpretation of the BMD findings remains controversial as the World Health Organization (WHO) guidelines are used to assess osteoporosis (T scores) in women. The International Society for Clinical Densitometry (ISCD) offers separate guidelines for BMD interpretation in men.
Initial biochemical panels and a complete blood count can reveal findings that warrant further investigation (Table 2). Examples include irregular levels of calcium, alkaline phosphatase, phosphorous, and liver enzymes. Skeletal biopsy can alternatively be evaluated in the absence of supportive laboratory findings and is often reserved in patients who present with continued history of bone pain and/ or fractures with the absence of irregular laboratory indices. Biochemical markers such as C-terminal and N-terminal propeptides of type I collagen and/or bone resorption crosslinking telopeptides are independent predictors of fractures in men and women.7
BMD Testing: Males presenting with back pain, height loss, or kyphosis may have lateral spinal radiography to evaluate the presence of vertebral fractures. Markers of bone turnover may also be measured in the serum or urine although they are not specific to bone and are not recommended at this time for the clinical assessment of osteoporosis in men. The National Osteoporosis Foundation (NOF) does not have official recommendations for men. BMD testing is primarily recommended in men older than 65 or in younger men if secondary causes are evident or there is risk for fractures (Table 1). The ISCD recommends BMD testing in men older than 70.8
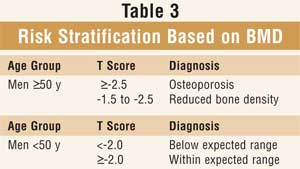
Interpreting T Scores: In males older than 50, osteoporosis is defined by the WHO as a T score of -2.5 or less. The z score (<-2.0) can be considered in males under age 50. Nomenclature has evolved from diagnosing osteopenia (T score between -2.5 and -1.5) to "reduced bone density." The decision to initiate therapy should not be made on T scores alone but in concert with risk factors, clinical presentation, and history (Table 3).
Treatment in Men
Treatment should involve a patient–clinician discussion about pharmacologic
options as well as prevention strategies. Prevention strategies are designed
to maximize peak bone mass, which occurs by age 30, and minimize bone loss.
Fall prevention, importance of regular weight-bearing exercise, tobacco
cessation, the amount of alcohol consumed (<2-3 oz./day), and the importance
of adequate calcium and vitamin D intake are all examples of prevention
measures that should be discussed.
Fall prevention measures should include, but are not limited to, use of a cane or walker for added stability; wearing appropriate footwear when going outside; floor safety (remove loose wires, cords, and throw rugs); bathroom safety (install grab bars, nonskid rails, and nonskid decals in tub/shower); and appropriate lighting (well-lit halls, stairways, and entrances, use of nightlights).
Weight-bearing activity refers to aerobic exercise on the feet, with the bones supporting the body weight. Examples include walking, jogging, dancing, low-impact aerobics, stair climbing, and gardening. Swimming and water aerobics offer many health benefits but have no impact on increasing bone density. Resistance exercises, such as weight lifting, are another form of activity that can also be used to help strengthen bone. Both weight-bearing and resistance training exercises provide additional benefits because they increase muscle strength, coordination, and overall health. All patients should be encouraged to discontinue smoking and limit the amount of alcohol consumed, as both can be damaging to the bones and lead to other health conditions.
