US Pharm. 2014;39(4):HS18-HS24.
There are more than 1 million people with HIV in the United States, with nearly 16% of them unaware that they are infected.1 Due to the nature of their work, healthcare personnel (HCP) have the potential for exposure to various types of infectious organisms. The Occupational Safety and Health Administration (OSHA) estimates that over 5.6 million workers are at risk for occupational exposure to bloodborne pathogens, including HIV.2 Guidelines for the management of occupational exposures to HIV and recommendations for post-exposure prophylaxis were recently updated by the U.S. Public Health Service Working Group.3 This article focuses on these guidelines and reviews the epidemiology of occupational exposure, risks for transmission of HIV in the workplace, prevention strategies, and current recommendations for treatment and monitoring.
Epidemiology
The updated guidelines define HCP as all paid and unpaid individuals working in a healthcare setting who have the potential for exposure to infectious body substances or contaminated supplies, equipment, or surfaces.3 There have been substantial improvements in safety devices and the use of barriers to help prevent HCP from accidental exposure to bloodborne pathogens, so occupational transmission of HIV is considered to be extremely rare in the U.S.3,4 There have been 57 documented transmissions and 143 possible transmissions of HIV to HCP in the U.S. as of 2010.4
It is important to note that reporting and tracking of occupational HIV transmission is a passive process, and that it is difficult to document an acquired case if the exposure is not reported at the time the injury occurred.5 Therefore, although the reported number of cases is low and there have not been any confirmed or possible cases reported since 1999 and 2009, respectively, there is concern of underreporting.4-6
Transmission
HCP are at risk for acquiring HIV from exposure to blood or other potentially infectious body fluids via a percutaneous injury (needlestick or cut with a sharp object) or by a splash exposure of a mucous membrane or nonintact skin to blood, tissue, or other body fluids that are potentially infectious (TABLE 1).3,7

The average risk for transmission after a percutaneous exposure to HIV-infected blood is estimated to be 0.3%, and the average risk for transmission after a mucous membrane exposure is estimated to be 0.09%.3,5 It should be noted that the percutaneous risk estimate represents an average of several different types of exposures from patients in various stages of infection with HIV and that occupational transmission has only been documented after exposure to blood or visibly bloody fluids.5 Therefore, there is a lower risk for transmission from exposure to other fluids or tissues that do not contain visible blood.3,5 The average risk for transmission from nonintact skin exposure has not been specifically quantified but is believed to be less than that for mucous membrane exposure.3,5
Multiple factors affect the risk of HIV transmission after occupational exposure, with HCP at an increased risk for infection in situations where there is exposure to a larger quantity of blood from the source patient (as indicated by a visibly contaminated needle, a procedure involving a needle placed directly into the source patient’s vein or artery, or a deep injury), or if the source patient is in a later stage of disease, as indicated by a high plasma titer of HIV or an advanced stage of illness.3,8 However, HIV transmission is possible even from a patient with an undetectable viral load, so appropriate follow-up testing and the need for treatment should be assessed even if the source patient does not have a detectable viral load.3
Prevention
The most important strategy for reducing the risk of occupational transmission of HIV is preventing exposures through the use of standard precautions.3 Both individual HCP and the institutions for which they work are responsible for ensuring that standard precautions are followed at all times, with HCP assuming that blood and body fluids from all patients are potentially infectious.3,4,9 Standard precautions include the use of infection-control measures such as the routine use of barriers (e.g., gloves, goggles, gowns, surgical masks, face shields) when contact with a patient’s blood or body fluids is likely.4,7,9 In order to prevent mucous membrane exposures, HCP should follow safe work practices such as keeping contaminated gloves or hands away from the face and positioning patients so that sprays and splatter are directed away from the face.9
In addition, HCP should follow appropriate hand hygiene measures, including immediately washing hands and skin surfaces after contact with blood or body fluids and carefully handling and disposing of sharp instruments during and especially after use.4,9 Safety devices should be used whenever possible, and all used syringes and sharp instruments should be placed into sharps containers for proper disposal.4
