US Pharm. 2009;34(1):30-38.
Peripheral neuropathy (PN) is one of the most common complications of diabetes.1 Up to 50% of elderly patients with type 2 diabetes mellitus (T2DM) have diabetes-related PN.2,3 Painful diabetic peripheral neuropathy (PDPN) accounts for 10% to 20% of PN cases, and these patients describe their pain as sharp, throbbing, aching, or burning; as being stabbed by tiny knives or bitten by hundreds of spiders or fire ants on their lower legs and feet; and as keeping them awake all night long.1-3 This article discusses the management of PDPN, focusing on the elderly.
Background
The most common form of PDPN is chronic or persistent sensorimotor neuropathy, which is abnormalities in feeling and movement due to nerve damage. PN is slow in onset and may be present in up 10% of patients at the time of T2DM diagnosis. It is often accompanied by autonomic dysfunction, and as it progresses, other diabetes-associated foot problems including foot ulceration and Charcot's foot may also progress.2,3
A study in 2003 looked at health care costs of PN in the United States and found the cost of treating PN at that time to be nearly $11 billion. Elderly patients with T2DM comprised 92.5% of the study population and accounted for 93.1% of the total costs ($10.2 billion).4
The prevalence of chronic PDPN increases with both a person's age and duration of diabetes and is more common in those whose glycemic control was previously suboptimal.2,3
Management
Prevention
Strategies that have been proposed to prevent and maintain PDPN include strict glycemic control, controlling hyperlipidemia and hypertension, smoking cessation, daily low-dose aspirin, moderate alcohol consumption, and maintaining good foot care.5 In the Diabetes Control and Complications Trial (DCCT), strict glycemic control decreased the incidence of PDPN in patients with type 1 diabetes mellitus (T1DM) and slowed its progression.6 Interestingly, PDPN can occur even when blood glucose levels are well controlled.7
Data from a small number of trials assessing the treatment of PDPN for patients with T2DM are not as strong as DCCT data, but the results suggest that those with T2DM should follow the same guidelines as those with T1DM to slow or prevent the progression of PDPN.5,7
Treatment
In addition to strict glycemic control, pain medications are needed for the treatment of PDPN. Antidepressants and antiepileptics are the main drugs that have been studied and used to treat PDPN. Carbamazepine (Tegretol) and amitriptyline (Elavil) were the first drugs studied in PDPN, but the only drugs approved are duloxetine (Cymbalta) and pregabalin (Lyrica).8-11
Antidepressants: Tricyclic antidepressants (TCAs) have been the most studied drug class for PDPN. They are norepinephrine (NE) and serotonin (5-HT) reuptake inhibitors. The tertiary amines--amitriptyline and imipramine--are nonselective and provide balanced inhibition of both neurotransmitters. The secondary amines, nortriptyline and desipramine, provide more reuptake of norepinephrine. Both secondary and tertiary amines appear to be effective in treating PDPN.12 It is not known if the reuptake inhibition of both NE and 5-HT is the analgesic mechanism of these drugs, but it is thought that TCAs decrease PDPN by reducing the perception of pain centrally.7 In studies, the efficacy of drugs for the treatment of PDPN was assessed by calculating the number needed to treat (NNT) to obtain one patient with 50% pain relief. The NNT for both amitriptyline and imipramine was 2.1 (95% confidence interval [CI], 1.8-2.6) and 2.5 (95% CI, 1.9-3.6) for desipramine. The NNT for nortriptyline and doxepin has not been established.5
TCAs have a high incidence of adverse effects, especially anticholinergic effects, and warnings for their use include the presence of arrhythmias, myocardial infarction (MI), glaucoma, and urinary retention. They have also been associated with syndrome of inappropriate antidiuretic hormone secretion, especially in the elderly.13 The secondary amines cause fewer anticholinergic side effects compared to the tertiary amines.12 Contraindications include recent MI and concurrent use of monoamine oxidase inhibitors (MAOIs). The tertiary amines are metabolized by and inhibit more CYP450 enzymes than the secondary amines and therefore have a greater potential for drug interactions. Monitoring parameters include blood pressure, heart rate, electrocardiography, and weight gain.13