The NOF recommends 1,200 mg of calcium per day for all adults age 50 and older.6,9,10 Calcium can be obtained through natural dietary sources and/or by calcium supplementation. Examples of dietary sources for calcium include one cup of milk (290-315 mg), low-fat yogurt (340-450 mg), collard greens (300-350 mg), and calcium-fortified foods such as soy milk (80-300 mg), orange juice (300 mg), and cereal (up to 1,000 mg). 9,11 Other sources of calcium-rich foods include fish and shellfish (salmon, shrimp), vegetables (broccoli, beans, cabbage, turnip greens, bok choy), tofu, almonds, and dried figs. If calcium supplements are needed, it is recommended to take 500 to 600 mg per dose and up to three doses per day. It is important to remember that calcium supplements offer different amounts of elemental calcium depending on the salt form. Calcium carbonate is 40% elemental calcium, and the absorption of this supplement can be impaired in patients with high gastric pH, including patients taking acid-suppressing medications (e.g., proton pump inhibitors) and the elderly. Calcium citrate is 21% elemental calcium, but it is better absorbed than calcium carbonate.11
The recommended daily allowance of vitamin D, as supported by the NOF, is 800 to 1,000 IU.6,9,10 Vitamin D is produced in the skin following exposure to direct sunlight and the amount produced varies depending on the time of day, season, latitude, and skin pigmentation. Exposure to the hands, arms, and face two to three times a week for 10 to 15 minutes is usually enough to meet the body's vitamin D requirement. There are a variety of factors, however, that decrease the skin's ability to manufacture vitamin D, including sunscreen use, clothing, and window glass that provides some level of sun protection factor; skin color (darker skin produces less vitamin D); and age (less vitamin D is made through the skin as adults age). Like calcium, Vitamin D can be obtained through diet and supplements. Examples of vitamin D–rich foods include egg yolks, saltwater fish, liver, and fortified dairy products.9 Remember to check the labels of calcium supplements and multivitamins to determine how much vitamin D each product contains.
Although there are no formal guidelines regarding the treatment of osteoporosis in men, a reasonable approach would be to recommend pharmacologic treatment for males at highest risk of a fracture. Those at highest risk include: 1) men older than 65 with a T score less than -2.5 (at any site measured); 2) men older than 50 with fragility or a vertebral compression fracture and a T score less than -1.5; 3) men receiving glucocorticoid therapy for more than three months with a T score less than -1.5; or 4) men with clinical hypogonadism and a T score less than -1.5. 7,12
Pharmacologic treatment options for osteoporosis in men include testosterone, bisphosphonates, and intermittent parathyroid hormone (PTH) (Table 4). Testosterone replacement therapy has been shown to decrease bone remodeling and increase trabecular BMD in men with acquired hypogonadism.13 However, the use of testosterone replacement in men older than 65 showed improvement in the lumbar BMD for those with low testosterone levels prior to treatment but did not show any improvement in the patients who had normal testosterone levels at the time of treatment.14 Due to the relatively small numbers of men included in the studies and the increase in the prostate-specific antigen, larger studies are needed to establish the safety and efficacy of testosterone therapy in males with osteoporosis and hypogonadism.13,14
Bisphosphonates inhibit the osteoclast-mediated resorption. Alendronate (Fosamax) and risedronate (Actonel) are FDA approved for the treatment of osteoporosis and glucocorticoid-induced osteoporosis. There have been several small studies that showed significant increases in BMD in the lumbar spine, femoral neck, trochanter, and total body, and a trend toward vertebral fracture reductions was observed.15-17 Orwoll et al. studied the effects of 10 mg of alendronate daily versus placebo on BMD over a two-year period. The study included 241 men (mean age, 63) and was a randomized, double-blind study. Those individuals who received 10 mg of alendronate daily had significant increases in the BMD of their lumbar spine, femoral neck, and total body. In addition, a decrease in the incidence of vertebral fractures was observed; however, this was not the primary end point of the trial.15 Sato et al. conducted an 18-month randomized, double-blind study of 280 Japanese men older than 65 (mean age, 76) who received 2.5 mg daily of risedronate or placebo poststroke. The authors reported that risedronate significantly increased BMD and reduced the number of hip fractures in the treatment group.16 Another study involving risedronate was a single-center, open-label, randomized trial conducted in 316 men with primary or secondary osteoporosis. The patients were randomized to risedronate 5 mg plus calcium 1,000 mg/day and vitamin D 800 IU/day; calcium 500 mg/day; or calcium 800 mg/day plus vitamin D 1,000 IU/day for one year. A significant increase in lumbar spine, total hip, and femoral hip BMD was demonstrated. There was a 60% reduction in the incidence of new vertebral fractures detected; however, this was a secondary end point.17 Limitations to these trials include the following: the majority of subjects were white (except Sato et al. study in Japan), the duration of the trials was one to two years, and the recommended dosages of bisphosphonates in the trials was daily rather than weekly as most commonly prescribed. The biophosphonate ibandronate (Boniva) does not have FDA indications for the treatment of osteoporosis in males, glucocorticoid-induced osteoporosis, or hypogonadal osteoporosis.
