US Pharm. 2023;48(7):HS-7-HS-10
ABSTRACT: According to CDC estimates, one in 13 people in the United States suffers from asthma. Asthma is a condition characterized by bronchoconstriction and inflammation of a person’s airway. With proper education, medication management, and adherence, asthma is reversible, and patients are able to live active lives with minimal symptoms. Treatment is determined by differentiating between mild-to-moderate, severe, and life-threatening exacerbations, as well as the management recommended by the 2022 Global Initiative for Asthma guidelines. Nirmatrelvir/ritonavir (Paxlovid) and molnupiravir are antivirals used to treat mild-or-moderate COVID-19 in patients with asthma who are likely to become seriously ill due to comorbid conditions.
Asthma is a chronic condition that affects a person’s airways and lungs. In a patient with asthma, the airways become inflamed and narrowed, making it difficult for them to breathe. The disease affects people of all ages and often starts in childhood. Examples of asthma triggers include pollen, dust, animal fur and feathers, exercise, viral infections (such as COVID-19), and cold air, among others.1 Underappreciated triggers include laughter, coughing, and some medications.1 Some medications that can lead to asthma exacerbation include nonselective beta-blockers (e.g., propranolol, carvedilol, labetalol) and nonsteroidal anti-inflammatory drugs.2
Common symptoms are coughing, shortness of breath, wheezing, chest tightness, and chest pain.3 Symptoms are intermittent and often worse at night or during exercise. Although there is no cure for asthma, management includes monitoring for signs and symptoms, avoiding triggers, and treating with medications such as bronchodilators and steroids. Bronchodilators work to relax the lung muscles and widen the bronchi to open up the air passages and relieve symptoms, while steroids reduce inflammation in the airways to improve symptoms and reduce the risk of severe asthma attacks that can lead to death.4 Left untreated, asthma can cause sleep disturbances, tiredness during the day, and poor concentration. In some cases, symptoms can become so severe that patients may need to receive emergency medical attention.
There is no single, direct cause of asthma, but there are many factors that have been linked to an increased risk of development. Individuals are more likely to develop asthma if they have a family history (especially a parent or sibling) of asthma, have an allergic condition such as eczema or rhinitis, live in an area with increased asthma prevalence, were born with low birth weight or prematurely, were exposed to tobacco smoke or other sources of air pollution, were exposed to many viral respiratory infections or environmental allergens, or are overweight or obese.4 To diagnose asthma, a healthcare provider will review a patient’s medical history, including information about the patient’s parents and siblings. The patient’s symptoms will be reviewed, as well as history of allergies, eczema, and other lung diseases. If asthma is suspected, a spirometry test will be ordered that measures the airflow through the lungs. Peak expiratory flow rate (PEFR) is a common spirometry test used in the diagnosis of asthma. PEFR measures the volume of air that is forcefully expelled from the patient’s lungs in one quick expiration. Chest x-rays, blood tests, and skin tests may also be used to diagnose asthma.
The goals of therapy for asthma management are to optimize the control of asthma symptoms and reduce the risk of asthma exacerbations, while also minimizing adverse effects from medications. A person with well-controlled asthma is expected to be able to participate in work, school, sports, and other activities of daily living without limitation due to difficulty breathing. The four essential components of asthma management include patient education, minimizing exposure to triggers, monitoring any changes in symptoms or lung function, and pharmacologic therapy.6
2022 GINA Guidelines Update
In 2022, the Global Initiative for Asthma (GINA) guidelines were updated to improve asthma prevention and management. The updates impacted first-line recommendations for inhaled controller and reliever medications. For over 50 years, short-acting beta-agonists (SABAs) were the first-line treatments for asthma; however, this class is no longer recommended as monotherapy. In adults and adolescents aged 12 years and older, inhaled corticosteroids (ICS) plus formoterol is the preferred treatment. Formoterol is a long-acting beta-agonist (LABA) that relaxes bronchial smooth muscle by acting on beta-2 receptors with little effect on heart rate. Formoterol induces significant bronchodil-tion with effects starting within minutes of inhalation of a therapeutic dose and persisting for up to 12 hours. This guideline recommendation reflects the SYGMA studies, which prove that the use of as-needed ICS + formoterol reduces the risk of exacerbations compared with a SABA alone. The results of these trials met their primary endpoint, demonstrating that as-needed budesonide-formoterol is superior in asthma symptom control to as-needed SABA (34.4% vs. 31.1%).7
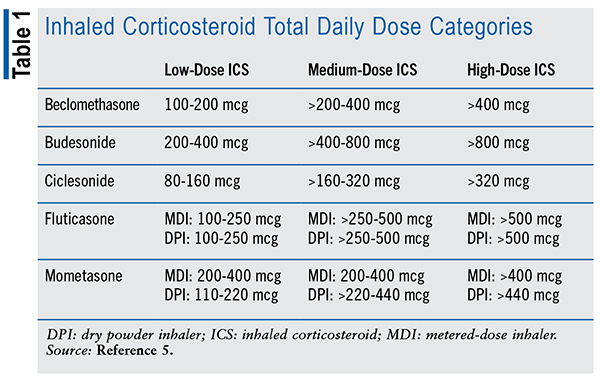
The 2022 GINA guidelines also recommend that each asthma treatment plan be individualized based on the frequency of symptoms and avoidance of triggers. The frequency of symptoms is assessed by how often the patient uses their rescue inhaler and if they have symptoms waking them during the night. Personalized asthma management should be assessed and adjusted using the step-up method. Steps 1 and 2 are defined as using low-dose ICS-formoterol, step 3 is defined as low-dose maintenance ICS-formoterol, step 4 is defined as medium-dose maintenance ICS-formoterol, and step 5 includes the recommendations from step 4 plus add-on long-acting muscarinic agonist. All steps include using the same low-dose ICS-formoterol inhaler as an as-needed symptom reliever, in addition to its daily use for maintenance therapy. TABLE 1 shows examples of ICS products and total daily doses that fall under each step. Every patient should be provided with their individual written asthma action plan, with instructions on what to do when their asthma symptoms worsen.