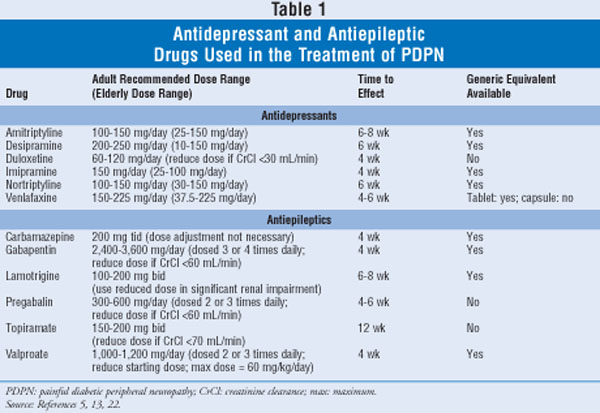
See TABLE 1 for recommended doses, time to effect, and generic availability of antidepressants used in the treatment of PDPN. The efficacy and safety of TCAs for PDPN are dose related.5,12 Many elderly patients cannot tolerate the anticholinergic side effects (i.e., dry mouth, blurred vision, constipation, urinary retention) of TCAs at the recommended doses for PDPN and often do not get adequate relief.2 Amitriptyline has been the most studied and used TCA for PDPN.5 The American Geriatrics Society, however, recommends that amitriptyline not be used in older persons, and it has been designated as potentially inappropriate for use in persons aged 65 years or older due to its strong anticholinergic and sedative properties.14,15
Because there are few drug classes to treat PDPN, elderly patients with PDPN often use TCAs or other inappropriate pain medications. A 2000 retrospective study of more than 20,000 patients age 65 or older found 50% of the patients received at least one inappropriate pain medication for a painful neuropathic disorder, including propoxyphene (26.7%) and amitriptyline (10.2%). Use increased with age and was more common in females, but due to the retrospective nature of the study, the investigators could not determine if the benefits of these drugs outweighed the risks in this elderly population.16
A retrospective study of TCAs for PDPN in patients 65 years and older was conducted between 1999 and 2001 to assess the prevalence of comorbidities and/or concurrent medications that might render TCAs inappropriate for elderly patients. TCAs were prescribed to 20% of the 1,749 patients included. Of the TCAs used, amitriptyline was prescribed 84% of the time, nortriptyline 14%, and imipramine 2%. Forty-eight percent of patients treated with a TCA had a potentially inappropriate use for it, including contraindications (0.3%), warnings (42.7%), and potential drug interactions (7.7%) listed in their package inserts. The average TCA dose in this study was 23 mg daily.12
Venlafaxine (Effexor) has also been studied for the treatment of PDPN.17 It is a potent inhibitor of neuronal serotonin reuptake, and at higher doses it also inhibits norepinephrine reuptake. The drug is thought to decrease pain centrally for PDPN, and its NNT is 5.5 (95% CI, 3.4-14).5
Adverse events are less frequent with venlafaxine compared to TCAs, and the most common include sleep disturbance (27%-46%), headache (25%-34%), and nausea (21%-58%). Venlafaxine has the potential to increase blood pressure and heart rate, so these vital signs should be monitored. A contraindication to venlafaxine is concurrent MAOI use. Venlafaxine is metabolized by and weakly inhibits CYP450 2D6 and 3A4.13
Duloxetine (Cymbalta) has also been studied for the treatment of PDPN and was the first FDA-approved agent in its treatment.18,19 It is a balanced inhibitor of both serotonin and norepinephrine transporters. Its mechanism for PDPN is unknown, and its NNT is 4 (95% CI, 3-9).5
Common adverse events include sleep disturbances (15%-26%), nausea (4%-22%), dizziness (1%-14%), and headache (13%). Contraindications include concurrent MAOI use and the presence of narrow-angle glaucoma. Duloxetine should be avoided in patients with hepatic impairment, substantial alcohol use, or creatinine clearance less than 30 mL/min. It is metabolized by CYP450 1A2 and 2D6 and moderately inhibits 2D6. Monitoring parameters include blood pressure, heart rate, serum creatinine, and liver enzymes.13
When choosing an antidepressant for PDPN, cost should also be considered. Extended-release venlafaxine and duloxetine both have a retail cost over $100 per month, while generic TCAs have a retail cost less than $20 per month.20
Selective serotonin reuptake inhibitors (SSRIs) have been studied for the treatment of PDPN and have not been proven effective.5
Antiepileptics: There are several antiepileptic agents that have been studied for the treatment of PDPN. See TABLE 1 for recommended doses, time to effect, and generic availability of antiepileptics used in the treatment of PDPN.