Treatment Guidelines
In September 2013, the U.S. Public Health Service released updated guidelines for managing occupational exposures to HIV and recommendations for postexposure prophylaxis (PEP).3 The guidelines continue to emphasize the use of primary prevention as the main strategy for safeguarding against occupational infections. In cases where an occupational exposure has occurred, the guidelines stress prompt management, use of a PEP regimen that is optimally tolerated with no drug interactions, expert consultation, rapid testing of source patients, and appropriate counseling and monitoring of exposed HCP. The guidelines recommend that all institutions have protocols for the management of occupational exposures, which should be distributed and readily available to healthcare providers who may be asked to manage these situations.3
A main change in the updated guidelines is that they simplify the selection of a PEP regimen by eliminating the need to assess the level of risk associated with an individual exposure.3 The clinician should be aware that the guidelines apply to situations in which HCP have been exposed to a source person who has confirmed or reasonable suspicion for HIV infection, and that no HIV medication currently has an approved indication by the FDA for use as PEP.3
Principles of Treatment
Initiation of Therapy: Factors for the clinician to consider when deciding to initiate PEP include the risk of HIV transmission from the type of exposure and the HIV status of the source patient. If the HIV status of the source patient is not known, rapid testing may be performed.3,10 No matter what type of HIV testing is used, administration of PEP should not be delayed while waiting for results, as PEP is most effective when started as soon as possible after an exposure. Occupational exposure should be considered an urgent medical need, with PEP ideally started within hours.3
In order to ensure that HCP will be adherent with a full course of therapy, potential side effects of agents should be discussed, and a regimen should be selected that is most likely to be tolerated. In addition, a PEP regimen should have minimal toxicity, a convenient dosing schedule, and no drug interactions with the individual’s OTC or prescription medications. Agents to alleviate anticipated side effects (such as antidiarrheals or antiemetics) should be preemptively prescribed in order to ensure adherence.3
Also at the initial evaluation meeting, HCP should be counseled to use precautions. These include the use of barrier contraception and avoidance of blood donation, pregnancy, and breastfeeding, especially during the first 6 to 12 weeks after exposure, in order to prevent secondary transmission of HIV.3
Duration of Therapy
The updated guidelines recommend that a three-drug (or more) regimen be routinely used as PEP, with all HCP receiving a full 4-week course of therapy. One exception to the duration of therapy is that if the HIV status of the source patient is unknown at time of exposure and he or she is later determined to be uninfected, PEP therapy may be discontinued.3
Selection of Specific Agents
The three-drug regimen that is recommended as first-line therapy for PEP in the updated guidelines is emtricitabine/tenofovir (available coformulated as Truvada) plus raltegravir. These three agents were selected for their favorable side-effect and drug-interaction profile, tolerability, and simple dosing schedule.3 Dosing, more common or serious side effects, select drug interactions, and therapeutic considerations for use of each of the preferred first-line agents are included in TABLE 2.3,11
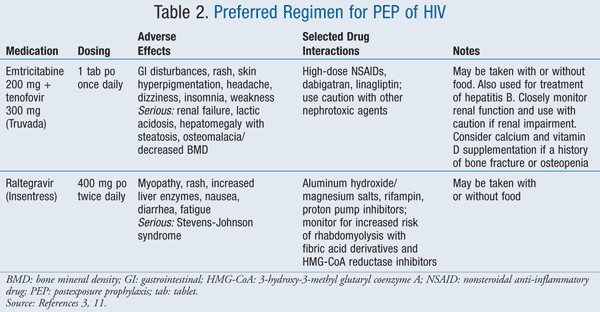
If the source patient has known or suspected drug resistance to any of the first-line agents, then alternative agents to which the virus is not resistant should be used, and an expert in infectious diseases or antiretroviral therapy should be consulted. Additional resources for clinicians include the National Clinicians’ Post-Exposure Prophylaxis Hotline (PEPline), which offers advice on the management of any occupational exposure. Information from PEPline is available online, and clinicians can be reached for consultation at 1-888-448-4911 between the hours of 9 AM and 2 AM EST.3,12
For situations in which a first-line agent cannot be used or is not tolerated, there are several preferred alternative regimens that are included in the updated guidelines (TABLE 3).3 These agents are listed in order of preference, and the clinician should choose one drug or drug pair from the left column with one pair of nucleoside/nucleotide reverse transcriptase inhibitors from the right column. Another option is for the clinician to use the combination product of elvitegravir, cobicistat, tenofovir, and emtricitabine (available coformulated as Stribild) as a fixed-dose formulation.