Managing Acute Asthma Exacerbations
Acute asthma exacerbations occur when a patient experiences episodes of worsening respiratory symptoms and lung function. Asthma exacerbation triggers vary from person to person and can be unpredictable. Triggers can result in serious adverse events in all patients with asthma, despite the severity. Comorbidities, such as infections, allergic rhinitis, obstructive sleep apnea, obesity, stress, and depression, can also result in acute asthma exacerbations. Signs and symptoms that warrant a visit to the emergency department include immense shortness of breath (unable to walk and talk), unable to complete usual daily activities, persistent or worsening symptoms after 24 hours, unimproved symptoms despite the use of rescue medications, blue lips or fingernails, or a PEFR <50% of personal best.6
A stepwise approach should be used to guide providers through the management of asthma exacerbations. When a patient is present in a healthcare setting, the first step to identifying an asthma exacerbation is to assess the patient’s symptoms, severity, and risk factors. TABLE 2 differentiates the symptoms between mild-to-moderate, severe, and life-threatening exacerbations, as well as the management recommended by the 2022 GINA guidelines.5,6,8,9

Currently, SABAs such as albuterol are the most commonly prescribed bronchodilators; however, formoterol has been found to have similar efficacy and safety. A systematic review consisting of nine randomized, controlled trials found no significant difference between the use of high-dose formoterol and SABAs for the management of acute asthma in the emergency department setting. Patients’ symptoms were assessed 30 to 40 minutes after the first administration of the study drug, 60 to 90 minutes after the last dose, and at the end of treatment. Additionally, there was no significant difference in potassium serum level, heart rate, QT interval, and hospitalization rate between the two study groups.10 The systemic review suggests that it is appropriate to use either a SABA or high-dose formoterol in the management of acute asthma.
Mild-to-moderate exacerbations with worsening symptoms after initial treatment warrant a patient transfer to an acute-care facility. Severe and life-threatening exacerbations are a medical emergency, and the patient should be transferred to an acute-care facility as soon as possible. Patients with severe exacerbations with worsening or persistent symptoms should also be assessed for alternative diagnoses that may contribute to these symptoms, such as pneumonia, heart failure, and chronic obstructive pulmonary disease (COPD).6
Azithromycin, a macrolide antibiotic, is known to have anti-inflammatory properties attributed to its action on macrophages. Banjanac et al showed that azithromycin inhibited the synthesis of all of the eicosanoids produced downstream of the COX pathway.11 The AZALEA trial, which looked at azithromycin use for acute asthma exacerbations, found poor evidence of benefit in patient outcomes for the use of empiric antibiotics. This was a randomized, double-blind, placebo-controlled clinical trial that took place in the United Kingdom. Inclusion criteria included adults with more than a 6-month history of asthma who presented with an asthma exacerbation within 48 hours of presentation. They were given azithromycin 500 mg daily or a matched placebo for 3 days. The primary outcome was a diary card symptom score 10 days after treatment. The mean (SD) symptoms were assessed on a scale of 0 to 6 (from 0 = no symptoms to 6 = severe symptoms). Symptoms scored include wheezing, breathlessness, and coughing. Results of the primary outcome were asthma symptom scores 4.14 (1.38) at exacerbation, 2.09 (1.71) at 10 days for the azithromycin group and 4.18 (1.48) at exacerbation, and 2.20 (1.51) for the placebo group. The AZALEA trial concluded that there is no statistically or clinically significant benefit in using azithromycin for the treatment of acute asthma exacerbations.12
Prior to discharge from an acute-care setting, patients should be monitored for their response to treatment or lack thereof. A good response to management is generally defined as no longer needing SABA, PEFR of >60% to 80% of personal best or predicted, and oxygen saturation of >94% on room air. Additionally, the patient’s home medication regimen should be assessed and altered as appropriate. Every patient should be discharged with reliever and controller therapy, a 5- to 7-day course of oral corticosteroids, education, and a follow-up appointment within 2 to 7 days. Education should also be provided on how to prevent future exacerbations. Ways to avert exacerbations include avoiding triggers to the best of their ability, practicing good adherence, and practicing proper inhaler techniques.