The proposed mechanism of action of carbamazepine (CBZ) for PDPN is its peripheral blockade of sodium channels on the A-gamma nerve fibers. Its NNT is 2.3 (95% CI, 1.6-3.9).5
CBZ can cause serious adverse events, such as aplastic anemia. More typical side effects include dizziness (44%), sedation (32%), nausea (29%), and headache (22%). Contraindications include TCA hypersensitivity, bone marrow suppression, recent use of MAOIs, and pregnancy. Because CBZ is a substrate (3A4) and inducer (1A2, 2C9, 2C19, 3A4) of CYP450 enzymes, it has the potential for many drug interactions. Common medications that would be affected by concurrent administration include theophylline and warfarin. Complete blood counts (CBCs) should be monitored routinely.13
Gabapentin (Neurontin) also acts peripherally to decrease pain perception in PDPN. The NNT for gabapentin at doses equal to or greater than 2,400 mg daily for PDPN is 3.9 (95% CI, 3.2-5.1).5
Gabapentin's most common side effects include sedation (20%), dizziness (17%-28%), ataxia (13%), and fatigue (11%). It is not hepatically metabolized, so there are no major CYP450 drug interactions. Gabapentin should be used cautiously in patients with severe kidney dysfunction. Patients on other antiepileptic drugs should have serum drug levels monitored periodically.13
Lamotrigine (Lamictal) is another antiepileptic medication that works peripherally and acts as a sodium channel blocker. Its NNT for PDPN is 4 (95% CI , 2.1-42).5
Lamotrigine's common side effects include sedation (9%), fatigue (8%), dizziness (7%), nausea (7%-14%), and abnormal vision (2%-5%). It also has rare, serious side effects, such as aplastic anemia and toxic epidermal necrolysis. Patients on other antiepileptic drugs should have serum drug levels monitored periodically.13
Pregabalin (Lyrica) is the second agent approved for the treatment of PDPN. It works peripherally on the gamma-aminobutyric acid (GABA) receptors, which help block the perception of pain. The NNT for pregabalin for PDPN is 4.2 (95% CI, 3.4-5.4).5
Pregabalin has many common side effects, including dizziness (8%-45%), somnolence (4%-28%), ataxia (up to 20%), peripheral edema (up to 16%), weight gain (up to 16%), dry mouth (1%-15%), headache (up to 14%), blurred vision (1%-12%), and tremor (up to 11%). Rare side effects include acute renal failure, rhabdomyolysis, and thrombocytopenia. Angioedema has also been reported. It is contraindicated in heart failure, in hypertension, and with thiazolidinediones (due to potential additive effects on weight gain and edema, especially in patients with prior cardiovascular disease). It is a schedule V drug. Creatine phosphokinase monitoring is recommended because of the risk of rhabdomyolysis.13
Topiramate (Topamax) is a peripherally acting sodium channel blocker, and it acts at the GABA receptor. Its NNT for PDPN is 7.4 (95% CI, 4.3-28).5
Topiramate has many adverse effects, including somnolence (15%-29%), fatigue (9%-30%), dizziness (4%-32%), cognitive slowing (3%-21%), and confusion (4%-14%). It also carries a small risk of kidney stones and closed-angle glaucoma. It is not well tolerated by many patients and is commonly discontinued because of side effects.13
Valproate (Depakene) is an antiepileptic agent that also acts peripherally. Its NNT for PDPN is 2.5 (95% CI, 1.8-4.1).5 Common side effects include sleep disturbances (27%-45%), tremor (19%-57%), nausea (26%-34%), alopecia (13%-24%), and dizziness (13%-18%). It is also associated with thrombocytopenia, pancreatitis, aplastic anemia, and toxic epidermal necrolysis. Contraindications include hepatic dysfunction, urea cycle disorders, pregnancy, and concurrent use of topiramate. Patients taking valproate must have serial monitoring of liver enzymes and CBC. Physicians may also consider monitoring serum drug levels.13
On January 31, 2008, the FDA issued an alert stating that patients on antiepileptics (0.43%) had approximately twice the risk of suicide behavior or ideation compared to patients on placebo (0.22%). There did not seem to be a specific demographic subgroup of patients who were at a higher risk, but the risk was higher in patients with epilepsy compared to patients on an antiepileptic for other conditions. All patients on antiepileptic drugs should be closely monitored for changes in behavior that suggest new or worsening suicidality or depression.21