The recommended treatment doses for alendronate and risedronate can be found in Table 4. The most common side effects for these agents are gastrointestinal (e.g., nausea, acid regurgitation, gastroesophageal reflux, abdominal pain). Patients should be instructed to take these agents with a full glass of water on an empty stomach 30 minutes before first food or beverage and remain in an upright position for the first 30 minutes to reduce the potential for esophageal irritation.

Another treatment option for osteoporosis and hypogonadal osteoporosis is PTH (teriparatide [Forteo]), which is the first bone-forming agent to be approved for the treatment of osteoporosis. Teriparatide is given at low, intermittent doses and works by stimulating osteoblasts within the cancellous bone. PTH has demonstrated substantial increases to the lumbar spine and femoral neck BMD, but additional studies are needed to evaluate the effect of teriparatide on fractures.18-20 Kurland et al. studied the effects of 400 IU PTH compared to placebo in an 18-month randomized, double-blind, placebo-controlled trial. All men received 1,500 mg calcium and 400 IU vitamin D daily. Those treated with PTH demonstrated a statistically significant increase in BMD of the lumbar spine and femoral neck.18 Orwoll et al. studied the effects of placebo, 20 mcg teriparatide, and 40 mcg teriparatide in 437 men with osteoporosis in a randomized, double-blind study. Patients who received teriparatide had statistically significant improvement in their BMD in the spine and femoral neck.19 A more recent study by Kaufman et al. observed the fracture rates and BMD of 355 men previously treated with teriparatide 20 mcg or 40 mcg daily for one year, following them for 30 months posttreatment. BMD showed a gradual decline following discontinuation of treatment, but the lumbar spine and total hip BMD remained significantly higher than baseline BMD. The risk of vertebral fractures was reduced by 51% in the combined teriparatide versus placebo, but this was not significant. The risk of moderate or severe vertebral fractures was significantly reduced (by 83%) in the combined teriparatide group versus the placebo group.20
The recommended dosage for teriparatide is found in Table 4. Adverse reactions associated with teriparatide are generally mild, with pain at the injection site, dizziness, and leg cramps the most commonly observed events. Patients should be instructed to rotate injection sites in the thigh or abdominal areas and to sit when administering the medication.
Conclusion
It is well known that osteoporosis is a health problem in women, yet this
condition remains underdiagnosed and underreported in men. Medicare
reimbursement for BMD testing is limited for men unless they meet one of the
following criteria: 1) vertebral abnormalities; 2) long-term glucocorticoid
therapy; 3) primary hyperparathyroidism; and 4) follow-up monitoring or
assessment of osteoporosis therapy. These criteria likely contribute to the
low numbers of men diagnosed with osteoporosis.21 In addition,
there remains a need for large, multicenter, randomized, double-blind,
placebo-controlled studies to address vertebral and nonvertebral fractures,
particularly hip fractures, as primary end points for treatment agents.
References
1. Stock H, Schneider A, Strauss E. Osteoporosis: a disease in men.
Clinical Orthopaedics and Related Research. 2004; 425:143-151.
2. America's Bone Health. Washington DC: National Osteoporosis Foundation.
Updated 2002. Available at: www.nof.org. Accessed May 20, 2007.
3. Osteoporosis in Men. Bethesda, MD: National Institutes of Health
Osteoporosis and Related Bone Diseases – National Resource Center. Updated
November 2006. Available at: www.niams.nih.gov/bone/hi/osteoporosis_men.htm.
