US Pharm. 2016;41(5):15-17.
Health experts have warned men against smoking tobacco, drinking alcohol, and taking unnecessary drugs if they are planning to be fathers. Conservative measures such as these are advised in an attempt to avoid increasing the risk of health conditions in children. Today, the focus on men as partners in reproductive health has expanded to include several international initiatives; the CDC encourages men, women, and all couples to have a reproductive life plan.1,2
The notion that men should be targeted for sexual and reproductive health promotion has evolved gradually; some experts think men should be encouraged to play a direct, active, and constructive role as part of a broader reproductive health agenda.3,4 Recently, researchers have given increased attention to identifying causes of male factor infertility (MFI), ranging from genetic Y-chromosome microdeletions to mechanisms of environmental damage in sperm production.5 Still evolving, however, is a clear understanding of how numerous medications may cause MFI, which is often treatable and reversible.5 In addition, knowledge about paternal age-related decline in sperm quality can potentially help couples with reproductive life planning.
Preconception Care for Men
According to the World Health Organization (WHO), preconception care is the provision of biomedical, behavioral, and social health interventions to women and couples before conception occurs.6 Experts indicate that much of the attention regarding preconception health promotion has been focused on women, with much less attention given to men’s preconception health and healthcare.4 Preconception care for men offers a number of benefits, including opportunities for disease prevention and health promotion and the improvement of family planning and pregnancy outcomes. TABLE 1 outlines these benefits and others.

Of note, benefits are not limited to an individual male’s health over a lifetime, but may also provide positive outcomes for the overall reproductive health of a nation (e.g., improved pregnancy outcomes); for a detailed discussion of recommendations to promote preconception care for men in the United States, refer to Reference 4. For further preconception-care perspectives, target groups, and specific regional considerations, as well as the full package of interventions, refer to Reference 7. Raising awareness of this issue can be an integral part of the pharmacist’s participation in health promotion and patient advocacy, particularly as it relates to a risk-versus-benefit analysis of medication therapy that is focused on patient preferences and participation in therapeutic outcomes.
Age-Related Decline in Sperm Quality
The age of a mother has frequently been considered a genetic risk factor for offspring; fathers, too, carry weight regarding this issue, particularly when it comes to their offspring’s mental health. In light of the lifelong fertility of the male sex, older men should understand that the rate of genetic mutations transmitted via sperm cells increases significantly with age; risk for psychiatric disorders in offspring, especially autism spectrum disorders (ASD) and schizophrenia, have been noted.4,8
Many men are currently unaware of the increasing body of evidence associating paternal age with schizophrenia in their children.4 One cohort study demonstrated an offspring’s increased risk for schizophrenia even after adjusting for family history of schizophrenia, birth exposures, socioeconomics, and early parental death.9 Another cohort study revealed that the relative risk for offspring with schizophrenia increases with every 5-year increase in paternal age; ultimately, fathers >50 years of age have an adjusted relative risk of 2.96.10 Furthermore, these authors indicated that for fathers aged 30 to 35 years, one out of 99 offspring is estimated to have schizophrenia, compared to one out of 47 for fathers aged >50 years.
And although paternal age is recognized as a contributing factor in ASD in offspring, evidence suggests a more complex picture than had been previously thought.11 Data from the largest study to date of parental age and ASD risk indicate a complex relationship between parental age and ASD risk.12 The researchers discovered that the risk for ASD in children increases with increasing age of both the mother and the father.12 Moreover, the risk is even greater when both parents are older and when either the mother or the father is at least 10 years younger than the other parent.11,12 These findings have been shown in multiple geographic regions; therefore, it has been suggested by these results that multiple mechanisms are contributing to the association between parental age and ASD risk.13
Exposure-Related Damage to Sperm Quality
Damage to sperm DNA can occur in many ways, including exposure to tobacco, alcohol, illicit drugs, medications, radiation, and chemotherapy (TABLE 2).4 Sperm count and quality may be reduced by untreated medical conditions such as diabetes mellitus, varicoceles, and epididymitis. Oxidative stress and DNA damage to sperm can be caused by xenobiotics (e.g., polychlorinated biphenyls [PCBs], dioxins).4 It is important to note that while exposure may cause infertility or spontaneous abortions, pregnancy may result in spite of DNA damage, and birth defects and childhood cancers may ensue.4 Pharmacists may refer patients to a useful resource from the National Institute for Occupational Safety and Health (NIOSH)—How Can Workers Be Protected From Reproductive Hazards?—as an integral part of an individual’s reproductive healthcare plan.14 Through increased awareness and education of the prospective father, male preconception care provides an opportunity to improve sperm quality, since damaged sperm can be replaced (new sperm is made every 42-76 days) within 3 months of mitigated exposures.4 Frey et al note that specialty care is provided by either a urologist or reproductive endocrinologist with an interest in andrology.4

Pharmacists should note that male preconception care is comprehensive in nature (i.e., for disease prevention and detection); routine screening for a number of conditions, such as hypertension, obesity, hyperlipidemia, diabetes mellitus, coronary heart disease, and mental health disorders is required; screening for testicular cancer in young men or prostate cancer and colorectal cancer in men aged >50 years is also recommended.4,15 Importantly, chronic inflammation, which can be caused by untreated infections (e.g., periodontal disease, sexually transmitted infections) can induce oxidative damage to sperm. Of note, family history and other conditions for which an individual may be at increased risk (e.g., cardiovascular disease, genetic risks) should not be overlooked.