In addition to safety and efficacy, cost should also be considered if choosing an antiepileptic agent for the elderly. All antiepileptics are available generically, except pregabalin and topiramate.22
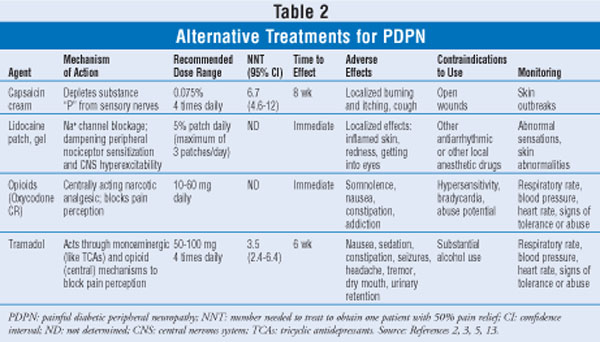
Other Pharmacologic Agents: Other agents (and their characteristics) that can be used for the treatment of PDPN are listed in TABLE 2. The American Society of Pain Educators published consensus guidelines in 2006 for the treatment of PDPN. Its drug recommendations were based on the number of randomized, controlled trials that show efficacy. First-line agents included duloxetine, oxycodone CR, pregabalin, and TCAs. Second-line agents included CBZ, gabapentin, lamotrigine, tramadol, and venlafaxine ER. Topical choices included lidocaine and capsaicin. The topical preparations may be viable options for elderly patients who cannot tolerate the recommended oral medications due to side effects or drug interactions. The complexity of capsaicin application (wearing gloves, four times a day dosing, very painful for first week of use) may limit its use. If an analgesic such as tramadol or an opioid is considered, one must keep in mind the abuse potential. Tramadol has side effects similar to the opioids, such as nausea, constipation, and sedation.23
Other Treatment Options for PDPN
Observational studies support the use of acupuncture for PDPN. One study reported that the benefits of acupuncture lasted up to six months and reduced the use of other analgesics.24
Evidence is available for other treatments such as percutaneous nerve stimulation (TENS units), static magnetic field therapy, low-intensive laser therapy, monochromatic infrared light therapy, and electrical spinal cord stimulation.25-29
There are many case reports that show the benefits of neurofeedback/biofeedback in pain relief. Experts state that elderly patients find biofeedback useful for the treatment of diabetic neuropathy, intermittent claudication, and arthritis. One case series reported that chronic pain can be reduced through electroencephalogram biofeedback.30,31
Pharmacist's Role
All patients with neuropathy should be considered at risk of a foot ulcer and require frequent foot exams, education in routine foot care, and regular podiatric assessments.32 Pharmacists can provide patients with information regarding the essentials of good foot care. See TABLE 3 for do's and don'ts of foot care. Counseling points regarding drug therapy should include potential side effects and when to expect benefit.

Conclusion
Up to 50% of elderly patients with T2DM have PN. Strict glycemic control alone improves PDPN in many patients. When drug treatment is needed, TCAs are more efficacious and less expensive than most other options, but their side effects make them an unfavorable choice in the elderly. If a TCA is prescribed, a secondary amine, such as desipramine, should be recommended. With Medicare Part D, elderly patients can more easily afford medications, such as duloxetine and pregabalin. These two medications are approved for PDPN and lack anticholinergic side effects. They do, however, require dose reduction in renal impairment. Neurofeedback is the only alternative nondrug therapy that has shown benefit in reducing neuropathic pain in the elderly; however, finding clinics that provide this type of therapy and that accept insurance or Medicare can be problematic. The best treatment for PDPN is prevention, which includes good glycemic, blood pressure, and lipid control; daily low-dose aspirin; moderate alcohol consumption; a healthy diet; and good foot care.
REFERENCES
1. Davies M, Brophy S, Williams R, Taylor A. The prevalence, severity, and impact of painful diabetic peripheral neuropathy in type 2 diabetes. Diabetes Care. 2006;29:1518-1522.
2. Boulton AJ. Management of diabetic peripheral neuropathy. Clinical Diabetes. 2005;23:9-15.
3. Boulton AJ, Vinik AI, Arezzo JC, et al. Diabetic neuropathies. A statement by the American Diabetes Association. Diabetes Care. 2005;28:956-962.
4. Gordois A, Scuffham P, Shearer A, et al. The health care costs of diabetic peripheral neuropathy in the U.S. Diabetes Care. 2003;26:1790-1795.
5. Huizinga MM, Peltier A. Painful diabetic neuropathy: a management-centered review. Clinical Diabetes. 2007;23:6-15.
6. Diabetes Control and Complications Trial Research Group. The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. N Engl J Med. 1993;329:977-986.