COVID-19 in Asthma Patients
In 2020 and 2021 during the peak of the COVID-19 pandemic, many countries saw a decrease in the number of asthma exacerbations. Although the exact reason is unknown, it is thought that certain public health measures, such as social distancing and increased handwashing, may have had an impact on this. Viral infections are one of the triggers of asthma exacerbations, and with many people implementing these public health measures, there was an overall decrease in the prevalence of respiratory-related illnesses as a whole.
According to the 2022 GINA guidelines, people with well-controlled or mild-to-moderate asthma are not at an increased risk of acquiring COVID-19 or COVID-19-related death. Although the risk of COVID-19-related death was not more likely in patients with well-controlled asthma, studies have shown that those who recently took oral corticosteroids (OCS) were at an increased risk of death.6 To avoid severe infection, it is crucial that patients with asthma maintain good symptom control, reduce their risk of exacerbations, and minimize their need for OCS.
A national multicenter, prospective cohort study showed that the use of an ICS in maintenance asthma therapy may be protective against developing severe COVID-19 symptoms. Patients with asthma, COPD, or both who were admitted to a hospital with a positive COVID-19 test were eligible to be included in this study. Patients were stratified based on age (<16 years, 16-49 years, and ≥50 years). Included were 75,463 patients, and patients with asthma were significantly more likely than those without asthma to receive critical care (patients aged 16-49 years: adjusted odds ratio [OR] 1.20 [95% CI 1.05-1.37]; P = .0080; patients aged ≥50 years: adjusted OR 1.17 [1.08-1.27]; P <.0001). Patients aged 50 years and older who used ICS within 2 weeks of hospital admission were associated with decreased mortality in those with asthma compared with those without an underlying respiratory condition.13
When COVID-19 is suspected or confirmed in a patient with asthma, the 2022 GINA guidelines recommend avoiding nebulizers whenever possible to reduce the risk of viral transmission to healthcare professionals and other personnel. It is preferred that bronchodilators be administered via the use of a metered-dose inhaler (MDI) and spacer in all patients with asthma and COVID-19, except for those with acute, severe asthma. A mask may be added to the spacer if needed to improve the amount of medication inhaled. Additionally, the use of in-line filters may be used in spirometry or peak flow measurements to minimize transmission. Healthcare providers and caregivers should follow local guidance on hygiene strategies and the use of personal protective equipment.
Nirmatrelvir/ritonavir (Paxlovid) and molnupiravir are antivirals used to treat mild-or-moderate COVID-19 in those who are likely to become seriously ill due to comorbid conditions. Examples of comorbid conditions include asthma, COPD, heart disease, or a compromised immune system. Nirmatrelvir/ritonavir has several drug-drug interactions. Those related to asthma management include LABAs and inhaled/systemic corticosteroids. LABAs, particularly salmeterol, may need to be withheld due to the increased risk of cardiovascular adverse events. Corticosteroids taken with nirmatrelvir/ritonavir may put the patient at an increased risk for adrenal suppression or Cushing syndrome.14 The short course (5-day therapy) of nirmatrelvir/ritonavir may outweigh the potential risks; however, providers should weigh the risks versus benefits for each patient with asthma and an active COVID-19 infection. Medications used for hospitalized patients for the treatment of COVID-19 include baricitinib, dexamethasone, remdesivir, sarilumab, tocilizumab, and tofacitinib citrate. None of these medications interact with the recommended asthma medications budesonide-formoterol or albuterol.
Role of the Pharmacist
Pharmacists play a vital role in providing patients with education on medication use and adverse effects. It is critical that patients with asthma are counseled on the proper use of inhalers to reduce the risk of exacerbations. Spacers should be recommended to all patients with an MDI who have dexterity issues. The sequence of inhalers is important, and pharmacists should counsel patients with multiple inhalers on the order of use. Because bronchodilators work faster than ICS, they should be given first to dilate the bronchi, which allows the ICS to travel deeper into the lungs. Pharmacists should advise patients to wait 60 seconds between using each inhaler. Additionally, patients with asthma should be educated on the side effects of overusing their rescue inhalers. Using a SABA too frequently can result in nervousness, tremors, tachycardia, and heart palpitations. An increase in the use of a patient’s rescue inhaler is a sign that their maintenance therapy may need to be adjusted. Patients using ICS should be counseled to rinse their mouth with water and spit it out to prevent a fungal infection known as thrush.