Medications That Affect Sperm
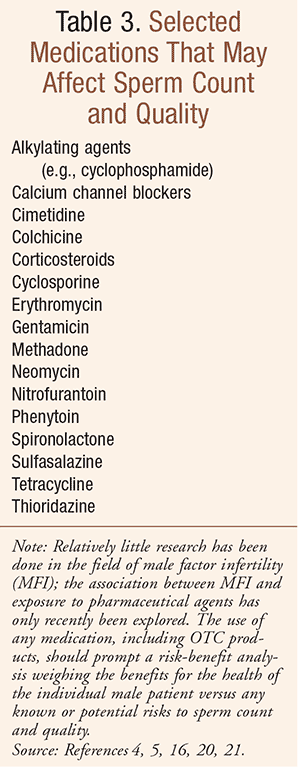
A variety of medications can affect sperm count and quality, including specific antihypertensive agents and antibiotics, among many others (see TABLE 3). A thorough medication history, including an individual’s past and current medication use—including prescription, nonprescription, and herbal products—should be reviewed.16 Of note, the use of any medication should prompt a risk-benefit analysis weighing the benefits for the health of the individual male patient versus any known or potential risks to sperm count and quality.16
Conclusion
While men have the advantage of lifelong fertility, proper preconception care for reproductive life planning is recommended. This should include a thorough and comprehensive evaluation of the medication regimen and involve patient preferences and participation in therapeutic outcomes.
REFERENCES
1. Wegner MN, Landry E, Wilkinson D, et al. Men as partners in reproductive health: from issues to action. Int Fam Plan Perspect. 1998;24:38-42.
2. Johnson K, Posner SF, Bierman J, et al. Recommendations to improve preconception health and health care—United States. MMWR Recomm Rep. 2006;55:1-23.
3. Sternberg P, Hubley J. Evaluating men’s involvement as a strategy in sexual and reproductive health promotion. Health Promot Int. 2004;19:389-396.
4. Frey KA, Navarro SM, Lu MC. The clinical content of preconception care: preconception care for men. Am J Obstet & Gynecol. 2008;199(6):S389-S395.
5. Brezina PR, Yunus FN, Zhao Y. Effects of pharmaceutical medications on male fertility. J Reprod Infertil. 2012;13(1):3-11.
6. Preconception care policy brief pdf. World Health Organization. February 2012. www.who.int/maternal_child_adolescent/documents/concensus_preconception_care/en/. Accessed April 18, 2016.
7. World Health Organization. February 6-7 2012 Meeting Report. Meeting to develop a global consensus on preconception care to reduce maternal and childhood mortality and morbidity. Geneva, Switzerland, 2013.http://apps.who.int/iris/handle/10665/78067. Accessed April 22, 2016.
8. Gray N. Father time: children with older dads at greater risk for mental illness. August 29, 2011. www.scientificamerican.com/article/children-with-older-dads-at-greater-mental-illness-risk/. Accessed April 1, 2016.
9. Sipos A, Rasmussen F, Harrison G, et al. Paternal age and schizophrenia: a population based cohort study. BMJ. 2004;329:1070.
10. Malaspina D, Harlap S, Fennig S. et al. Advancing paternal age and the risk of schizophrenia. Arch Gen Psychiatry. 2001;58:361-367.
11. Medscape. Complex relationship between parental age, autism risk. June 11, 2015. www.medscape.com/viewarticle/846265. Accessed April 12, 2016.
12. Sandin S, Schendel D, Magnusson P, et al. Autism risk associated with parental age and with increasing difference in age between the parents. Mol Psychiatry. 2016;21:693-700.
13. D’Onofrio BM, Rickert ME, Frans E, et al. Paternal age at childbearing and offspring psychiatric and academic morbidity. JAMA Psychiatry. 2014;71(4):432-438.
14. CDC. National Institute for Occupational Safety and Health (NIOSH). The effects of workplace hazards on male reproductive health. DHHS (NIOSH) Publication Number 96-132. Last reviewed and updated: June 6, 2014. www.cdc.gov/niosh/docs/96-132/. Accessed April 12, 2016.
15. U.S. Preventive Services Task Force. Guide to clinical preventive services, 2014: recommendations of the U.S. Preventive Services Task Force. AHRQ Pub. No. 14-05158. Rockville, MD: Agency for Healthcare Research and Quality. U.S. Department of Health and Human Services. May 2014. www.ahrq.gov/professionals/clinicians-providers/guidelines-recommendations/guide/index.html. Accessed April 19, 2016.
16. CDC. Preconception health and health care: exposures. www.cdc.gov/preconception/careformen/exposures.html. Accessed April 11, 2016.
17. Marinelli D, Gaspari L, Pedotti P, and Taioli E. Mini-review of studies on the effect of smoking and drinking habits on semen parameters. Int J Hyg Environ Health. 2004;207:185-192.
18. Muthusami KR, Chinnaswamy P. Effect of chronic alcoholism on male fertility hormones and semen quality. Fertil Steril. 2005;84:919-924.
19. Jensen TK, Bonde JP, Joffe M. The influence of occupational exposure on male reproductive function. Occup Med (Lond). 2006;56:544-553.
20. Epocrates.com. Epocrates Plus Version 15.12.1. Updated March 23, 2016. Accessed April 22, 2016.
21. Semla TP, Beizer JL, Higbee MD. Geriatric Dosage Handbook. 20th ed. Hudson, OH: Lexicomp; 2015:1402-1407.
To comment on this article, contact rdavidson@uspharmacist.com.