7. Belgrade MJ, Cole BE, McCarberg BH, McLean MJ. Diabetic peripheral neuropathic pain: case studies. Mayo Clin Proc. 2006;81(suppl 4):S26-S32.
8. Rull JA, Quibrera R, Gonzalez-Millan H, et al. Symptomatic treatment of peripheral diabetic neuropathy with carbamazepine (Tegretol): double blind crossover trial. Diabetologia. 1969;5:215-218.
9. Davis JL, Lewis SB, Gerich JE, et al. Peripheral diabetic neuropathy treated with amitriptyline and fluphenazine. JAMA. 1977;238:2291-2292.
10. Cymbalta (duloxetine) package insert. Indianapolis, IN: Eli Lilly & Co; 2006.
11. Lyrica (pregabalin) package insert. New York, NY: Pfizer Inc; 2006.
12. Berger A, Dukes E, Edelsberg J, et al. Use of tricyclic antidepressants in older patients with diabetic peripheral neuropathy. Clin J Pain. 2007;23:251-258.
13. Micromedex Healthcare Series [intranet database]. Version 5.1. Greenwood Village, CO: Thomson Healthcare; 2008.
14. American Geriatrics Society. The management of persistent pain in older persons: AGS panel on persistent pain in older persons. JAGS. 2002;50:S205-S224.
15. Fick DM, Cooper JW, Wade WE, et al. Updating the Beers criteria for potentially inappropriate medication use in older adults: results of a US consensus panel of experts. Arch Intern Med. 2003;163:2716-2724.
16. Oster G, Berger A, Dukes E, et al. Use of potentially inappropriate pain-related medications in older adults with painful neuropathic disorders. Am J Geriatr Pharmacother. 2004;2:163-170.
17. Rowbotham MC, Goli V, Kunz NR, Lei D. Venlafaxine extended release in the treatment of painful diabetic neuropathy: a double-blind, placebo-controlled study. Pain. 2004;110:697-706.
18. Goldstein DJ, Lu Y, Detke MJ, et al. Duloxetine vs. placebo in patients with painful diabetic neuropathy. Pain. 2005;116:109-118.
19. Raskin JR, Pritchett YL, Wang F, et al. A double-blind, randomized multicenter trial comparing duloxetine with placebo in the management of diabetic peripheral neuropathic pain. Pain Medicine. 2005;6:346-356.
20. Retail prices found at www.drugstore.com. Accessed May 14, 2008.
21. U.S. Food and Drug Administration. Suicidality and antiepileptic drugs. www.fda.gov/cder/drug/
22. Lexi-Comp/UptoDate [intranet database]. Online Version 16.1. Waltham, MA: UptoDate, Inc; 2008.
23. Argoff CE, Backonia MM, Belgrade MJ, et al. Consensus guidelines: treatment planning and options. Mayo Clin Proc. 2006:81(suppl 4):S12-S25.
24. Abusaisha BB, Constanzi JB, Boulton AJ. Acupuncture for the treatment of chronic painful diabetic neuropathy: a long-term study. Diabetes Res Clin Pract. 1998;39:115-121.
25. Hamza MA, White PF, Craig WF, et al. Percutaneous electrical nerve stimulation: a novel analgesic therapy for diabetic neuropathic pain. Diabetes Care. 2000;23:365-370.
26. Weintraub MI, Wolfe GI, Barohn RA, et al. Static magnetic field therapy for symptomatic diabetic neuropathy: a randomized double-blind placebo-controlled trial. Arch Phys Med Rehabil. 2003;86:736-746.
27. Zinman LH, Ngo M, Ng ET, et al. Low-intensity laser therapy for painful symptom diabetic sensorimotor polyneuropathy: a controlled trial. Diabetes Care. 2004;27;921-924.
28. Leonard DR, Farooqui MH, Myers S. Restoration of sensation, reduced pain, and improved balance in subjects with diabetic peripheral neuropathy: a double-blind placebo-controlled study with monochromatic infrared treatment. Diabetes Care. 2004;27:168-172.29. Tesfaye S, Watt J, Benbow SJ, et al. Electrical spinal-cord stimulation for painful-diabetic peripheral neuropathy. Lancet. 1996;348:1696-1701.
30. Your Total Health. Biofeedback & Pain Management. 2008. http://yourtotalhealth.
31. WebMD. Biofeedback to relieve pain from peripheral neuropathy. 2008. http://diabetes.webmd.com/peripheral-neuropathy-
32. American Diabetes Association. Preventative foot care in diabetes. Diabetes Care. 2004;27:S63-S64.