Pharmacists also play a critical role in administering and counseling patients on vaccines. The CDC recommends that all patients 6 months and older, whether they have asthma or not, be fully vaccinated against COVID-19, including the two- or three-dose primary series and a booster. The CDC also recommends that all patients aged older than 6 months receive an annual influenza vaccine, especially if that person also suffers from asthma. If a patient is not up-to-date with their vaccines or has any questions about which vaccines they should receive, a pharmacist is able to educate, administer, and counsel patients on possible side effects that they may experience after receiving a vaccine.
Conclusion
First-line therapy for asthma, despite disease severity, is an ICS-formoterol inhaler. Dosing and frequency should be adjusted as appropriate for an individual patient’s needs. A short course of OCS should be used when indicated for severe asthma exacerbations. Every patient with asthma should have an individualized written asthma action plan advising them to increase controller and reliever medications when symptoms worsen.
According to the CDC, patients with moderate-to-severe or uncontrolled asthma are more likely to be hospitalized from COVID-19. Studies have shown that patients with well-controlled asthma or mild-to-moderate asthma are not at an increased risk of being hospitalized due to COVID-19. All patients, especially those with underlying conditions such as asthma, should make sure that they are up to date on all vaccines, especially the COVID-19 vaccine series plus booster and their annual influenza vaccine. Patients can utilize pharmacists to educate them on the proper technique of their inhalers, reinforce adherence to medications, administer any vaccines they may need, and counsel them on the potential side effects or drug interactions that may exist in their drug regimens.
REFERENCES
1. National Heart, Lung, and Blood Institute. What is asthma? 2022. www.nhlbi.nih.gov/health/asthma. Accessed April 9, 2023.
2. Bombatch C, Brian S, Colley P, et al, eds. Asthma. In: 2023 NAPLEX Course Book. Chap 41. El Segundo, CA: RxPrep; 2023:571-587.
3. Asthma & Allergy Foundation of America. Asthma 2023. https://aafa.org/asthma/. Accessed April 9, 2023.
4. World Health Organization. Asthma. 2022. www.who.int/newsroom/fact-sheets/detail/asthma. Accessed April 9, 2023.
5. GINA. 2022 Gina Main Report–Global Initiative for Asthma. 2023. https://ginasthma.org/gina-reports/. Accessed April 9, 2023.
6. Fanta CH. An overview of asthma management. UpToDate. 2023. www.uptodate.com/contents/an-overview-of-asthma-management#H1. Accessed April 9, 2023.
7. O’Byrne PM, FitzGerald JM, Bateman ED, et al. Effect of a single day of increased as-needed budesonide-formoterol use on short-term risk of severe exacerbations in patients with mild asthma: a post-hoc analysis of the SYGMA 1 study. Lancet Respir Med. 2021;9:149-158.
8. World Health Organization. WHO coronavirus (COVID-19) dashboard. https://covid19.who.int/. Accessed April 9, 2023.
9. CDC. Basics of COVID-19. www.cdc.gov/coronavirus/2019-ncov/your-health/about-covid-19/basics-covid-19.html. Accessed April 10, 2023.
10. Rodrigo GJ, Neffen H, Colodenco FD, Castro-Rodriguez JA. Formoterol for acute asthma in the emergency department: a systematic review with meta-analysis. Ann Allergy Asthma Immunol. 2010;104(3):247-252.
11. Banjanac M, Munić Kos V, Nujić K. Anti-inflammatory mechanism of action of azithromycin in LPS-stimulated J774A.1 cells. Pharmacol Res. 2012;66(4):357-362.
12. Johnston SL, Szigeti M, Cross M, et al. AZALEA Trial Team. Azithromycin for acute exacerbations of asthma: the AZALEA randomized clinical trial. JAMA Intern Med. 2016;176(11):1630-1637. Erratum in: JAMA Intern Med. 2018;178(7):1003.
13. Bloom CI, Drake TM, Docherty AB, et al. ISARIC investigators. Risk of adverse outcomes in patients with underlying respiratory conditions admitted to hospital with COVID-19: a national, multicentre prospective cohort study using the ISARIC WHO Clinical Characterisation Protocol UK. Lancet Respir Med. 2021;9(7):699-711.
14. Allergy & Asthma Network. COVID antiviral pill news and updates. https://allergyasthmanetwork.org/news/covid-pill/. Accessed April 9, 2023.
The content contained in this article is for informational purposes only. The content is not intended to be a substitute for professional advice. Reliance on any information provided in this article is solely at your own risk.
To comment on this article, contact rdavidson@uspharmacist.com.