Monitoring and Follow-up
All HCP who have had an occupational exposure (whether or not they are receiving PEP) should be reevaluated within 72 hours of the incident. The purpose of this visit is to provide another opportunity for HCP to ask questions and for the provider to ensure that they understand the risks for infection, as well as to discuss any potential or experienced side effects of PEP, reiterate the importance of adherence, and reinforce precautions in order to prevent secondary transmission of infection. Also, any additional information about the source patient should be known by this time, and the need for continued treatment with PEP can be assessed. Psychological counseling should also be available and routinely included as part of therapy for HCP who have had an occupational exposure.3
HCP should have baseline HIV testing at the time of occupational exposure and follow-up testing at 6 weeks, 12 weeks, and 6 months later. An exception to this is that if the individual received baseline testing using the newer HIV p24 antigen-HIV antibody test, follow-up testing is only required at 6 weeks and 4 months after exposure.3
If PEP is used, HCP should be monitored for drug toxicity, with laboratory testing for complete blood counts (CBC) and renal and hepatic function performed at baseline and 2 weeks after exposure, with additional testing as needed. The importance of adherence should be emphasized, and information about potential drug interactions and side effects of the medication regimen should be provided. If it is determined that a healthcare provider has acquired HIV infection through an occupational exposure, that person should be referred to an HIV specialist for treatment. Cases should be reported to the state health department and to the CDC’s “Cases of Public Health Importance” (CDC COPHI) coordinator at 404-639-2050.3,6
Conclusion
Pharmacists can play an integral role in the management of occupational exposures by educating HCP about the risks of HIV transmission in the workplace and the appropriate use of PEP. Pharmacists should work with clinicians to select the most suitable PEP regimen for a particular patient and can work with their healthcare facilities to ensure that protocols are in place to handle these situations in a timely manner. Pharmacists can assist by ensuring that PEP medications are readily available and should provide counseling on behavioral modifications, side effects, and strategies to alleviate adverse effects if symptoms occur. Pharmacists are in a key position to actively work with exposed HCP and providers to ensure the proper management of occupational exposures in order to prevent HIV transmission.
REFERENCES
1. CDC. HIV in the United States: at a glance. www.cdc.gov/hiv/statistics/basics/ataglance.html. Accessed January 9, 2014.
2. U.S. Department of Labor. Healthcare wide
hazards—needlestick/sharps injuries.
www.osha.gov/SLTC/etools/hospital/hazards/sharps/sharps.html. Accessed
January 9, 2014.
3. Kuhar D, Henderson D, Struble K, et al; US Public
Health Service Working Group. Updated U.S. Public Health Service
guidelines for the management of occupational exposures to human
immunodeficiency virus and recommendations for postexposure prophylaxis.
Infect Control Hosp Epidemiol. 2013;34:875-892.
4. CDC. Occupational HIV transmission and prevention among
health care workers. www.cdc.gov/hiv/risk/other/occupational.html.
Accessed January 9, 2014.
5. Bell D. Occupational risk of human immunodeficiency virus infection in healthcare workers: an overview. Am J Med. 1997;102:9-15.
6. CDC. Surveillance of occupationally acquired HIV/AIDS
in healthcare personnel, as of December 2010.
www.cdc.gov/HAI/organisms/hiv/Surveillance-Occupationally-Acquired-HIV-AIDS.html.
Accessed January 9, 2014.
7. Wu H, Dhara R, Czarkowski A. Occupational exposure to HIV. In: CDC Health Information for International Travel 2014: The Yellow Book.
http://wwwnc.cdc.gov/travel/yellowbook/2014/chapter-2-the-pre-travel-consultation/occupational-exposure-to-hiv.
Accessed January 9, 2014.
8. Cardo D, Culver D, Ciesielski C, et al. A case-control
study of HIV seroconversion in health care workers after percutaneous
exposure. New Engl J Med. 1997;337:1485-1490.
9. Siegel J, Rhinehart E, Jackson M, et al. 2007 guideline
for isolation precautions: preventing transmission of infectious agents
in health care settings. Am J Infect Control. 2007;35(suppl 2):S65-S164.
10. Masciotra S, McDougal J, Feldman J, et al. Evaluation
of an alternative HIV diagnostic algorithm using specimens from
seroconversion panels and persons with established HIV infections. J Clin Virol. 2011;52(suppl 1):S17-S22.
11. Lexi-Comp Online [Internet database]. Hudson, OH: Lexi-Comp, Inc; 2014. www.lexi.com. Accessed January 9, 2014.
12. Clinicians’ Consultation Center. Post-Exposure
Prophylaxis (PEP).
http://nccc.ucsf.edu/clinician-consultation/post-exposure-prophylaxis-pep/.
Accessed March 24, 2014.
To comment on this article, contact rdavidson@uspharmacist.com.