Accessed May 20, 2007.
4. Osteoporosis Management: Osteoporosis in Men. Chicago, Ill: American
Medical Association. Updated December 2004 – 2006. Available at:
www.amacmeonline.com/osteo_mgmt/module07/pdf/osteo_mgmt_07.pdf. Accessed May
20, 2007.
5. Licata A. Osteoporosis in men: suspect secondary disease first. Cleve
Clin J Med. 2003;70:247-254.
6. Campion JM, Maricic MJ. Osteoporosis in men. Am Fam Physician.
2003;67:1521-1526.
7. Seeman E, Bianchi G, Khosla S, et al. Bone fragility in men–where are we?
Osteoporos Int. 2006;17:1577-1583.
8. International Society for Clinical Densitometry Official Position. West
Hartford, Conn: International Society for Clinical Densitometry. Updated
September 2005. Available at:
www.iscd.org/Visitors/positions/OfficialPositionsText.cfm. Accessed May 20,
2007.
9. National Osteoporosis Foundation's Updated Recommendations for Calcium and
Vitamin D3 Intake. Washington, DC: National Osteoporosis
Foundation. Updated March 13, 2007. Available at:
www.nof.org/prevention/calcium_and_VitaminD.htm. Accessed May 25, 2007.
10. Boonen S, Vanderschueren D, Haentjens P, Lips P. Calcium and vitamin D in
the prevention and treatment of osteoporosis--a clinical update. J Int
Med. 2006;259:539-552.
11. Calcium and vitamin D supplementation: who needs it? Pharmacist's
Letter/Prescriber's Letter. 2007;23:230304.
12. Khan AA, Hodsman AB, Papaioannou A, et al. Management of osteoporosis in
men: an update and case example. CMAJ. 2007;176:345-348.
13. Katznelson L, Finkelstein JS, Schoenfeld DA, et al. Increase in bone
density and lean body mass during testosterone administration in men with
acquired hypogonadism. J Clin Endocrinol Metab. 1996;81:4358-4365.
14. Synder PJ, Peachey H, Hannoush P, et al. Effect of testosterone treatment
on bone mineral density in men over 65 years of age. J Clin Endocrinol Metab
. 1999;84:1966-1972.
15. Orwoll E, Ettinger M, Weiss S, et al. Alendronate for the treatment of
osteoporosis in men. N Engl J Med. 2000;343:604-610.
16. Sato Y, Iwamoto J, Kanoko T, et al. Risedronate sodium therapy for
prevention of hip fracture in men 65 years or older after stroke. Arch
Intern Med. 2005;165:1743-1748.
17. Ringe JD, Faber H, Farahmand P, et al. Efficacy of risedronate in men with
primary and secondary osteoporosis: results of a 1-year study. Rheumatol Int
. 2006;26:427-243.
18. Kurland ES, Cosman F, McMahon, et al. Parathyroid hormone as a therapy for
idiopathic osteoporosis in men: effects on bone mineral density and bone
markers. J Clin Endocrinol Metab. 2000;85:3069-3076.
19. Orwoll ES, Scheele WH, Paul S, et al. The effect of teriparatide [human
parathyroid hormone (1-34)] therapy on bone density in men with osteoporosis
. J Bone Min Res. 2003;18:9-17.
20. Kaufman J-M, Orwoll E, Goemaere S, et al. Teriparatide effects on
vertebral fractures and bone mineral density in men with osteoporosis:
treatment and discontinuation of therapy. Osteoporos Int.
2005;16:510-516.
21. Frequently asked questions regarding bone density testing for Medicare
beneficiaries. Washington DC: National Osteoporosis Foundation. Updated 2007.
Available at: www.nof.org/professionals/reimbursement/reimbursement_faqs.htm.
Accessed May 25, 2007.
To comment on this article, contact editor@uspharmacist.com.






